Meloxicam by ReadyMeds MELOXICAM tablet
Meloxicam by
Drug Labeling and Warnings
Meloxicam by is a Prescription medication manufactured, distributed, or labeled by ReadyMeds. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use meloxicam tablets safely and effectively. See full prescribing information for meloxicam tablets.
Meloxicam Tablets, USP
Initial U.S. Approval: 2000WARNING: CARDIOVASCULAR and GASTROINTESTINAL RISKS
See full prescribing information for complete boxed warning.
Cardiovascular Risk
- NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk. (5.1)
- Meloxicam is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery. (4.2, 5.1)
Gastrointestinal Risk- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (5.2)
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
- Tablets: 7.5 mg, 15 mg (3)
CONTRAINDICATIONS
- Known hypersensitivity (e.g., anaphylactoid reactions and serious skin reactions) to meloxicam (4.1)
- History of asthma, urticaria or other allergic-type reactions after taking aspirin or other NSAIDs (4.1)
- Use during the peri-operative period in the setting of coronary artery bypass graft (CABG) surgery (4.2)
WARNINGS AND PRECAUTIONS
- Serious and potentially fatal cardiovascular (CV) thrombotic events, myocardial infarction and stroke. Patients with known CV disease/risk factors may be at greater risk. (5.1)
- Serious gastrointestinal (GI) adverse events which can be fatal. The risk is greater in patients with a prior history of ulcer disease or GI bleeding and in patients at higher risk for GI events, especially the elderly. (5.2)
- Elevated liver enzymes, and rarely, severe hepatic reactions. Discontinue use immediately if abnormal liver enzymes persist or worsen. (5.3)
- New onset or worsening of hypertension. Blood pressure should be monitored closely during treatment. (5.4)
- Fluid retention and edema. Should be used with caution in patients with fluid retention or heart failure. (5.5)
- Renal papillary necrosis and other renal injury with long-term use. Use with caution in the elderly, those with impaired renal function, heart failure, liver dysfunction and those taking diuretics, ACE-inhibitors or angiotensin II antagonists. The use of meloxicam in patients with severe renal impairment is not recommended. (5.6)
- Serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN), which can be fatal and can occur without warning. Discontinue meloxicam at first appearance of rash or skin reactions. (5.8)
ADVERSE REACTIONS
- Most common (≥ 5% and greater than placebo) adverse events in adults are diarrhea, upper respiratory tract infections, dyspepsia and influenza-like symptoms (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Mylan Pharmaceuticals Inc. at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Concomitant use of meloxicam and warfarin may result in increased risk of bleeding complications (7.7)
- Concomitant use of meloxicam and aspirin is not generally recommended because of the potential of increased adverse effect including increased GI bleeding (7.2)
- Concomitant use with meloxicam increases lithium plasma levels (7.4)
- Concomitant use with NSAIDs may reduce the antihypertensive effect of ACE-inhibitors (7.1)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2014
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISK OF SERIOUS CARDIOVASCULAR and GASTROINTESTINAL EVENTS
1 INDICATIONS AND USAGE
1.1 Osteoarthritis (OA)
1.2 Rheumatoid Arthritis (RA)
2 DOSAGE AND ADMINISTRATION
2.1 General Instructions
2.2 Osteoarthritis
2.3 Rheumatoid Arthritis
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 Allergic Reactions
4.2 Coronary Surgery
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Thrombotic Events
5.2 Gastrointestinal (GI) Effects - Risk of GI Ulceration, Bleeding and Perforation
5.3 Hepatic Effects
5.4 Hypertension
5.5 Congestive Heart Failure and Edema
5.6 Renal Effects
5.7 Anaphylactoid Reactions
5.8 Adverse Skin Reactions
5.9 Pregnancy
5.10 Corticosteroid Treatment
5.11 Masking of Inflammation and Fever
5.12 Hematological Effects
5.13 Use in Patients with Preexisting Asthma
5.14 Monitoring
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-Marketing Experience
7 DRUG INTERACTIONS
7.1 ACE-Inhibitors
7.2 Aspirin
7.3 Diuretics
7.4 Lithium
7.5 Methotrexate
7.6 Cyclosporine
7.7 Warfarin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
8.8 Females of Reproductive Potential
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Osteoarthritis and Rheumatoid Arthritis
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Medication Guide
17.2 Cardiovascular Effects
17.3 Gastrointestinal Effects
17.4 Hepatotoxicity
17.5 Adverse Skin Reactions
17.6 Weight Gain and Edema
17.7 Anaphylactoid Reactions
17.8 Effects During Pregnancy
17.9 Effects On Female Fertility
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISK OF SERIOUS CARDIOVASCULAR and GASTROINTESTINAL EVENTS
Cardiovascular Risk
- Non-steroidal anti-inflammatory drugs (NSAIDs) may cause an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk [see Warnings and Precautions (5.1)].
- Meloxicam is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4.2) and Warnings and Precautions (5.1)].
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse reactions including bleeding, ulceration and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
1.1 Osteoarthritis (OA)
Meloxicam tablets are indicated for relief of the signs and symptoms of osteoarthritis [see Clinical Studies (14.1)].
1.2 Rheumatoid Arthritis (RA)
Meloxicam tablets are indicated for relief of the signs and symptoms of rheumatoid arthritis [see Clinical Studies (14.1)].
-
2 DOSAGE AND ADMINISTRATION
2.1 General Instructions
Carefully consider the potential benefits and risks of meloxicam tablets and other treatment options before deciding to use meloxicam tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5.4)].
After observing the response to initial therapy with meloxicam tablets, adjust the dose to suit an individual patient's needs.
In adults, the maximum recommended daily oral dose of meloxicam tablets is 15 mg regardless of formulation. In patients with hemodialysis, a maximum daily dosage of 7.5 mg is recommended [see Warnings and Precautions (5.6), Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Meloxicam tablets may be taken without regard to timing of meals.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
4.1 Allergic Reactions
Meloxicam tablets are contraindicated in patients with known hypersensitivity (e.g., anaphylactoid reactions and serious skin reactions) to meloxicam.
