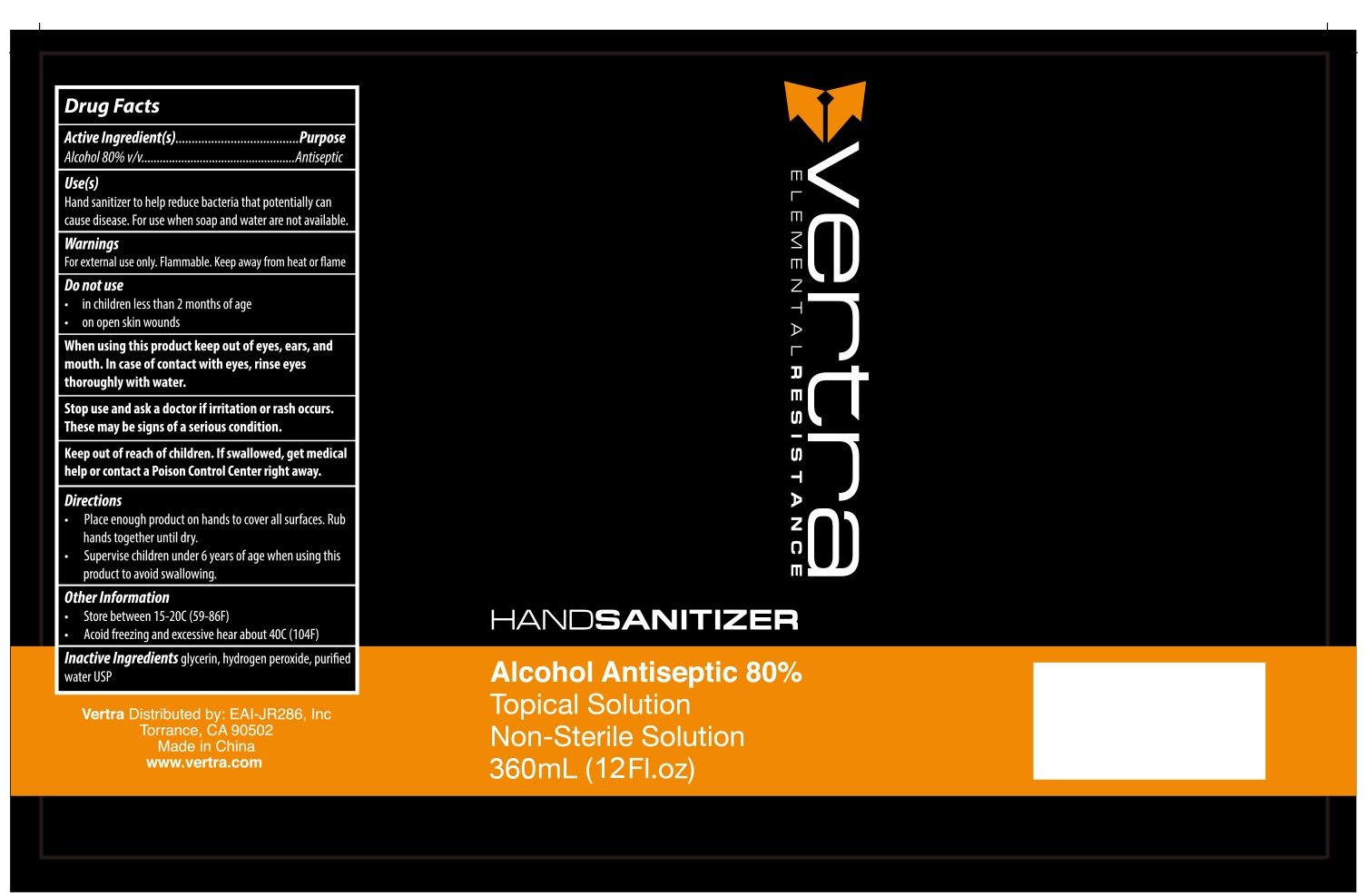
79740-003 Vertra Hand Sanitizer topical solution
Vertra Hand Sanitizer topical solution by
Drug Labeling and Warnings
Vertra Hand Sanitizer topical solution by is a Otc medication manufactured, distributed, or labeled by ANTHOS GROUP, INC, THE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
VERTRA HAND SANITIZER TOPICAL SOLUTION- hand sanitizer topical solution liquid
ANTHOS GROUP, INC, THE
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
79740-003
Vertra Hand Sanitizer topical solution
Use
Hand sanitizer to help reduce bacteria that potentially can cause disease. For use when soap and water are not available
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Keep out of reach of children If swallowed, get medical help or contact a Poison Control Center right away
| VERTRA HAND SANITIZER TOPICAL SOLUTION
hand sanitizer topical solution liquid |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - ANTHOS GROUP, INC, THE (117511051) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ANTHOS GROUP, INC, THE | 117511051 | manufacture(79740-003) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.