51414-911 Antibacterial wet wipes 75% Alcohol
Antibacterial Wet Wipes by
Drug Labeling and Warnings
Antibacterial Wet Wipes by is a Otc medication manufactured, distributed, or labeled by YUYAO JESSIE COMMODITY CO.,LTD. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ANTIBACTERIAL WET WIPES- alcohol cloth
YUYAO JESSIE COMMODITY CO.,LTD
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
51414-911 Antibacterial wet wipes 75% Alcohol
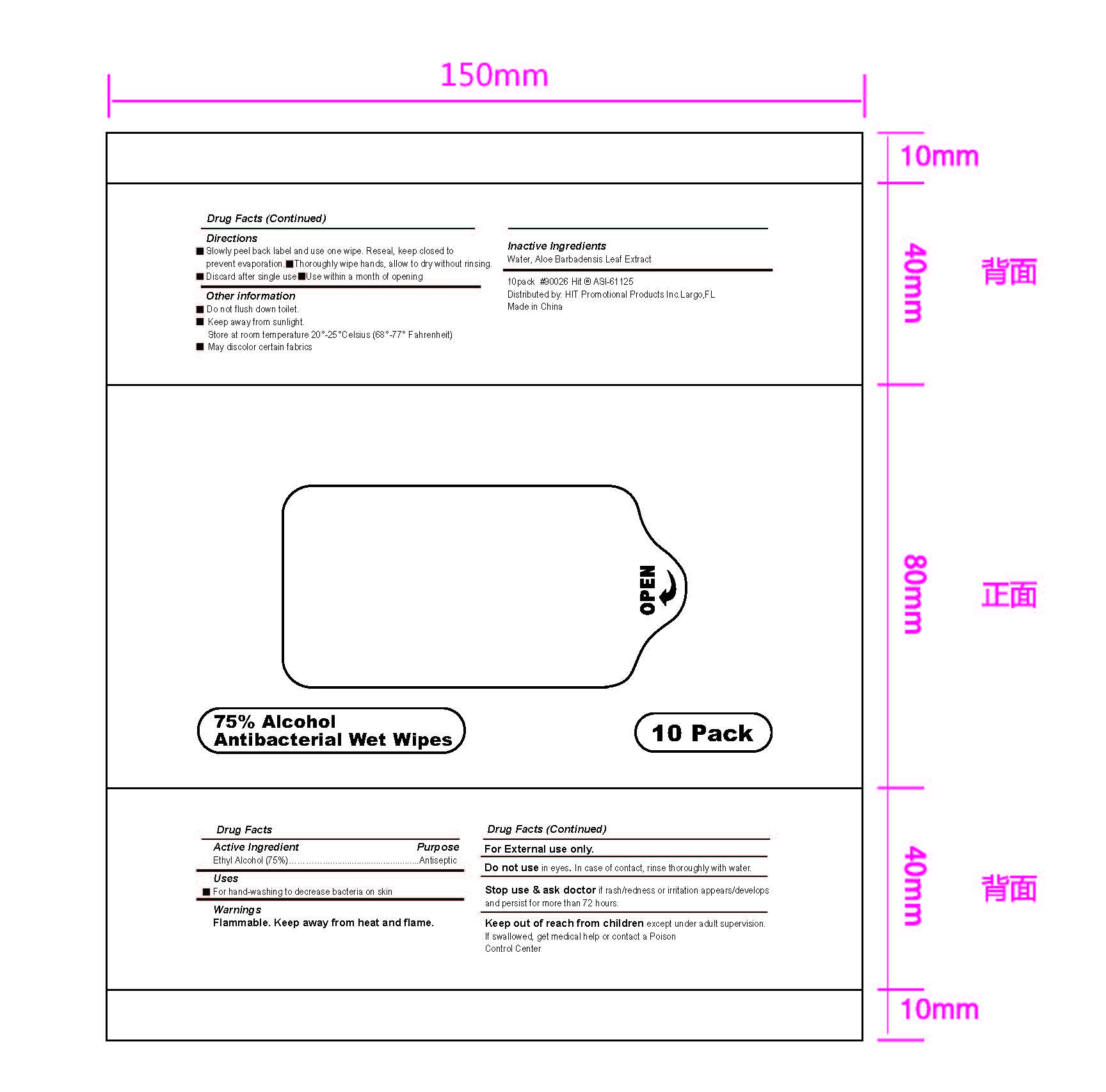
Warnings
For External use only.
Do not use in eyes. In case of contact, rinse thoroughly with water.
Stop use & ask doctor if rash/redness or irritation appears/develops
and persist for more than 72 hours.
Flammable. Keep away from heat and flame.
Keep out of reach from children except under adult supervision.
If swallowed, get medical help or contact a Poison
Control Center
Directions
1 Slowly peel back label and use one wipe. Reseal, keep closed to
prevent evaporation.I I Thoroughly wipe hands, allow to dry without rinsing.
I 1 Discard after single use e I Use within a month of opening
Other information
■Do not fush down toilet.
■Keep away from sunlight.
Store at room temperature 20° 25°Celsius (68°-77° Fahrenheit)
■May discolor certain fabrics
| ANTIBACTERIAL WET WIPES
alcohol cloth |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - YUYAO JESSIE COMMODITY CO.,LTD (529892305) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| YUYAO JESSIE COMMODITY CO.,LTD | 529892305 | manufacture(51414-911) | |