Meloxicam should not be given to patients who have experienced asthma, urticaria or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7, 5.13)].
4.2 Coronary Surgery
Meloxicam tablets are contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to 3 years’ duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction and stroke, which can be fatal. All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with an NSAID, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV events and the steps to take if they occur.
Two large, controlled, clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10 to 14 days following CABG surgery found an increased incidence of myocardial infarction and stroke [see Contraindications (4.2)].
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID does increase the risk of serious GI events [see Warnings and Precautions (5.2)].
5.2 Gastrointestinal (GI) Effects - Risk of GI Ulceration, Bleeding and Perforation
NSAIDs, including meloxicam, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration and perforation of the stomach, small intestine or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding or perforation caused by NSAIDs, occur in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. These trends continue with longer duration of use, increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk.
Prescribe NSAIDs, including meloxicam, with extreme caution in those with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease and/or gastrointestinal bleeding who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore, special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event in patients treated with an NSAID, use the lowest effective dose for the shortest possible duration. Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during meloxicam therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of meloxicam until a serious GI adverse event is ruled out. For high-risk patients, consider alternate therapies that do not involve NSAIDs.
5.3 Hepatic Effects
Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including meloxicam. These laboratory abnormalities may progress, may remain unchanged or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported [see Adverse Reactions (6.1)].
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with meloxicam. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue meloxicam [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.4 Hypertension
NSAIDs, including meloxicam, can lead to onset of new hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. NSAIDs, including meloxicam, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
Patients taking ACE inhibitors, thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs.
5.5 Congestive Heart Failure and Edema
Fluid retention and edema have been observed in some patients taking NSAIDs. Use meloxicam with caution in patients with fluid retention, hypertension or heart failure.
5.6 Renal Effects
Long-term administration of NSAIDs, including meloxicam, can result in renal papillary necrosis, renal insufficiency, acute renal failure and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a non-steroidal anti-inflammatory drug may cause a dose dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics, ACE-inhibitors, and angiotensin II receptor antagonists and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
A pharmacokinetic study in patients with mild and moderate renal impairment revealed that no dosage adjustments in these patient populations are required. Patients with severe renal impairment have not been studied. The use of meloxicam in patients with severe renal impairment with CrCl less than 20 mL/min is not recommended. A study performed in patients on hemodialysis revealed that although overall Cmax was diminished in this population, the proportion of free drug not bound to plasma was increased. Therefore it is recommended that meloxicam dosage in this population not exceed 7.5 mg per day. Closely monitor the renal function of patients with impaired renal function who are taking meloxicam [see Dosage and Administration (2.1), Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
Use caution when initiating treatment with meloxicam in patients with considerable dehydration. It is advisable to rehydrate patients first and then start therapy with meloxicam. Caution is also recommended in patients with preexisting kidney disease.
The extent to which metabolites may accumulate in patients with renal impairment has not been studied with meloxicam. Because some meloxicam metabolites are excreted by the kidney, monitor patients with significant renal impairment closely.
5.7 Anaphylactoid Reactions
As with other NSAIDs, anaphylactoid reactions have occurred in patients without known prior exposure to meloxicam. Meloxicam should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs [see Contraindications (4.1) and Warnings and Precautions (5.12)]. Seek emergency help in cases where an anaphylactoid reaction occurs.
5.8 Adverse Skin Reactions
NSAIDs, including meloxicam, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin manifestations and discontinue use of the drug at the first appearance of skin rash or any other sign of hypersensitivity.
5.9 Pregnancy
Starting at 30 weeks gestation, avoid the use of meloxicam because it may cause premature closure of the ductus arteriosus [see Use in Specific Populations (8.1) and Patient Counseling Information (17.8)].
5.10 Corticosteroid Treatment
Meloxicam cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Slowly taper patients on prolonged corticosteroid therapy if a decision is made to discontinue corticosteroids.
5.11 Masking of Inflammation and Fever
The pharmacological activity of meloxicam in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
5.12 Hematological Effects
Anemia may occur in patients receiving NSAIDs, including meloxicam. This may be due to fluid retention, occult or gross GI blood loss or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including meloxicam, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Carefully monitor patients treated with meloxicam who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants.
5.13 Use in Patients with Preexisting Asthma
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm, which can be fatal. Since cross-reactivity, including bronchospasm, between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, meloxicam should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with preexisting asthma.
5.14 Monitoring
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, meloxicam should be discontinued.
-
6 ADVERSE REACTIONS
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The following serious adverse reactions are discussed elsewhere in the labeling:
- Cardiovascular thrombotic events [see Boxed Warning and Warnings and Precautions (5.1)]
- Gastrointestinal effects – risk of GI ulceration, bleeding and perforation [see Boxed Warning and Warnings and Precautions (5.2)]
- Hepatic effects [see Warnings and Precautions (5.3)]
- Hypertension [see Warnings and Precautions (5.4)]
- Congestive heart failure and edema [see Warnings and Precautions (5.5)]
- Renal effects [see Warnings and Precautions (5.6)]
- Anaphylactoid reactions [see Warnings and Precautions (5.7)]
- Adverse skin reactions [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Adults
Osteoarthritis and Rheumatoid Arthritis
The Meloxicam Phase 2/3 clinical trial database includes 10,122 OA patients and 1,012 RA patients treated with meloxicam 7.5 mg/day, 3,505 OA patients and 1,351 RA patients treated with meloxicam 15 mg/day. Meloxicam at these doses was administered to 661 patients for at least 6 months and to 312 patients for at least one year. Approximately 10,500 of these patients were treated in ten placebo- and/or active-controlled osteoarthritis trials and 2,363 of these patients were treated in ten placebo- and/or active-controlled rheumatoid arthritis trials. Gastrointestinal (GI) adverse events were the most frequently reported adverse events in all treatment groups across meloxicam trials.
A 12-week multicenter, double-blind, randomized trial was conducted in patients with osteoarthritis of the knee or hip to compare the efficacy and safety of meloxicam with placebo and with an active control. Two 12-week multicenter, double-blind, randomized trials were conducted in patients with rheumatoid arthritis to compare the efficacy and safety of meloxicam with placebo.
