NERVE PAIN- arabica coffee bean, silicon dioxide, and hypericum perforatum solution/ drops
Nerve Pain by
Drug Labeling and Warnings
Nerve Pain by is a Homeopathic medication manufactured, distributed, or labeled by Forces of Nature. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Also Contains
- Indications
- Directions
-
Warnings
Some individuals may be sensitive to essential oils. Begin with a small drop to determine if the product causes any increased redness or irritation and discontinue use if it irritates your skin.
For external use only. If accidental ingestion of more than several drops, and adverse reaction occurs, get medical help or contact a Poison Control Center. Do not apply to the eyes. If you are pregnant or breast - feeding, ask a health professional before use. Not for use on children under the age of 3.
- SPL UNCLASSIFIED SECTION
-
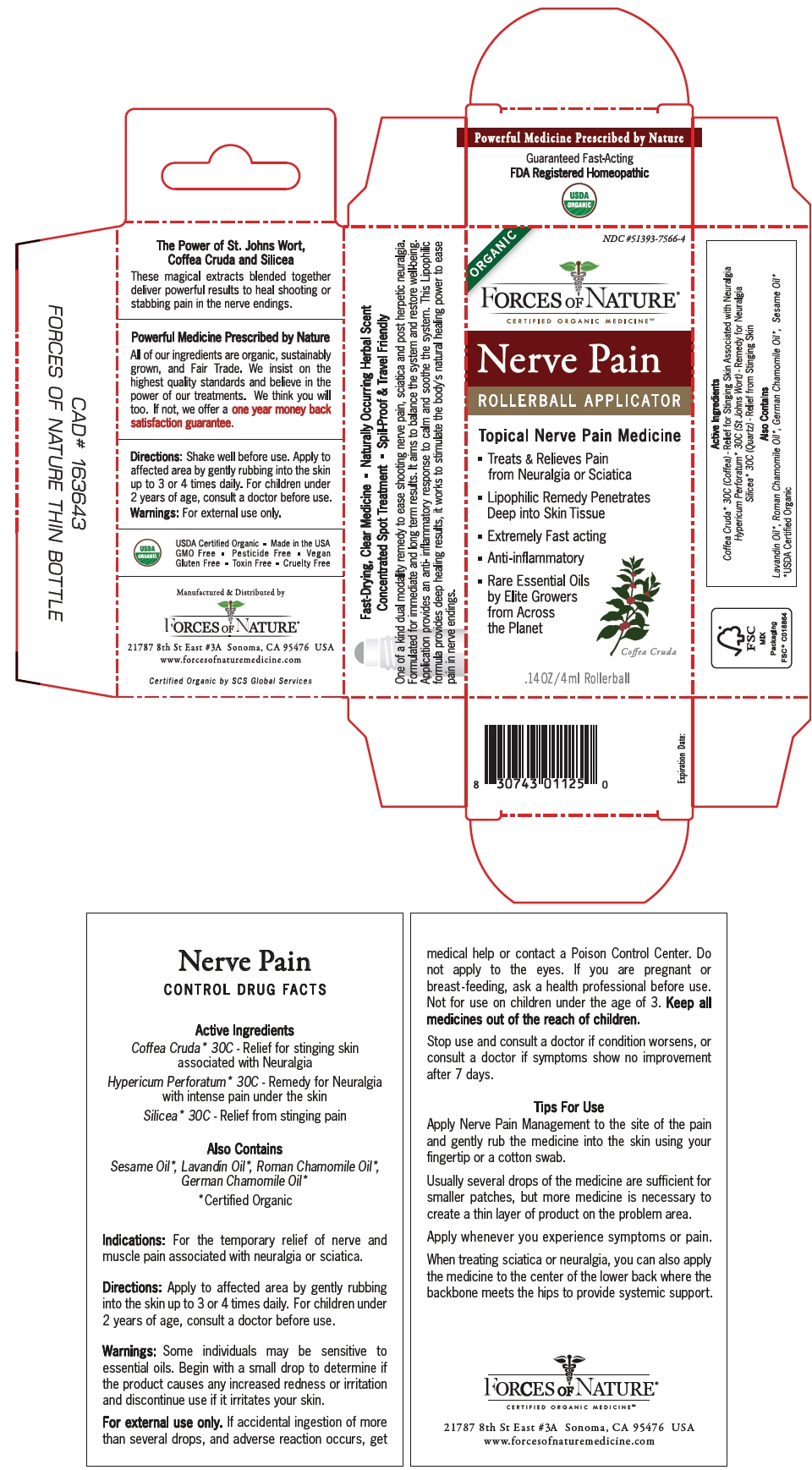
PRINCIPAL DISPLAY PANEL - 4 ml Bottle Box
ORGANIC
NDC #51393-7566-4
FORCES OF NATURE®
CERTIFIED ORGANIC MEDICINESMNerve Pain
ROLLERBALL APPLICATORTopical Nerve Pain Medicine
- Treats & Relieves Pain
from Neuralgia or Sciatica - Lipophilic Remedy Penetrates
Deep into Skin Tissue - Extremely Fast acting
- Anti-inflammatory
- Rare Essential Oils
by Elite Growers
from Across
the Planet
Coffea Cruda
.14 OZ/4 ml Rollerball
- Treats & Relieves Pain
-
INGREDIENTS AND APPEARANCE
NERVE PAIN
arabica coffee bean, silicon dioxide, and hypericum perforatum solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51393-7566 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Arabica Coffee Bean (UNII: 3SW678MX72) (Arabica Coffee Bean - UNII:3SW678MX72) Arabica Coffee Bean 30 [hp_C] in 100 mL Silicon Dioxide (UNII: ETJ7Z6XBU4) (Silicon Dioxide - UNII:ETJ7Z6XBU4) Silicon Dioxide 30 [hp_C] in 100 mL Hypericum Perforatum (UNII: XK4IUX8MNB) (Hypericum Perforatum - UNII:XK4IUX8MNB) Hypericum Perforatum 30 [hp_C] in 100 mL Inactive Ingredients Ingredient Name Strength Lavandula Dentata Whole (UNII: ISP4E5T96U) Chamaemelum Nobile (UNII: 7NF3GE7IWR) Chamaemelum Nobile Flower (UNII: O2T154T6OG) Sesame Oil (UNII: QX10HYY4QV) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51393-7566-1 1 in 1 BOX 01/01/2013 1 11 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product 2 NDC: 51393-7566-2 1 in 1 BOX 01/01/2013 2 33 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product 3 NDC: 51393-7566-4 1 in 1 BOX 07/01/2016 3 4 mL in 1 BOTTLE, DISPENSING; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED HOMEOPATHIC 01/01/2013 Labeler - Forces of Nature (050169130)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.