K2L COMMERCE - Dramaholic Hand Sanitizer
Dramaholic Hand Sanitizer by
Drug Labeling and Warnings
Dramaholic Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by DONG IL PHARMS CO., LTD.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
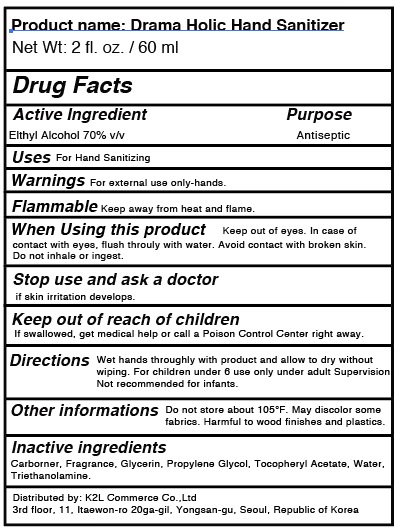
DRAMAHOLIC HAND SANITIZER- alcohol gel
DONG IL PHARMS CO., LTD.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
K2L COMMERCE - Dramaholic Hand Sanitizer
Do not use on the following body parts.
Around the eyes and ears, in the oral cavity, a wide range of body parts and damaged skin (may have irritating effects)
If the following symptoms appear, stop using them immediately and consult a doctor or pharmacist.
Symptoms of skin irritation
Other precautions
For external use only
Be careful not to get into your eyes, and if so, rinse well with clean water and consult a doctor or pharmacist.
Be careful not to inhale vapor when using it extensively or for a long time. (If you drink ethanol vapor in large quantities or repeatedly, irritation to the mucous membrane, headache, etc. may occur).
When repeated use on the same site, be careful as the skin may become rough due to degreasing.
When used in sealed bandages, cast bandages, packs, etc., irritation symptoms may appear.
Do not use for any other purpose.
| DRAMAHOLIC HAND SANITIZER
alcohol gel |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - DONG IL PHARMS CO., LTD. (557810721) |
| Registrant - DONG IL PHARMS CO., LTD. (557810721) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| DONG IL PHARMS CO., LTD. | 557810721 | manufacture(73242-0200) | |