Table 1a depicts adverse events that occurred in ≥ 2% of the meloxicam treatment groups in a 12-week placebo- and active-controlled osteoarthritis trial.
Table 1b depicts adverse events that occurred in ≥ 2% of the meloxicam treatment groups in two 12-week placebo-controlled rheumatoid arthritis trials.
Table 1a Adverse Events (%) Occurring in ≥ 2% of Meloxicam Patients in a 12-Week Osteoarthritis Placebo- and Active-Controlled Trial - * WHO preferred terms edema, edema dependent, edema peripheral and edema legs combined
- † WHO preferred terms rash, rash erythematous and rash maculo-papular combined
Placebo Meloxicam
7.5 mg dailyMeloxicam
15 mg dailyDiclofenac
100 mg dailyNo. of Patients 157 154 156 153 Gastrointestinal 17.2 20.1 17.3 28.1 Abdominal Pain 2.5 1.9 2.6 1.3 Diarrhea 3.8 7.8 3.2 9.2 Dyspepsia 4.5 4.5 4.5 6.5 Flatulence 4.5 3.2 3.2 3.9 Nausea 3.2 3.9 3.8 7.2 Body as a Whole Accident Household 1.9 4.5 3.2 2.6 Edema* 2.5 1.9 4.5 3.3 Fall 0.6 2.6 0 1.3 Influenza-Like Symptoms 5.1 4.5 5.8 2.6 Central and Peripheral Nervous System Dizziness 3.2 2.6 3.8 2 Headache 10.2 7.8 8.3 5.9 Respiratory Pharyngitis 1.3 0.6 3.2 1.3 Upper Respiratory Tract Infection 1.9 3.2 1.9 3.3 Skin Rash† 2.5 2.6 0.6 2 Table 1b Adverse Events (%) Occurring in ≥ 2% of Meloxicam Patients in Two 12-Week Rheumatoid Arthritis Placebo-Controlled Trials - * MedDRA preferred term: nausea, abdominal pain NOS, influenza like illness, headaches NOS and rash NOS
- † MedDRA high level term (preferred terms): dyspeptic signs and symptoms (dyspepsia, dyspepsia aggravated, eructation, gastrointestinal irritation), upper respiratory tract infections-pathogen unspecified (laryngitis NOS, pharyngitis NOS, sinusitis NOS), joint related signs and symptoms (arthralgia, arthralgia aggravated, joint crepitation, joint effusion, joint swelling)
Placebo Meloxicam
7.5 mg dailyMeloxicam
15 mg
dailyNo. of Patients 469 481 477 Gastrointestinal Disorders 14.1 18.9 16.8 Abdominal Pain NOS* 0.6 2.9 2.3 Dyspeptic Signs and Symptoms† 3.8 5.8 4 Nausea* 2.6 3.3 3.8 General Disorders and Administration Site Conditions Influenza-Like Symptoms* 2.1 2.9 2.3 Infection and Infestations Upper Respiratory Tract Infections-
Pathogen Class Unspecified†4.1 7 6.5 Musculoskeletal and Connective Tissue Disorders Joint Related Signs and Symptoms † 1.9 1.5 2.3 Nervous System Disorders Headaches NOS* 6.4 6.4 5.5 Skin and Subcutaneous Tissue Disorders Rash NOS* 1.7 1 2.1 The adverse events that occurred with meloxicam in ≥ 2% of patients treated short-term (4 to 6 weeks) and long-term (6 months) in active-controlled osteoarthritis trials are presented in Table 2.
Table 2 Adverse Events (%) Occurring in ≥ 2% of Meloxicam Patients in 4 to 6 Weeks and 6 Month Active-Controlled Osteoarthritis Trials - * WHO preferred terms edema, edema dependent, edema peripheral and edema legs combined
- † WHO preferred terms rash, rash erythematous and rash maculo-papular combined
4 to 6 Weeks Controlled Trials 6 Month Controlled Trials Meloxicam
7.5 mg dailyMeloxicam
15 mg dailyMeloxicam
7.5 mg dailyMeloxicam
15 mg dailyNo. of Patients 8,955 256 169 306 Gastrointestinal 11.8 18 26.6 24.2 Abdominal Pain 2.7 2.3 4.7 2.9 Constipation 0.8 1.2 1.8 2.6 Diarrhea 1.9 2.7 5.9 2.6 Dyspepsia 3.8 7.4 8.9 9.5 Flatulence 0.5 0.4 3 2.6 Nausea 2.4 4.7 4.7 7.2 Vomiting 0.6 0.8 1.8 2.6 Body as a Whole Accident Household 0 0 0.6 2.9 Edema* 0.6 2 2.4 1.6 Pain 0.9 2 3.6 5.2 Central and Peripheral Nervous System Dizziness 1.1 1.6 2.4 2.6 Headache 2.4 2.7 3.6 2.6 Hematologic Anemia 0.1 0 4.1 2.9 Musculoskeletal Arthralgia 0.5 0 5.3 1.3 Back Pain 0.5 0.4 3 0.7 Psychiatric Insomnia 0.4 0 3.6 1.6 Respiratory Coughing 0.2 0.8 2.4 1 Upper Respiratory 0.2 0 8.3 7.5 Tract Infection Skin Pruritus 0.4 1.2 2.4 0 Rash† 0.3 1.2 3 1.3 Urinary Micturition
Frequency0.1 0.4 2.4 1.3 Urinary Tract
Infection0.3 0.4 4.7 6.9 Higher doses of meloxicam (22.5 mg and greater) have been associated with an increased risk of serious GI events; therefore, the daily dose of meloxicam should not exceed 15 mg.
The following is a list of adverse drug reactions occurring in < 2% of patients receiving meloxicam in clinical trials involving approximately 16,200 patients.
