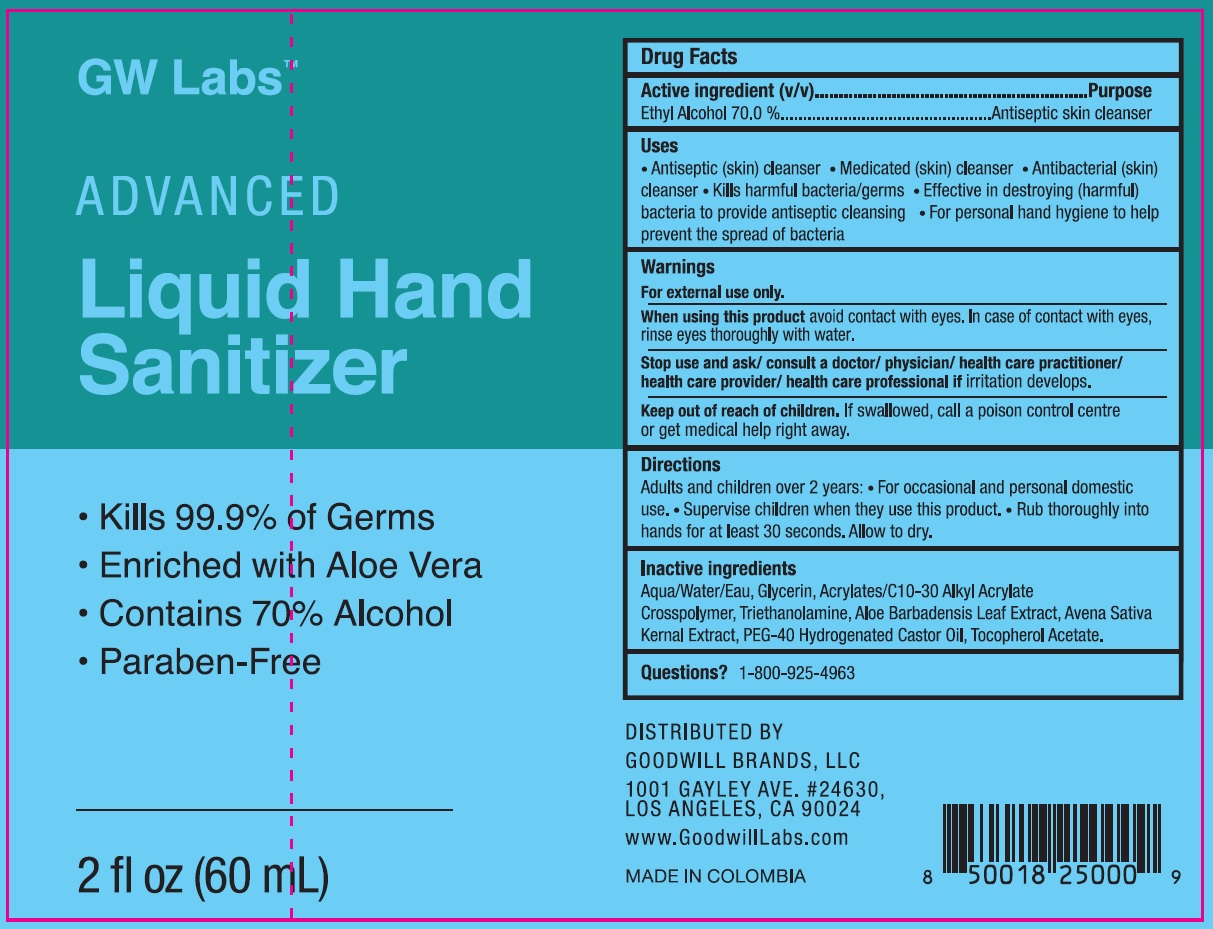
Goodwill Labs Advanced Hand Sanitizer
Goodwill Labs Advanced Hand Sanitizer by
Drug Labeling and Warnings
Goodwill Labs Advanced Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by Goodwill Brands, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
GOODWILL LABS ADVANCED HAND SANITIZER- alcohol gel
Goodwill Brands, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Goodwill Labs Advanced Hand Sanitizer
Uses
- Antiseptic (skin) cleanser
- Medicated (skin) cleanser
- Antibacerial (skin) cleanser
- Kills harmful bacteria/germs
- Effective in destroying (harmful) bacteria to provide antiseptic cleansing
- For personal hand hygiene to help prevent the spread of bacteria
Warnings
For external use only.
When using this product
avoid contact with eyes. In case of contact with eyes, rinse eyes thoroughly with water.
Directions
Adults and children over 2 years:
- For occasional and personal domestic use.
- Supervise children when they use this product.
- Rub thoroughly into hands for at least 30 seconds. Allow to dry.
| GOODWILL LABS ADVANCED HAND SANITIZER
alcohol gel |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Goodwill Brands, LLC (130677488) |
Revised: 2/2022
Document Id: d850b2ca-3ef1-2747-e053-2995a90aa79e
Set id: a7e8d5a3-b0d3-1d70-e053-2a95a90aa435
Version: 2
Effective Time: 20220218
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.