AVOPEF- etoposide injection
Avopef by
Drug Labeling and Warnings
Avopef by is a Prescription medication manufactured, distributed, or labeled by Avyxa Pharma, LLC, Avyxa Holdings, LLC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use AVOPEF safely and effectively. See full prescribing information for AVOPEF.
AVOPEFTM (etoposide) injection, for intravenous use
Initial U.S. Approval: 1983WARNING: SEVERE MYELOSUPPRESSION
See full prescribing information for complete boxed warning.
- AVOPEF can cause severe myelosuppression resulting in infection or bleeding. (5.1)
- Do not administer AVOPEF to patients with absolute neutrophil counts of less than 500 cells/mm3or platelets less than 50,000 cells/mm3. (5.1)
- Monitor complete blood cell counts, prior to the administration of AVOPEF and before each subsequent cycle, and at appropriate intervals during and after therapy. (5.1)
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- Refractory Testicular Cancer: 50 mg/m2 to 100 mg/m2 administered intravenously daily on Days 1 to 5, or 100 mg/m2 administered intravenously daily on Days 1, 3 and 5. (2.2)
- Small Cell Lung Cancer: 35 mg/m2 administered intravenously daily on days 1 to 4, or 50 mg/m2 administered intravenously daily on days 1 to 5. (2.3)
- Dilute AVOPEF prior to intravenous infusion over 30- to 60-minutes. (2.5)
DOSAGE FORMS AND STRENGTHS
- Injection: 100 mg/5 mL (20 mg/mL) in a multiple-dose vial. (3)
CONTRAINDICATIONS
Hypersensitivity to etoposide or any of its excipients. (4)
WARNINGS AND PRECAUTIONS
- Severe Myelosuppression: Monitor complete blood counts before each AVOPEF administration and at appropriate intervals during and after therapy. Interrupt AVOPEF for absolute neutrophil counts below 500 cells/mm3or platelet counts below 50,000 cells/mm3. (5.1)
- Hypersensitivity and Infusion-Related Reactions: At the first sign of hypersensitivity, stop the infusion and administer volume expanders, corticosteroids, antihistamines, and pressor agents as appropriate. Permanently discontinue AVOPEF in patients who experience a severe hypersensitivity reaction. Hypotension has occurred after rapid intravenous injection. (5.2)
- Extravasation Resulting in Tissue Necrosis: Extravasation of etoposide can result in swelling, pain, cellulitis, and tissue necrosis. (5.3)
- Secondary Leukemia: Secondary leukemia has occurred with use of etoposide. (5.4)
- Risk of Increased AVOPEF Toxicity with Low Serum Albumin: Monitor serum albumin during treatment with AVOPEF. Patients with low serum albumin may have increased concentrations of unbound etoposide and may be at an increased risk for etoposide associated adverse reactions. (5.5)
- Alcohol Content: The alcohol content in a dose of AVOPEF may affect the central nervous system. This may include impairment of a patient's ability to drive or use machines immediately after infusion. (5.6)
- Embryo-Fetal Toxicity: AVOPEF can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. (5.7)
ADVERSE REACTIONS
The most common adverse reactions are myelosuppression, hypersensitivity, nausea/vomiting, and alopecia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Avyxa Pharma, LLC at 1-888-520-0954 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- CYP3A Inhibitors and CYP3A Inducers: Avoid concomitant use of strong CYP3A inhibitors and strong CYP3A inducers with AVOPEF. (7.1)
- Vitamin K Antagonists: Monitor INR more frequently and modify the dosage of the vitamin K antagonists as appropriate. Co-administration of AVOEPF with warfarin can result in elevated international normalized ratio (INR). (7.2)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: SEVERE MYELOSUPPRESSION
1 INDICATIONS AND USAGE
1.1 Refractory Testicular Cancer
1.2 Small Cell Lung Cancer
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Information
2.2 Recommended Dosage for Refractory Testicular Cancer
2.3 Recommended Dosage for Small Cell Lung Cancer
2.4 Recommended Dosage in Patients with Renal Impairment
2.5 Preparation for Intravenous Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Severe Myelosuppression
5.2 Hypersensitivity and Infusion-Related Reactions
5.3 Extravasation Resulting in Tissue Necrosis
5.4 Secondary Leukemia
5.5 Risk of Increased AVOPEF Toxicity with Low Serum Albumin
5.6 Alcohol Content
5.7 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on AVOPEF
7.2 Effect of AVOPEF on Other Drugs
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: SEVERE MYELOSUPPRESSION
- AVOPEF can cause severe myelosuppression resulting in infection or bleeding [see Warnings and Precautions (5.1)].