Body as a Whole allergic reaction, face edema, fatigue, fever, hot flushes, malaise, syncope, weight decrease, weight increase Cardiovascular angina pectoris, cardiac failure, hypertension, hypotension, myocardial infarction, vasculitis Central and Peripheral Nervous System convulsions, paresthesia, tremor, vertigo Gastrointestinal colitis, dry mouth, duodenal ulcer, eructation, esophagitis, gastric ulcer, gastritis, gastroesophageal reflux, gastrointestinal hemorrhage, hematemesis, hemorrhagic duodenal ulcer, hemorrhagic gastric ulcer, intestinal perforation, melena, pancreatitis, perforated duodenal ulcer, perforated gastric ulcer, stomatitis ulcerative Heart Rate and Rhythm arrhythmia, palpitation, tachycardia Hematologic leukopenia, purpura, thrombocytopenia Liver and Biliary System ALT increased, AST increased, bilirubinemia, GGT increased, hepatitis Metabolic and Nutritional dehydration Psychiatric abnormal dreaming, anxiety, appetite increased, confusion, depression, nervousness, somnolence Respiratory asthma, bronchospasm, dyspnea Skin and Appendages alopecia, angioedema, bullous eruption, photosensitivity reaction, pruritus, sweating increased, urticaria Special Senses abnormal vision, conjunctivitis, taste perversion, tinnitus Urinary System albuminuria, BUN increased, creatinine increased, hematuria, renal failure 6.2 Post-Marketing Experience
The following adverse reactions have been identified during post approval use of meloxicam. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions about whether to include an adverse event from spontaneous reports in labeling are typically based on one or more of the following factors: (1) seriousness of the event, (2) number of reports or (3) strength of causal relationship to the drug. Adverse reactions reported in worldwide post-marketing experience or the literature include: acute urinary retention; agranulocytosis; alterations in mood (such as mood elevation); anaphylactoid reactions including shock; erythema multiforme; exfoliative dermatitis; interstitial nephritis; jaundice; liver failure; Stevens-Johnson Syndrome and toxic epidermal necrolysis.
-
7 DRUG INTERACTIONS
See also Clinical Pharmacology (12.3).
7.1 ACE-Inhibitors
NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking meloxicam concomitantly with ACE-inhibitors.
7.2 Aspirin
When meloxicam is administered with aspirin (1000 mg 3 times daily) to healthy volunteers, an increase in the AUC (10%) and Cmax (24%) of meloxicam was noted. The clinical significance of this interaction is not known; however, as with other NSAIDs concomitant administration of meloxicam and aspirin is not generally recommended because of the potential for increased adverse effects.
Concomitant administration of low-dose aspirin with meloxicam may result in an increased rate of GI ulceration or other complications, compared to use of meloxicam alone. Meloxicam is not a substitute for aspirin for cardiovascular prophylaxis.
7.3 Diuretics
Clinical studies, as well as post-marketing observations, have shown that NSAIDs can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single- and multiple-dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam. Nevertheless, during concomitant therapy with meloxicam, patients should be observed closely for signs of renal failure [see Warnings and Precautions (5.6)], as well as to ensure diuretic efficacy.
7.4 Lithium
In a study conducted in healthy subjects, mean pre-dose lithium concentration and AUC were increased by 21% in subjects receiving lithium doses ranging from 804 mg to 1072 mg twice daily with meloxicam 15 mg every day as compared to subjects receiving lithium alone. These effects have been attributed to inhibition of renal prostaglandin synthesis by meloxicam. Closely monitor patients on lithium treatment for signs of lithium toxicity when meloxicam is introduced, adjusted or withdrawn.
7.5 Methotrexate
NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. Therefore, NSAIDs may reduce the elimination of methotrexate, thereby enhancing the toxicity of methotrexate. Use caution when meloxicam is administered concomitantly with methotrexate [see Clinical Pharmacology (12.3)].
7.6 Cyclosporine
Meloxicam, like other NSAIDs, may affect renal prostaglandins, thereby altering the renal toxicity of certain drugs. Therefore, concomitant therapy with meloxicam may increase cyclosporine's nephrotoxicity. Use caution when meloxicam is administered concomitantly with cyclosporine.
7.7 Warfarin
The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
Monitor anticoagulant activity, particularly in the first few days after initiating or changing meloxicam therapy in patients receiving warfarin or similar agents, since these patients are at an increased risk of bleeding than with the use of either drug alone. Use caution when administering meloxicam with warfarin since patients on warfarin may experience changes in INR and an increased risk of bleeding complications when a new medication is introduced [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C; Category D Starting 30 Weeks Gestation
There are no adequate and well controlled studies in pregnant women. Meloxicam crosses the placental barrier. Prior to 30 weeks gestation, use meloxicam during pregnancy only if the potential benefit justifies the potential risk to the fetus. Starting at 30 weeks gestation, avoid meloxicam and other NSAIDs, in pregnant women as premature closure of the ductus arteriosus in the fetus may occur. If this drug is used during this time period in pregnancy, inform the patient of the potential hazard to a fetus [see Warnings and Precautions (5.9) and Patient Counseling Information (17.8)].
Teratogenic Effects
Meloxicam was not teratogenic when administered to pregnant rats during fetal organogenesis at oral doses up to 4 mg/kg/day (2.6-fold greater than the maximum recommended human daily dose [MRHD] based on body surface area [BSA] comparison). Administration of meloxicam to pregnant rabbits throughout embryogenesis produced an increased incidence of septal defects of the heart at an oral dose of 60 mg/kg/day. The no effect level was 20 mg/kg/day (26-fold greater than the MRHD based on BSA conversion).
8.2 Labor and Delivery
The effects of meloxicam on labor and delivery of pregnant women are unknown. Oral administration of meloxicam to pregnant rats during late gestation through lactation increased the incidence of dystocia, delayed parturition and decreased offspring survival at meloxicam doses of 0.125 mg/kg/day or greater (at least 12.5 times lower than the maximum recommended human daily dose based on body surface area comparison).
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk; however, meloxicam was excreted in the milk of lactating rats at concentrations higher than those in plasma. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from meloxicam, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Use of this drug for a pediatric indication is protected by marketing exclusivity.
8.5 Geriatric Use
As with any NSAID, caution should be exercised in treating the elderly (65 years and older).
