Hydrolatum by Denison Pharmaceuticals, LLC Hydrolatum
Hydrolatum by
Drug Labeling and Warnings
Hydrolatum by is a Otc medication manufactured, distributed, or labeled by Denison Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
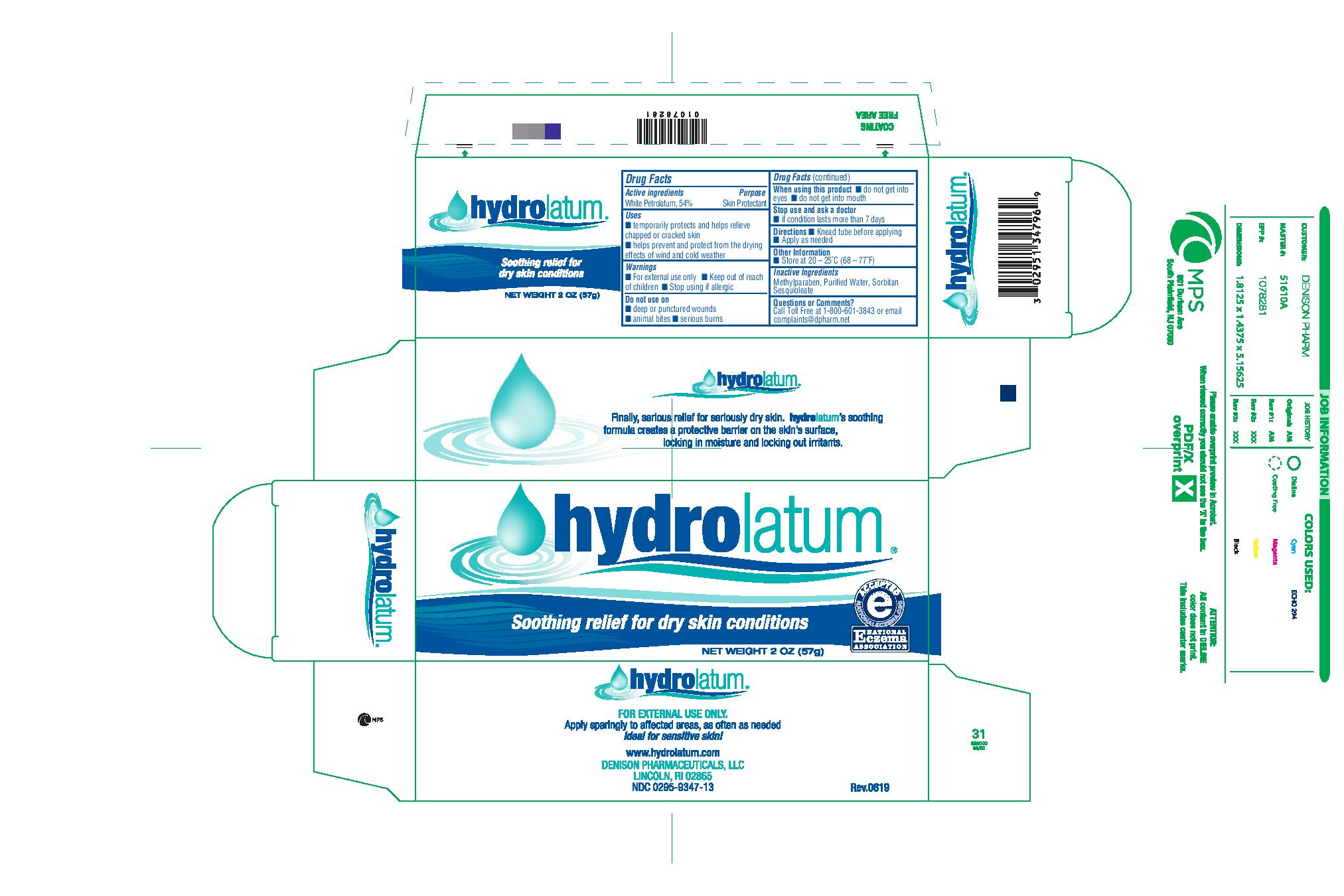
HYDROLATUM- white petrolatum cream
Denison Pharmaceuticals, LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Hydrolatum
| HYDROLATUM
white petrolatum cream |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Denison Pharmaceuticals, LLC (001207208) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Denison Pharmaceuticals, LLC | 001207208 | manufacture(0295-9374) | |
Revised: 8/2020
Document Id: adde34a3-bb15-4925-e053-2995a90a701b
Set id: aae0618d-c778-f0fc-e053-2a95a90a9465
Version: 2
Effective Time: 20200827
Trademark Results [Hydrolatum]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 HYDROLATUM 76691543 3610776 Live/Registered |
DENISON PHARMACEUTICALS, LLC 2008-07-24 |
 HYDROLATUM 74700964 2033025 Dead/Cancelled |
Denison Pharmaceuticals, Inc. 1995-07-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.