- Do not administer AVOPEF to patients with absolute neutrophil counts of less than 500 cells/mm3or platelets less than 50,000 cells/mm3[see Warnings and Precautions (5.1)].
- Monitor complete blood cell counts, prior to the administration of AVOPEF and before each subsequent cycle, and at appropriate intervals during and after therapy [see Warnings and Precautions (5.1)].
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Information
- Dilute AVOPEF to a final concentration of 0.2 to 0.4 mg/mL prior to administration [see Dosage and Administration (2.5)].
- Administer diluted AVOPEF by intravenous infusion over 30 to 60 minutes to reduce the risk of infusion-related reactions including hypotension [see Dosage and Administration (2.5)].
- Before each AVOPEF administration and at appropriate intervals during and after therapy, monitor complete blood counts with differential and serum albumin [see Warnings and Precautions (5.1,5.5)].
- If severe reactions occur, reduce the dosage or discontinue AVOPEF and take appropriate corrective measures according to the clinical judgment of the healthcare provider.
2.2 Recommended Dosage for Refractory Testicular Cancer
The recommended dosage of AVOPEF is:
- 50 mg/m2 to 100 mg/m2 administered intravenously daily on Days 1 to 5, or
- 100 mg/m2 administered intravenously daily on Days 1, 3 and 5
2.3 Recommended Dosage for Small Cell Lung Cancer
The recommended dosage of AVOPEF is:
- 35 mg/m2 administered intravenously daily on Days 1 to 4, or
- 50 mg/m2administered intravenously daily on Days 1 to 5
2.4 Recommended Dosage in Patients with Renal Impairment
No dosage modification is recommended for patients with creatinine clearance (CLcr) > 50 mL/min. The recommended dosage of AVOPEF in patients with CLcr of 15 to 50 mL/min is listed in Table 1.
Table 1: Recommended Dosage of AVOPEF in Patients with Creatine Clearance of 15 to 50 mL/min *Repeat treatment cycles every 3 to 4 weeks.
Recommended AVOPEF Dosage*
Refractory Testicular Cancer
37 mg/m2 to 75 mg/m2
Days 1 through 5
75 mg/m2
Days 1, 3 and 5
Small Cell Lung Cancer
26 mg/m2
Days 1 through 4
37 mg/m2
Days 1 through 5
A recommended dosage of AVOPEF has not been established for patients with creatinine clearance < 15 mL/min.
2.5 Preparation for Intravenous Administration
- AVOPEF is a hazardous drug. Follow applicable special handling and disposal procedures. [see References (15)].
- Dilute AVOPEF with either 5% Dextrose Injection, USP, or 0.9% Sodium Chloride Injection, USP, to a final concentration of 0.2 to 0.4 mg/mL. If solutions of AVOPEF are prepared at concentrations above 0.4 mg/mL, precipitation may occur.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Do not administer AVOPEF by rapid intravenous injection. To reduce the risk of infusion-related reactions including hypotension, administer diluted AVOPEF intravenously over 30 to 60 minutes. A longer duration of administration may be used if there is a large volume of fluid to be infused.
- AVOPEF, diluted to a concentration of 0.2 mg/mL is stable for 96 hours or diluted to a concentration of 0.4 mg/mL is stable for 24 hours at room temperature (25°C) under normal room fluorescent light in polyvinyl chloride (PVC) container.
- After first use, store the partially used multiple-dose vial in the original carton at controlled room temperature, 20°C to 25° C (68°F to 77° F) for up to 28 days. Discard unused portion of the multiple-dose vial after 28 days.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Severe Myelosuppression
AVOPEF can cause severe and fatal myelosuppression, including neutropenia, febrile neutropenia, anemia, and thrombocytopenia.