Of the total number of subjects in clinical studies, 5,157 were age 65 and over (4,044 in OA studies and 1,113 in RA studies). No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
No dose adjustment is necessary in patients with mild to moderate hepatic impairment. Patients with severe hepatic impairment have not been adequately studied. Since meloxicam is significantly metabolized in the liver; the use of meloxicam in these patients should be done with caution [see Warnings and Precautions (5.3) and Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dose adjustment is necessary in patients with mild to moderate renal impairment. Patients with severe renal impairment have not been studied. The use of meloxicam in subjects with severe renal impairment is not recommended. Following a single-dose of meloxicam, the free Cmax plasma concentrations were higher in patients with renal failure on chronic hemodialysis (1% free fraction) in comparison to healthy volunteers (0.3% free fraction). Therefore, it is recommended that meloxicam dosage in this population not exceed 7.5 mg per day. Hemodialysis did not lower the total drug concentration in plasma; therefore, additional doses are not necessary after hemodialysis. Meloxicam is not dialyzable [see Dosage and Administration (2.1), Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
8.8 Females of Reproductive Potential
Data from several small studies in humans and from studies in animals indicate that NSAIDs, including meloxicam, may be associated with a reversible delay in ovulation. Therefore, in women who have difficulties conceiving, or who are undergoing investigation of infertility, use of meloxicam is not recommended.
-
10 OVERDOSAGE
There is limited experience with meloxicam overdose. Four cases have taken 6 to 11 times the highest recommended dose; all recovered. Cholestyramine is known to accelerate the clearance of meloxicam.
Symptoms following acute NSAID overdose include lethargy, drowsiness, nausea, vomiting and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Severe poisoning may result in hypertension, acute renal failure, hepatic dysfunction, respiratory depression, coma, convulsions, cardiovascular collapse and cardiac arrest. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Patients should be managed with symptomatic and supportive care following an NSAID overdose. Administration of activated charcoal is recommended for patients who present 1 to 2 hours after overdose. For substantial overdose or severely symptomatic patients, activated charcoal may be administered repeatedly. Accelerated removal of meloxicam by 4 g oral doses of cholestyramine given 3 times a day was demonstrated in a clinical trial. Administration of cholestyramine may be useful following an overdose. Forced diuresis, alkalinization of urine, hemodialysis or hemoperfusion may not be useful due to high protein binding.
For additional information about overdose treatment, call a poison control center (1-800-222-1222).
-
11 DESCRIPTION
Meloxicam, an oxicam derivative, is a member of the enolic acid group of non-steroidal anti-inflammatory drugs (NSAIDs). Each meloxicam tablet contains 7.5 mg or 15 mg meloxicam for oral administration. Meloxicam is chemically designated as 4-hydroxy-2-methyl-N-(5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide-1,1-dioxide. The molecular weight is 351.4. Its molecular formula is C14H13N3O4S2 and it has the following structural formula:
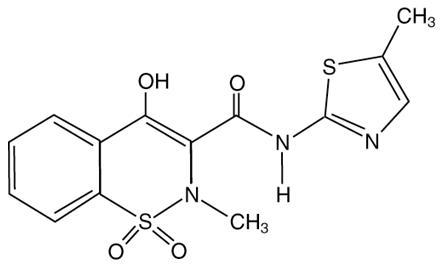
Meloxicam, USP is a pale yellow powder. Practically insoluble in water, slightly soluble in acetone, soluble in dimethylformamide, very slightly soluble in ethanol and in methanol. Meloxicam has an apparent partition coefficient (log P)app = 0.1 in n-octanol/buffer pH 7.4. Meloxicam has pKa values of 1.1 and 4.2.
Meloxicam is available as a tablet for oral administration containing 7.5 mg or 15 mg meloxicam.
The inactive ingredients in meloxicam tablets, USP include colloidal silicon dioxide, crospovidone, lactose monohydrate, magnesium stearate, microcrystalline cellulose and sodium lauryl sulfate.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of meloxicam, like that of other NSAIDs, may be related to prostaglandin synthetase (cyclo-oxygenase) inhibition which is involved in the initial steps of the arachidonic acid cascade, resulting in the reduced formation of prostaglandins, thromboxanes and prostacylin. It is not completely understood how reduced synthesis of these compounds results in therapeutic efficacy.
12.3 Pharmacokinetics
Absorption
The absolute bioavailability of meloxicam capsules was 89% following a single oral dose of 30 mg compared with 30 mg IV bolus injection. Following single intravenous doses, dose proportional pharmacokinetics were shown in the range of 5 mg to 60 mg. After multiple oral doses the pharmacokinetics of meloxicam capsules were dose proportional over the range of 7.5 mg to 15 mg. Mean Cmax was achieved within 4 to 5 hours after a 7.5 mg meloxicam tablet was taken under fasted conditions, indicating a prolonged drug absorption. With multiple dosing, steady-state concentrations were reached by Day 5. A second meloxicam concentration peak occurs around 12 to 14 hours post-dose suggesting biliary recycling.
Table 3 Single-Dose and Steady-State Pharmacokinetic Parameters for Oral 7.5 mg and 15 mg Meloxicam (Mean and % CV)* - * The parameter values in the table are from various studies
- † Not under high fat conditions
- ‡ Meloxicam tablets
- § Vz/f =Dose/(AUCKel)
Steady-State Single-Dose Pharmacokinetic
Parameters
(% CV)Healthy
Male Adults
(Fed)†Elderly
Males
(Fed)†Elderly
Females
(Fed)†Renal
Failure
(Fasted)Hepatic
Insufficiency
(Fasted)7.5 mg‡
tablets15 mg capsules 15 mg capsules 15 mg capsules 15 mg capsules N 18 5 8 12 12 Cmax [mcg/mL] 1.05 (20) 2.3 (59) 3.2 (24) 0.59 (36) 0.84 (29) tmax [h] 4.9 (8) 5 (12) 6 (27) 4 (65) 10 (87) t1/2 [h] 20.1 (29) 21 (34) 24 (34) 18 (46) 16 (29) CL/f [mL/min] 8.8 (29) 9.9 (76) 5.1 (22) 19 (43) 11 (44) Vz/f§ [L] 14.7 (32) 15 (42) 10 (30) 26 (44) 14 (29) Food and Antacid Effects
Administration of meloxicam capsules following a high fat breakfast (75 g of fat) resulted in mean peak drug levels (i.e., Cmax) being increased by approximately 22% while the extent of absorption (AUC) was unchanged. The time to maximum concentration (Tmax) was achieved between 5 and 6 hours. In comparison, neither the AUC nor the Cmax values for meloxicam suspension were affected following a similar high fat meal, while mean Tmax values were increased to approximately 7 hours. No pharmacokinetic interaction was detected with concomitant administration of antacids. Based on these results, meloxicam can be administered without regard to timing of meals or concomitant administration of antacids.
