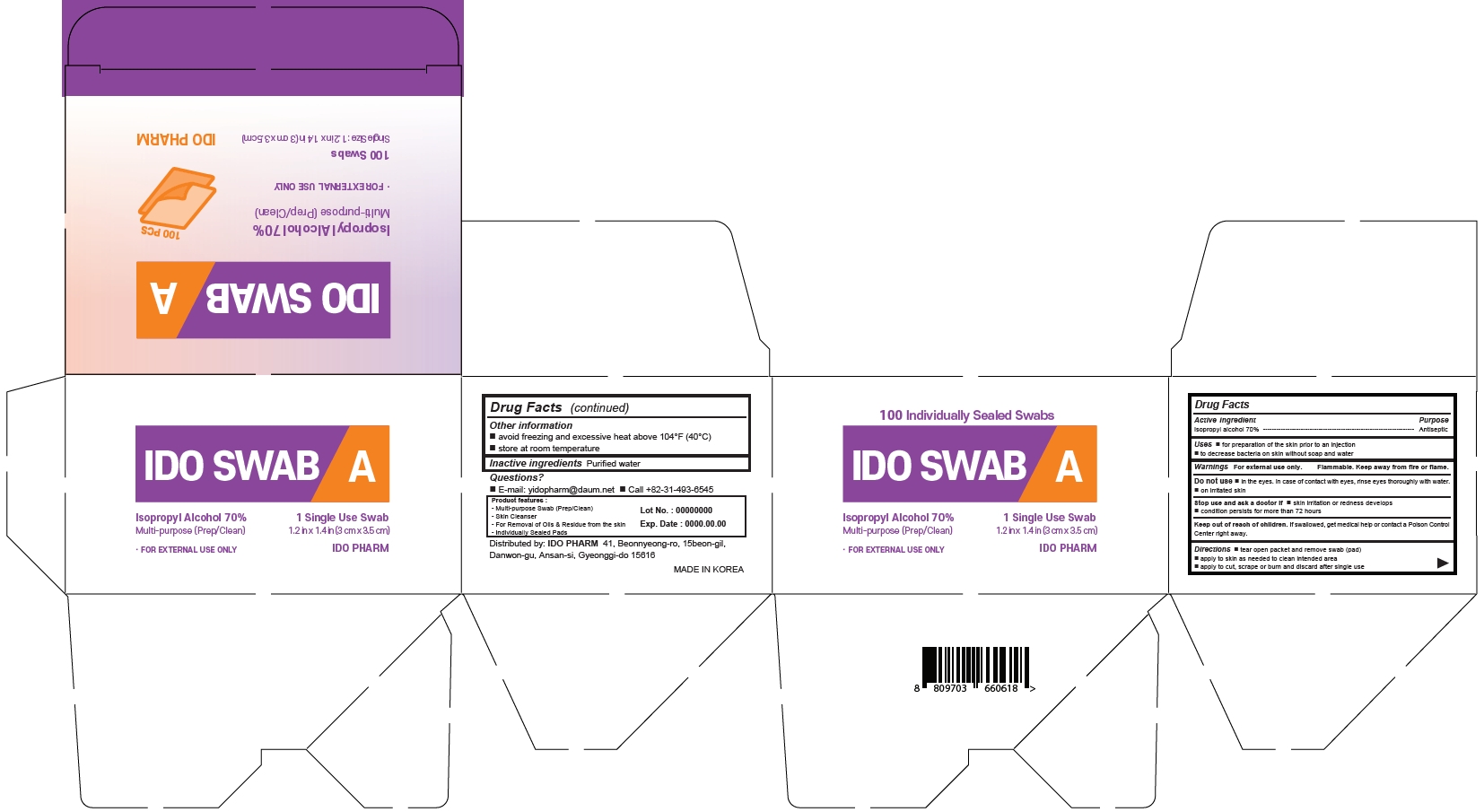
IDO Isopropyl Alcohol Pad by IDO PHARM IDO SWAB A Alcohol Swab
IDO Isopropyl Alcohol Pad by
Drug Labeling and Warnings
IDO Isopropyl Alcohol Pad by is a Otc medication manufactured, distributed, or labeled by IDO PHARM. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
IDO ISOPROPYL ALCOHOL PAD- isopropyl alcohol swab
IDO PHARM
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
IDO SWAB A Alcohol Swab
Uses
- for preparation of the skin prior to an injection
- to decrease bacteria on skin without soap and water
Warnings
For external use only.
Flammable. Keep away from heat or flame.
Do not use
- in eyes. In case of contact with eyes, rinse eyes thoroughly with water
- on irritated skin
| IDO ISOPROPYL ALCOHOL PAD
isopropyl alcohol swab |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - IDO PHARM (694853523) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| IDO PHARM | 694853523 | manufacture(77039-001) | |
Revised: 12/2021
Document Id: d3e5f9ca-1c66-754a-e053-2a95a90a488e
Set id: aa18319f-64eb-6baf-e053-2995a90a0172
Version: 2
Effective Time: 20211224
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.