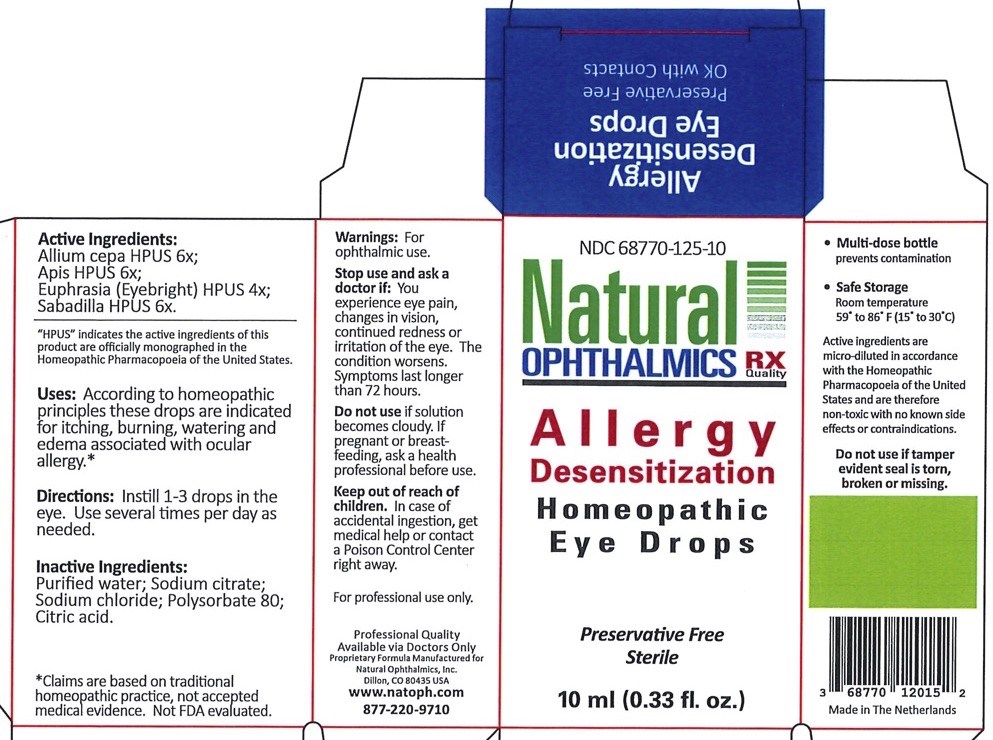
Allergy Desensitization by Natural Ophthalmics, Inc Allergy Desensitization
Allergy Desensitization by
Drug Labeling and Warnings
Allergy Desensitization by is a Homeopathic medication manufactured, distributed, or labeled by Natural Ophthalmics, Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ALLERGY DESENSITIZATION- onion, apis mellifera, schoenocaulon officinale seed, euphrasia stricta liquid
Natural Ophthalmics, Inc
Disclaimer: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
----------
Allergy Desensitization
Homeopathic Purpose
| Allium cepa | 6x | Relieves itching, burning |
| Apis | 6x | Relieves inflammation with burning and stinging |
| Sabadilla | 6x | Relieve hay fever type symptoms |
| Euphrasia (Eye-bright) | 4x | General dryness, redness and irritation of the eye and lids |
Uses
According to homeopathic principles these drops are indicated for itching, burning, watering and edema associated with ocular allergy.*
Do not use
Do not use if solution becomes cloudy. If pregnant or breastfeeding, ask a health professional before use.
| ALLERGY DESENSITIZATION
onion, apis mellifera, schoenocaulon officinale seed, euphrasia stricta liquid |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Natural Ophthalmics, Inc (118039333) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Altaire Pharmaceuticals Inc. | 786790378 | manufacture(68770-125) | |
Revised: 12/2025
Document Id: 46aa4b91-bad0-b697-e063-6294a90aa229
Set id: a9ceae40-0ca7-12d0-e053-2a95a90adb78
Version: 3
Effective Time: 20251223