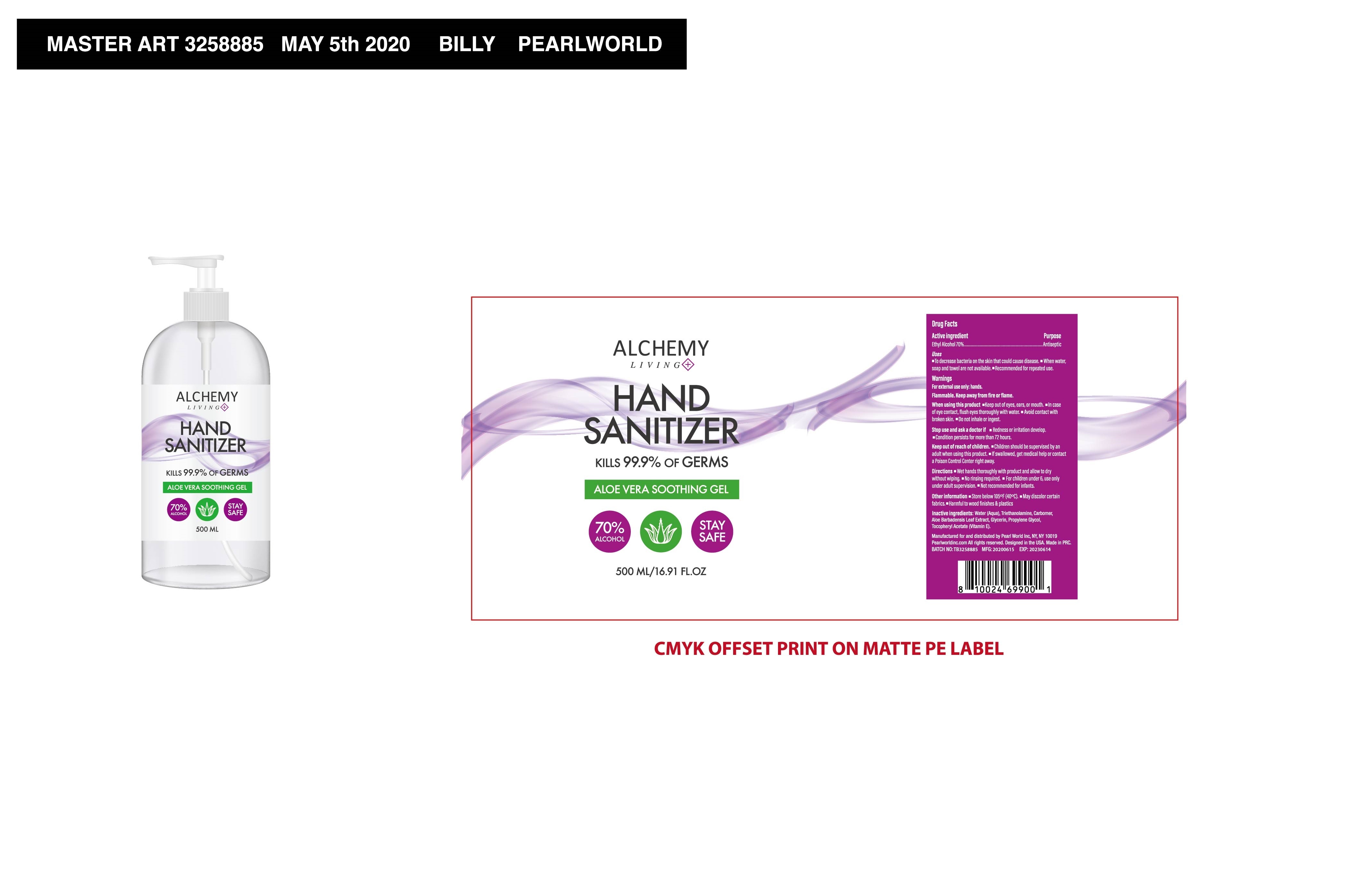
006 Alchemy Living Hand Sanitizer
Alchemy Living Hand Sanitizer by
Drug Labeling and Warnings
Alchemy Living Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by COSBE LABORA TORY INC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ALCHEMY LIVING HAND SANITIZER- alchemy living hand sanitizer gel
COSBE LABORA TORY INC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
006 Alchemy Living Hand Sanitizer
Use
To decrease bacteria on the skin that could cause disease. When water, soap and towel are not available. Recommended for repeated use.
When using this product
When using this product Keep out of eyes, ears, or mouth. -In case of eye contact, flush eyes thoroughly with water. Avoid contact with broken skin, Do not inhale or ingest
Stop use and ask a doctor if Redness or irritation develop.
Condition persists for more than 72 hours
Keep out of reach of children
Keep out of reach of children. Children should be supervised by an adult when using this product. lf swallowed, get medical help or contact a Poison Control Center right away
Directions
Wet hands thoroughly with product and allow to dry without wiping. No rinsing required. For children under 6, use only under adult supervision. Not recommended for infants.
Other information
Store below 105°F(40°C). May discolor certain fabrics, Harmful to wood finishes & plastics
| ALCHEMY LIVING HAND SANITIZER
alchemy living hand sanitizer gel |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - COSBE LABORA TORY INC. (543033650) |
| Registrant - COSBE LABORA TORY INC. (543033650) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| COSBE LABORA TORY INC. | 543033650 | manufacture(74620-006) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.