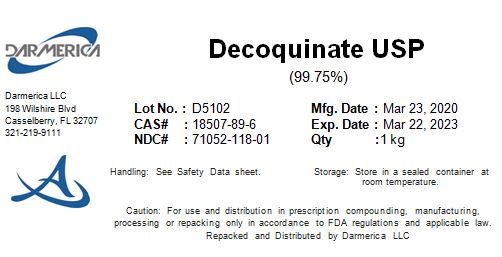
Decoquinate by DARMERICA, LLC Decoquinate
Decoquinate by
Drug Labeling and Warnings
Decoquinate by is a Other medication manufactured, distributed, or labeled by DARMERICA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DECOQUINATE- decoquinate powder
DARMERICA, LLC
----------
Decoquinate
| DECOQUINATE
decoquinate powder |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - DARMERICA, LLC (080233052) |
Revised: 12/2021
Document Id: d2a605d0-0fc0-5a16-e053-2995a90ab6b6
Set id: a70d1270-001f-6de2-e053-2a95a90adb60
Version: 2
Effective Time: 20211208
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.