Distribution
The mean volume of distribution (Vss) of meloxicam is approximately 10 L. Meloxicam is ~ 99.4% bound to human plasma proteins (primarily albumin) within the therapeutic dose range. The fraction of protein binding is independent of drug concentration, over the clinically relevant concentration range, but decreases to ~ 99% in patients with renal disease. Meloxicam penetration into human red blood cells, after oral dosing, is less than 10%. Following a radiolabeled dose, over 90% of the radioactivity detected in the plasma was present as unchanged meloxicam.
Meloxicam concentrations in synovial fluid, after a single oral dose, range from 40% to 50% of those in plasma. The free fraction in synovial fluid is 2.5 times higher than in plasma, due to the lower albumin content in synovial fluid as compared to plasma. The significance of this penetration is unknown.
Metabolism
Meloxicam is extensively metabolized in the liver. Meloxicam metabolites include 5'-carboxy meloxicam (60% of dose), from P-450 mediated metabolism formed by oxidation of an intermediate metabolite 5'-hydroxymethyl meloxicam which is also excreted to a lesser extent (9% of dose). In vitro studies indicate that CYP2C9 (cytochrome P450 metabolizing enzyme) plays an important role in this metabolic pathway with a minor contribution of the CYP3A4 isozyme. Patients’ peroxidase activity is probably responsible for the other two metabolites which account for 16% and 4% of the administered dose, respectively. All the four metabolites are not known to have any in vivo pharmacological activity.
Excretion
Meloxicam excretion is predominantly in the form of metabolites, and occurs to equal extents in the urine and feces. Only traces of the unchanged parent compound are excreted in the urine (0.2%) and feces (1.6%). The extent of the urinary excretion was confirmed for unlabeled multiple 7.5 mg doses: 0.5%, 6%, and 13% of the dose were found in urine in the form of meloxicam, and the 5'-hydroxymethyl and 5'-carboxy metabolites, respectively. There is significant biliary and/or enteral secretion of the drug. This was demonstrated when oral administration of cholestyramine following a single IV dose of meloxicam decreased the AUC of meloxicam by 50%.
The mean elimination half-life (t1/2) ranges from 15 hours to 20 hours. The elimination half-life is constant across dose levels indicating linear metabolism within the therapeutic dose range. Plasma clearance ranges from 7 to 9 mL/min.
Special Populations
Geriatric
Elderly males (≥ 65 years of age) exhibited meloxicam plasma concentrations and steady-state pharmacokinetics similar to young males. Elderly females (≥ 65 years of age) had a 47% higher AUCss and 32% higher Cmax,ss as compared to younger females (≤ 55 years of age) after body weight normalization. Despite the increased total concentrations in the elderly females, the adverse event profile was comparable for both elderly patient populations. A smaller free fraction was found in elderly female patients in comparison to elderly male patients.
Gender
Young females exhibited slightly lower plasma concentrations relative to young males. After single doses of 7.5 mg meloxicam, the mean elimination half-life was 19.5 hours for the female group as compared to 23.4 hours for the male group. At steady-state, the data were similar (17.9 hours vs. 21.4 hours). This pharmacokinetic difference due to gender is likely to be of little clinical importance. There was linearity of pharmacokinetics and no appreciable difference in the Cmax or Tmax across genders.
Hepatic Impairment
Following a single 15 mg dose of meloxicam there was no marked difference in plasma concentrations in patients with mild (Child-Pugh Class I) or moderate (Child-Pugh Class II) hepatic impairment compared to healthy volunteers. Protein binding of meloxicam was not affected by hepatic impairment. No dosage adjustment is necessary in patients with mild to moderate hepatic impairment. Patients with severe hepatic impairment (Child-Pugh Class III) have not been adequately studied [see Warnings and Precautions (5.3) and Use in Specific Populations (8.6)].
Renal Impairment
Meloxicam pharmacokinetics have been investigated in subjects with mild and moderate renal impairment. Total drug plasma concentrations of meloxicam decreased and total clearance of meloxicam increased with the degree of renal impairment while free AUC values were similar in all groups. The higher meloxicam clearance in subjects with renal impairment may be due to increased fraction of unbound meloxicam which is available for hepatic metabolism and subsequent excretion. No dosage adjustment is necessary in patients with mild to moderate renal impairment. Patients with severe renal impairment have not been adequately studied. The use of meloxicam in subjects with severe renal impairment is not recommended [see Warnings and Precautions (5.6) and Use in Specific Populations (8.7)].
Hemodialysis
Following a single-dose of meloxicam, the free Cmax plasma concentrations were higher in patients with renal failure on chronic hemodialysis (1% free fraction) in comparison to healthy volunteers (0.3% free fraction). Hemodialysis did not lower the total drug concentration in plasma; therefore, additional doses are not necessary after hemodialysis. Meloxicam is not dialyzable [see Dosage and Administration (2.1), Warnings and Precautions (5.6) and Use in Specific Populations (8.7)].
Drug Interactions
Aspirin
When meloxicam is administered with aspirin (1000 mg 3 times daily) to healthy volunteers, it tended to increase the AUC (10%) and Cmax (24%) of meloxicam. The clinical significance of this interaction is not known [see Drug Interactions (7.2)].
Cholestyramine
Pretreatment for 4 days with cholestyramine significantly increased the clearance of meloxicam by 50%. This resulted in a decrease in t1/2, from 19.2 hours to 12.5 hours, and a 35% reduction in AUC. This suggests the existence of a recirculation pathway for meloxicam in the gastrointestinal tract. The clinical relevance of this interaction has not been established.
