Pro1tek Hand Sanitizer by Priority Environmental Solutions Inc. Pro1tek Hand Sanitizer
Pro1tek Hand Sanitizer by
Drug Labeling and Warnings
Pro1tek Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by Priority Environmental Solutions Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
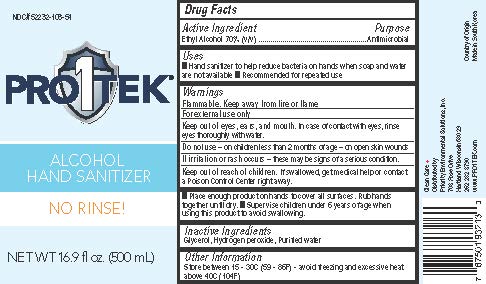
PRO1TEK HAND SANITIZER- ethyl alcohol 70% gel
Priority Environmental Solutions Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Pro1tek Hand Sanitizer
Uses
Hand Sanitizer to help reduce bacteria on hands when soap and water are not available. Recommended for repeated use
When using this product
Keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor.
If irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
| PRO1TEK HAND SANITIZER
ethyl alcohol 70% gel |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Priority Environmental Solutions Inc. (827067880) |
| Registrant - Priority Environmental Solutions Inc. (827067880) |