LORATADINE- loratadine oral solution
Loratadine by
Drug Labeling and Warnings
Loratadine by is a Otc medication manufactured, distributed, or labeled by BluePoint Laboratories, Aurobindo Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
- Uses
- Warnings
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- use only with enclosed dosing cup
- mL= milliliter
adults and children 6 years and over 10mL daily; do not take more than 10mL in 24 hours children 2 to under 6 years of age 5mL daily; do not take more than 5mL in 24 hours children under 2 years of age ask a doctor consumers with liver or kidney disease ask a doctor - Other information
- Inactive ingredients
- Questions or comments?
-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/5 mL (120 mL Bottle)
 NDC: 68001-449-98
NDC: 68001-449-98
Ages
2 years
and olderLoratadine
Oral Solution USP
5 mg/5 mL
Antihistamine
Non-Drowsy*
24 Hour Relief of:- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
Do not use if carton is opened,
or if cap safety seal is broken
or missing.- Dye-Free
- Sugar-Free
- Alcohol Free
Indoor & Outdoor Allergies
Contains sodium metabisulfite,
a sulfite that may cause
allergic-type reactions.* When taken as directed. See Drug Facts Panel.
Grape Flavor
4FL OZ (120 mL)
-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/5 mL Carton (120 mL)
 NDC: 68001-449-98
NDC: 68001-449-98
#Compare to the
active ingredient in
children’s Claritin®
Ages2years
and older
Loratadine
Oral Solution USP
5 mg/5 mL
Antihistamine
Non-Drowsy*
24 Hour Relief of:- Sneezing
- Runny Nose
- Itchy,watery Eyes
- Itchy Throat or Nose
Indoor & Outdoor Allergies
Dosing Cup Included- Dye-Free
- Sugar-Free
- Alcohol Free
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions.
* When taken as directed.
See Drug Facts Panel.
GrapeFlavor
4 FL OZ (120 mL)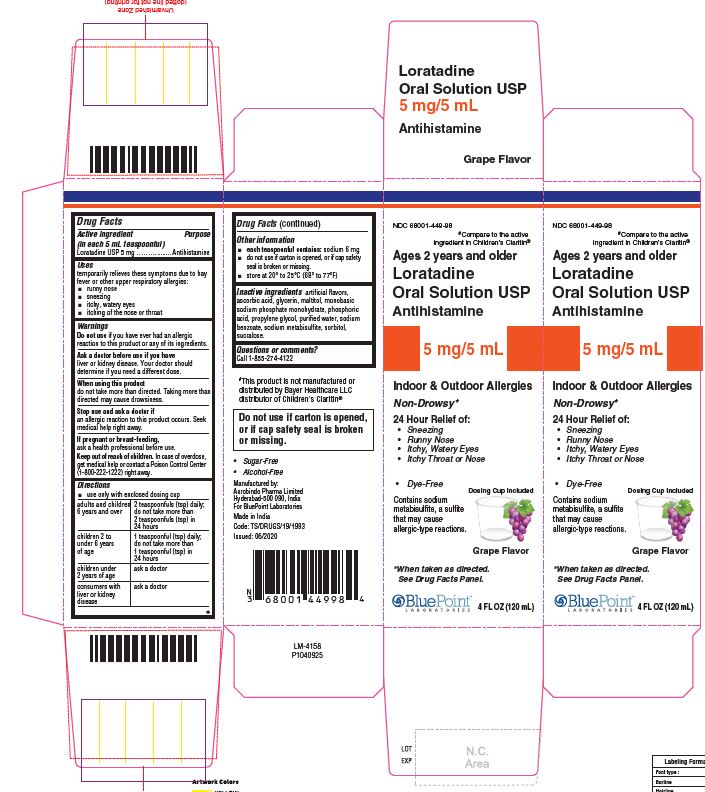
-
INGREDIENTS AND APPEARANCE
LORATADINE
loratadine oral solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68001-449 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 5 mg in 5 mL Inactive Ingredients Ingredient Name Strength GRAPE (UNII: 6X543N684K) ASCORBIC ACID (UNII: PQ6CK8PD0R) GLYCERIN (UNII: PDC6A3C0OX) MALTITOL (UNII: D65DG142WK) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) PHOSPHORIC ACID (UNII: E4GA8884NN) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM METABISULFITE (UNII: 4VON5FNS3C) SORBITOL (UNII: 506T60A25R) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color yellow (colorless to light yellow) Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68001-449-98 1 in 1 CARTON 07/31/2020 1 120 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208931 07/31/2020 Labeler - BluePoint Laboratories (985523874) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 918917642 analysis(68001-449) , manufacture(68001-449)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.