DOCOSANOL by H2-Pharma, LLC DOCOSANOL
DOCOSANOL by
Drug Labeling and Warnings
DOCOSANOL by is a Otc medication manufactured, distributed, or labeled by H2-Pharma, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DOCOSANOL- docosanol cream
H2-Pharma, LLC
----------
DOCOSANOL
Uses
- treats cold sores/fever blisters on the face or lips
- shortens healing time and duration of symptoms:
- tingling, pain, burning, and/or itching
Warnings
Allergy Alert
This product may cause a severe allergic reaction. Symptoms may include:
- hives
- facial swelling
- wheezing/difficulty breathing
- shock
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
For external use only
When using this product
- apply only to the affected areas
- do not use in or near the eyes
- avoid applying directly inside your mouth
- do not share this product with anyone. This may spread the infection.
Directions
-
adults and children 12 years or over:
- wash hands before and after applying cream
- apply to affected area on the face or lips at the first sign of cold sore/fever blister (tingle)
- early treatment ensures the best results
- rub in gently but completely
- use 5 times a day until healed
- children under 12 years: ask a doctor
Inactive ingredients
benzyl alcohol, light mineral oil, propylene glycol, purified water, sucrose distearate, sucrose stearate
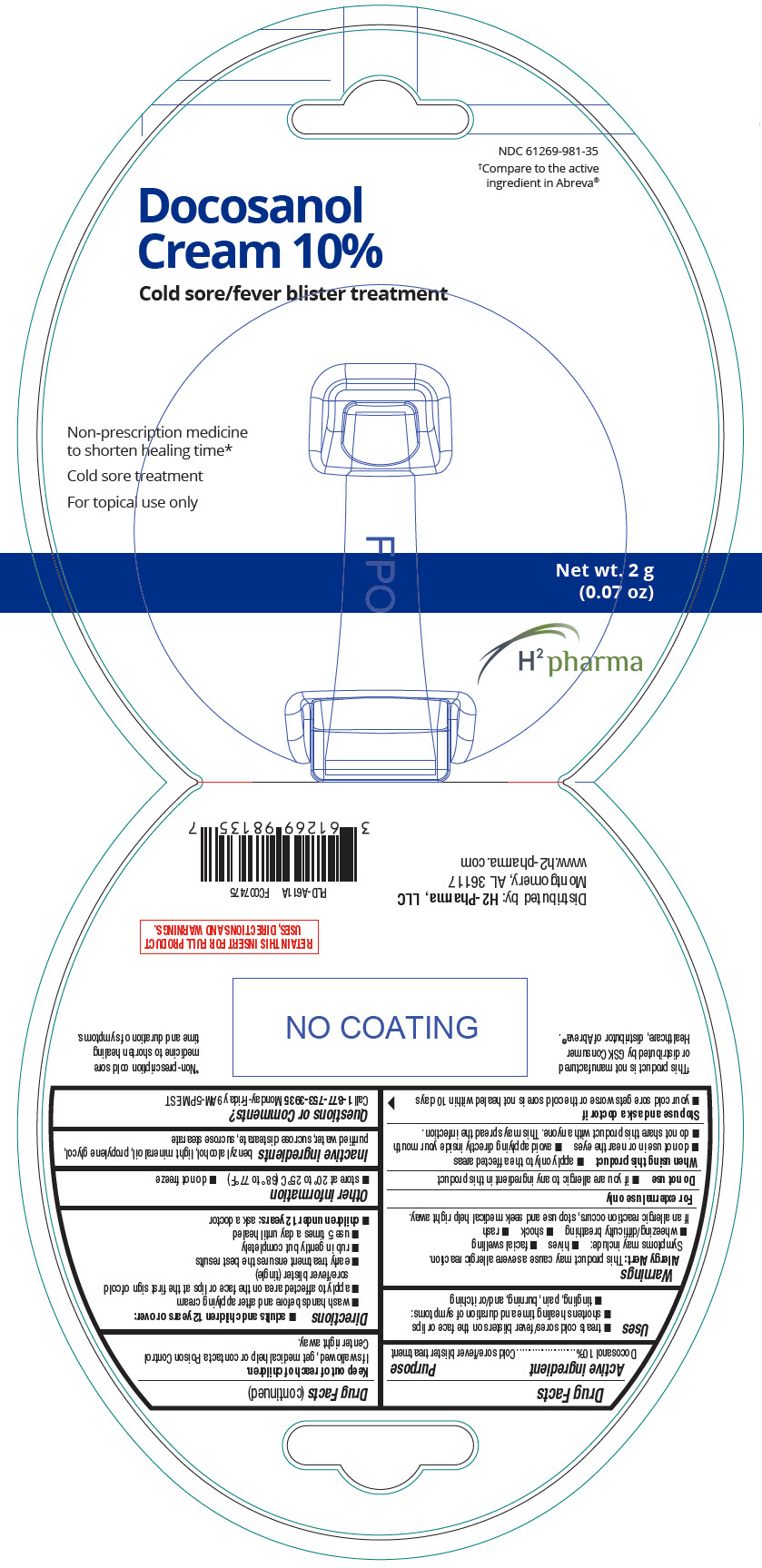
PRINCIPAL DISPLAY PANEL - 2 g Tube Package
NDC: 61269-981-35
†Compare to the active
ingredient in Abreva®
Docosanol
Cream 10%
Cold sore/fever blister treatment
Non-prescription medicine
to shorten healing time*
Cold sore treatment
For topical use only
Net wt. 2 g
(0.07 oz)
H2 pharma
| DOCOSANOL
docosanol cream |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - H2-Pharma, LLC (028473634) |
Revised: 4/2025
Document Id: a702c716-68dd-4a12-b584-44d31dfd84e7
Set id: a921a653-aedd-43c2-b84f-429a4aada79a
Version: 3
Effective Time: 20250411
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.