Cimetidine
Concomitant administration of 200 mg cimetidine 4 times daily did not alter the single-dose pharmacokinetics of 30 mg meloxicam.
Digoxin
Meloxicam 15 mg once daily for 7 days did not alter the plasma concentration profile of digoxin after ß-acetyldigoxin administration for 7 days at clinical doses. In vitro testing found no protein binding drug interaction between digoxin and meloxicam.
Lithium
In a study conducted in healthy subjects, mean pre-dose lithium concentration and AUC were increased by 21% in subjects receiving lithium doses ranging from 804 mg to 1072 mg twice daily with meloxicam 15 mg QD every day as compared to subjects receiving lithium alone [see Drug Interactions (7.4)].
Methotrexate
A study in 13 rheumatoid arthritis (RA) patients evaluated the effects of multiple doses of meloxicam on the pharmacokinetics of methotrexate taken once weekly. Meloxicam did not have a significant effect on the pharmacokinetics of single doses of methotrexate. In vitro, methotrexate did not displace meloxicam from its human serum binding sites [see Drug Interactions (7.5)].
Warfarin
The effect of meloxicam on the anticoagulant effect of warfarin was studied in a group of healthy subjects receiving daily doses of warfarin that produced an INR (International Normalized Ratio) between 1.2 and 1.8. In these subjects, meloxicam did not alter warfarin pharmacokinetics and the average anticoagulant effect of warfarin as determined by prothrombin time. However, one subject showed an increase in INR from 1.5 to 2.1. Caution should be used when administering meloxicam with warfarin since patients on warfarin may experience changes in INR and an increased risk of bleeding complications when a new medication is introduced [see Drug Interactions (7.7)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
There was no increase in tumor incidence in long-term carcinogenicity studies in rats (104 weeks) and mice (99 weeks) administered meloxicam at oral doses up to 0.8 mg/kg/day in rats and up to 8 mg/kg/day in mice (up to 0.5- and 2.6-fold, respectively, the maximum recommended human daily dose based on body surface area comparison).
-
14 CLINICAL STUDIES
14.1 Osteoarthritis and Rheumatoid Arthritis
The use of meloxicam for the treatment of the signs and symptoms of osteoarthritis of the knee and hip was evaluated in a 12-week, double-blind, controlled trial. Meloxicam (3.75 mg, 7.5 mg, and 15 mg daily) was compared to placebo. The four primary endpoints were investigator’s global assessment, patient global assessment, patient pain assessment and total WOMAC score (a self-administered questionnaire addressing pain, function and stiffness). Patients on meloxicam 7.5 mg daily and meloxicam 15 mg daily showed significant improvement in each of these endpoints compared with placebo.
The use of meloxicam for the management of signs and symptoms of osteoarthritis was evaluated in six double-blind, active-controlled trials outside the U.S. ranging from 4 weeks’ to 6 months’ duration. In these trials, the efficacy of meloxicam, in doses of 7.5 mg/day and 15 mg/day, was comparable to piroxicam 20 mg/day and diclofenac SR 100 mg/day and consistent with the efficacy seen in the U.S. trial.
The use of meloxicam for the treatment of the signs and symptoms of rheumatoid arthritis was evaluated in a 12-week, double-blind, controlled multinational trial. Meloxicam (7.5 mg, 15 mg, and 22.5 mg daily) was compared to placebo. The primary endpoint in this study was the ACR20 response rate, a composite measure of clinical, laboratory and functional measures of RA response. Patients receiving meloxicam 7.5 mg and 15 mg daily showed significant improvement in the primary endpoint compared with placebo. No incremental benefit was observed with the 22.5 mg dose compared to the 15 mg dose.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Meloxicam Tablets, USP are available containing 7.5 mg or 15 mg of meloxicam, USP.
The 7.5 mg tablet is a yellow round, unscored tablet debossed with M over 66 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 0378-1066-01
bottles of 100 tabletsNDC: 0378-1066-05
bottles of 500 tabletsThe 15 mg tablet is a yellow round, unscored tablet debossed with M over 89 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 0378-1089-01
bottles of 100 tabletsNDC: 0378-1089-05
bottles of 500 tabletsStore at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from moisture.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved Medication Guide
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy.
17.1 Medication Guide
Inform patients of the availability of a Medication Guide for NSAIDs that accompanies each prescription dispensed, and instruct them to read the Medication Guide prior to using meloxicam tablets.
17.2 Cardiovascular Effects
NSAIDs including meloxicam tablets may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up [see Warnings and Precautions (5.1)].
17.3 Gastrointestinal Effects
NSAIDs including meloxicam tablets, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena and hematemesis. Patients should be apprised of the importance of this follow-up [see Warnings and Precautions (5.2)].
17.4 Hepatotoxicity
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness and "flu-like" symptoms). If these occur, instruct patients to stop therapy and seek immediate medical therapy [see Warnings and Precautions (5.3)].
17.5 Adverse Skin Reactions
NSAIDs including meloxicam tablets, can cause serious skin side effects such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which may result in hospitalization and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Advise patients to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible [see Warnings and Precautions (5.8)].
17.6 Weight Gain and Edema
Advise patients to promptly report signs or symptoms of unexplained weight gain or edema to their physicians [see Warnings and Precautions (5.5)].
17.7 Anaphylactoid Reactions
Inform patients of the signs of an anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). Instruct patients seek immediate emergency help [see Warnings and Precautions (5.7)].
17.8 Effects During Pregnancy
Starting at 30 weeks gestation, meloxicam tablets should be avoided as premature closure of the ductus arteriosus in the fetus may occur [see Warnings and Precautions (5.9) and Use in Specific Populations (8.1)].
17.9 Effects On Female Fertility
Advise females of reproductive potential who desire pregnancy that NSAIDs, including meloxicam tablets, may be associated with a reversible delay in ovulation For women who have difficulties conceiving, or who are undergoing investigation of infertility, use of meloxicam is not recommended [see Use in Specific Populations 8.8)].
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Repackaged By:
ReadyMeds
Dothan, AL 36301
REVISED JUNE 2012
MLXC:R4mc -
SPL UNCLASSIFIED SECTION
Medication Guide
for
Non-Steroidal Anti-Inflammatory Drugs
(NSAIDs)(See the end of this Medication Guide for a list of prescription NSAID medicines.)
