XYZMUNE- ascorbic acid, cholecalciferol, folic acid, zinc, echinacea, l-lysine hcl, tumeric, ginger capsule
XYZMUNE by
Drug Labeling and Warnings
XYZMUNE by is a Other medication manufactured, distributed, or labeled by Basiem, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HEALTH CLAIM:
XYZMUNE Capsules - Dietary Supplement
Dispensed by Prescription‡
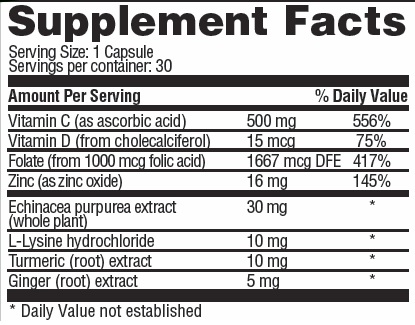
OTHER INGREDIENTS: gelatin, calcium stearate, silica, organic mushroom (Pleurotus eryngii), organic turkey tail (Trametes versicolor), organic cordyceps (Cordyceps militaris), organic reishi (Ganoderna lucidum) organic himematsutake (Agaricus blazei), organic lion's mane (Hericium erincaces), organic antrodia (Taiwanofungus camphoratus (Antrodia camphorate)), organic maitake (Grifola frondosa)
-
DESCRIPTION:
XYZMUNE is an orally administered prescription vitamin formulation for the clinical dietary management of suboptimal nutritional status in patients where advanced supplementation is required and nutritional supplementation in physiologically stressful conditions for maintenance of good immune health is needed.†
XYZMUNE is manufactured in accordance with Current Good Manufacturing Practice for foods, using ingredients that have been approved by the U.S. Food and Drug Administration (FDA) as food additives or are “Generally Recognized as Safe” (GRAS) for their intended use.
‡ XYZMUNE should be administered under the supervision of a licensed medical practitioner.
† These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent disease.
-
WARNING AND PRECAUTIONS
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients. XYZMUNE capsules should only be used under the direction and supervision of a licensed medical practitioner.‡ Use with caution in patients that may have a medical condition, are pregnant, lactating, trying to conceive, under the age of 18, or taking medications.
KEEP OUT OF REACH OF CHILDREN
PRECAUTION
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B is deficient. Folic acid in doses above 1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress. XYZMUNE capsules should only be used under the direction and supervision of a licensed medical practitioner. - ADVERSE REACTIONS
- DOSAGE & ADMINISTRATION
-
HOW SUPPLIED HEALTH CLAIM:
XYZMUNE Dietary Supplement
Capsules are clear with a yellow powder inside
Bottles contain 30 capsules, 69597-355-30*
Dispensed by Prescription‡Manufactured in USA for:
Basiem, LLC
Madisonville, LA 70447
Distributed by: Solubiomix, LLC
Rev. 04/2020
* Basiem does not represent these product codes to be National Drug Codes (NDC). Product codes are formatted according to standard industry practice, to meet the formatting requirement by pedigree reporting and supply-chain control including pharmacies.
‡ The most appropriate way to ensure pedigree reporting consistent with these regulatory guidelines and safety monitoring is to dispense this product by prescription. This is not an Orange Book product. This product may be administered only under a physician’s supervision and all prescriptions using this product shall be pursuant to state statutes as applicable. The ingredients, indication or claims of this product are not to be construed to be drug claims.1. Federal Register Notice of August 2, 1973 (38 FR 20750)
2. Federal Register Notice of October 17, 1980 (45 FR 69043, 69044)
3. Federal Register Notice of March 5, 1996 (61 FR 8760) - STORAGE AND HANDLING:
- PACKAGE LABEL:
-
INGREDIENTS AND APPEARANCE
XYZMUNE
ascorbic acid, cholecalciferol, folic acid, zinc, echinacea, l-lysine hcl, tumeric, ginger capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:69597-355 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 500 mg cholecalciferol (UNII: 1C6V77QF41) (cholecalciferol - UNII:1C6V77QF41) cholecalciferol 0.015 mg ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 16 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg ECHINACEA PURPUREA WHOLE (UNII: QI7G114Y98) (ECHINACEA PURPUREA WHOLE - UNII:QI7G114Y98) ECHINACEA PURPUREA WHOLE 30 mg LYSINE HYDROCHLORIDE (UNII: JNJ23Q2COM) (LYSINE - UNII:K3Z4F929H6) LYSINE 10 mg TURMERIC (UNII: 856YO1Z64F) (TURMERIC - UNII:856YO1Z64F) TURMERIC 10 mg GINGER (UNII: C5529G5JPQ) (GINGER - UNII:C5529G5JPQ) GINGER 5 mg Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) CALCIUM STEARATE (UNII: 776XM7047L) SILICA DIMETHYL SILYLATE (UNII: EU2PSP0G0W) PLEUROTUS ERYNGII WHOLE (UNII: 2WGH02ZJ1G) TRAMETES VERSICOLOR WHOLE (UNII: 2YD82VN5CM) CORDYCEPS MILITARIS WHOLE (UNII: J617U5X7NN) GANODERMA LUCIDUM WHOLE (UNII: J5P04QW0CF) AGARICUS BLAZEI WHOLE (UNII: 11MSQ4JG7G) HERICIUM AMERICANUM WHOLE (UNII: UF625TXW2A) TAIWANOFUNGUS CAMPHORATUS WHOLE (UNII: 944OR6BL20) GRIFOLA FRONDOSA WHOLE (UNII: O238UC53ET) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:69597-355-30 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 04/08/2020 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 size (solid drugs) 23 mm shape Labeler - Basiem, LLC (079686680)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