What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines may increase the chance of a heart attack or stroke that can lead to death. This chance increases:
- with longer use of NSAID medicines
- in people who have heart disease
NSAID medicines should never be used right before or after a heart surgery called a “coronary artery bypass graft (CABG).”
NSAID medicines can cause ulcers and bleeding in the stomach and intestines at any time during treatment. Ulcers and bleeding:
- can happen without warning symptoms
- may cause death
The chance of a person getting an ulcer or bleeding increases with:
- taking medicines called “corticosteroids” and “anticoagulants”
- longer use
- smoking
- drinking alcohol
- older age
- having poor health
NSAID medicines should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
NSAID medicines are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as:
- different types of arthritis
- menstrual cramps and other types of short-term pain
Who should not take a Non-Steroidal Anti-Inflammatory Drug (NSAID)?
Do not take an NSAID medicine:
- if you had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAID medicine
- for pain right before or after heart bypass surgery
Tell your healthcare provider:
- about all of your medical conditions.
- about all of the medicines you take. NSAIDs and some other medicines can interact with each other and cause serious side effects. Keep a list of your medicines to show to your healthcare provider and pharmacist.
- if you are pregnant. NSAID medicines should not be used by pregnant women late in their pregnancy.
- if you are breastfeeding. Talk to your doctor.
What are the possible side effects of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
Serious side effects include:
- heart attack
- stroke
- high blood pressure
- heart failure from body swelling (fluid retention)
- kidney problems including kidney failure
- bleeding and ulcers in the stomach and intestine
- low red blood cells (anemia)
- life-threatening skin reactions
- life-threatening allergic reactions
- liver problems including liver failure
- asthma attacks in people who have asthma
Other side effects include:
- stomach pain
- constipation
- diarrhea
- gas
- heartburn
- nausea
- vomiting
- dizziness
Get emergency help right away if you have any of the following symptoms:
- shortness of breath or trouble breathing
- chest pain
- weakness in one part or side of your body
- slurred speech
- swelling of the face or throat
Stop your NSAID medicine and call your healthcare provider right away if you have any of the following symptoms:
- nausea
- more tired or weaker than usual
- itching
- your skin or eyes look yellow
- stomach pain
- flu-like symptoms
- vomit blood
- there is blood in your bowel movement or it is black and sticky like tar
- unusual weight gain
- skin rash or blisters with fever
- swelling of the arms and legs, hands and feet
These are not all the side effects with NSAID medicines. Talk to your healthcare provider or pharmacist for more information about NSAID medicines.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
- Aspirin is an NSAID medicine but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some of these NSAID medicines are sold in lower doses without a prescription (over-the-counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.
NSAID medicines that need a prescription - * Vicoprofen contains the same dose of ibuprofen as over-the-counter (OTC) NSAIDs, and is usually used for less than 10 days to treat pain. The OTC NSAID label warns that long term continuous use may increase the risk of heart attack or stroke.
Generic Name Tradename Celecoxib Celebrex Diclofenac Cataflam, Voltaren, Arthrotec (combined with misoprostol) Diflunisal Dolobid Etodolac Lodine, Lodine XL Fenoprofen Nalfon, Nalfon 200 Flurbiprofen Ansaid Ibuprofen Motrin, Tab-Profen, Vicoprofen* (combined with hydrocodone), Combunox (combined with oxycodone) Indomethacin Indocin, Indocin SR, Indo-Lemmon, Indomethagan Ketoprofen Oruvail Ketorolac Toradol Mefenamic Acid Ponstel Meloxicam Mobic Nabumetone Relafen Naproxen Naprosyn, Anaprox, Anaprox DS, EC-Naprosyn, Naprelan, Naprapac (copackaged with lansoprazole) Oxaprozin Daypro Piroxicam Feldene Sulindac Clinoril Tolmetin Tolectin, Tolectin DS, Tolectin 600 This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised 06/2009
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 7.5 mg
NDC: 64205-106-30
Meloxicam
Tablets, USP
7.5 mgPHARMACIST: Dispense the Medication
Guide provided separately to each patient.Rx only 30 Tablets
Each tablet contains:
Meloxicam, USP 7.5 mgDispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Protect from moisture.
Usual Dosage: See accompanying
prescribing information.Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Mylan.com
Repackaged By:
ReadyMeds
Dothan, AL 36301
RM1066A4

-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 15 mg
NDC: 64205-108-30
Meloxicam
Tablets, USP
15 mgPHARMACIST: Dispense the Medication
Guide provided separately to each patient.Rx only 30 Tablets
Each tablet contains:
Meloxicam, USP 15 mgDispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.Keep container tightly closed.
Keep this and all medication
out of the reach of children.Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]Protect from moisture.
Usual Dosage: See accompanying
prescribing information.Mylan Pharmaceuticals Inc.Morgantown, WV 26505 U.S.A.
Mylan.com
Repackaged By:
ReadyMeds
Dothan, AL 36301
RM1089A4

-
INGREDIENTS AND APPEARANCE
MELOXICAM
meloxicam tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 64205-106(NDC: 0378-1066) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MELOXICAM (UNII: VG2QF83CGL) (MELOXICAM - UNII:VG2QF83CGL) MELOXICAM 7.5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 68401960MK) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color YELLOW Score no score Shape ROUND Size 6mm Flavor Imprint Code M;66 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 64205-106-30 30 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077923 08/28/2012 MELOXICAM
meloxicam tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 64205-108(NDC: 0378-1089) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MELOXICAM (UNII: VG2QF83CGL) (MELOXICAM - UNII:VG2QF83CGL) MELOXICAM 15 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSPOVIDONE (UNII: 68401960MK) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color YELLOW Score no score Shape ROUND Size 8mm Flavor Imprint Code M;89 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 64205-108-30 30 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077923 08/28/2012 Labeler - ReadyMeds (072115132) Establishment Name Address ID/FEI Business Operations ReadyMeds 072115132 repack(64205-106, 64205-108)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.