Monitor complete blood counts with differential before each AVOPEF administration and at appropriate intervals during and after treatment with AVOPEF. Do not administer AVOPEF to patients with absolute neutrophil counts of less than 500 cells/mm3or platelets less than 50,000 cells/mm3.
5.2 Hypersensitivity and Infusion-Related Reactions
AVOPEF can cause severe and fatal infusion-related reactions including anaphylactic reactions characterized by chills, fever, tachycardia, bronchospasm, dyspnea and hypotension [see Adverse Reactions (6.1)]. Hypertension and flushing have occurred.
At the first sign of hypersensitivity, stop the infusion and administer volume expanders, corticosteroids, antihistamines, and pressor agents as appropriate. Permanently discontinue AVOPEF in patients who experience a severe hypersensitivity reaction. Hypotension due to rapid intravenous injection has also occurred. To reduce the risk of hypotension due to an infusion-related reaction, administer AVOPEF by intravenous infusion over 30 to 60 minutes [see Dosage and Administration (2.5)].
5.3 Extravasation Resulting in Tissue Necrosis
Extravasation of etoposide can result in swelling, pain, cellulitis, and tissue necrosis.
5.5 Risk of Increased AVOPEF Toxicity with Low Serum Albumin
Etoposide is highly protein-bound. Patients with low serum albumin may have increased concentrations of unbound etoposide and may be at an increased risk for etoposide associated adverse reactions. Monitor for increased adverse reactions during treatment with AVOPEF in patients with low serum albumin.
5.6 Alcohol Content
The alcohol content in a dose of AVOPEF may affect the central nervous system and should be taken into account for patients in whom alcohol intake should be avoided or minimized. Consideration should be given to the alcohol content in AVOPEF on the ability to drive or use machines immediately after the infusion. Each administration of AVOPEF at 100 mg/m2 delivers 1.5 g/m2 of ethanol. For a patient with a BSA of 2.0 m2 this would deliver 3.0 grams of ethanol [see Description (11)]. Other etoposide products may have a different amount of alcohol or no alcohol.
5.7 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, AVOPEF can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, intravenous or intraperitoneal administration of etoposide to pregnant animals during the period of organogenesis caused embryo-fetal mortality and structural abnormalities at doses below the recommended human dose of 50 mg/m2 based on body surface area (BSA).
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise female patients of reproductive potential to use effective contraception during treatment with AVOPEF and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with AVOPEF and for 4 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in more detail in other sections of the labeling.
- Severe Myelosuppression [see Warnings and Precautions (5.1)]
- Hypersensitivity and Infusion-Related Reactions [see Warnings and Precautions (5.2)]
- Extravasation Resulting in Tissue Necrosis [see Warnings and Precautions (5.3)]
- Secondary Leukemia [see Warnings and Precautions (5.4)]
- Risk of Increased AVOPEF Toxicity with Low Serum Albumin [see Warnings and Precautions (5.5)]
- Alcohol Content [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Blood and lymphatic system disorders: acute leukemia, myelosuppression
Eye disorders: transient cortical blindness
Gastrointestinal disorders: abdominal pain, anorexia, constipation, diarrhea, dysgeusia, dysphagia, esophagitis, mucositis, nausea, stomatitis and vomiting
General disorders and administration site conditions: fatigue, fever, and infusion site extravasation with necrosis
Hepatobiliary disorders: hepatoxicity, metabolic acidosis
Immune system disorders: allergic reaction, anaphylactic reaction (including chills, fever, tachycardia, bronchospasm, dyspnea, hypotension, hypertension, flushing, facial swelling, tongue swelling, coughing, diaphoresis, cyanosis, tightness in throat, laryngospasm, back pain, loss of consciousness, hypersensitivity-associated apnea), hypersensitivity, infusion-related reaction.
Infections: febrile neutropenia
Nervous system disorders: optic neuritis, peripheral neuropathy and seizure
Respiratory, thoracic and mediastinal disorders: interstitial pneumonitis and pulmonary fibrosis
Skin and subcutaneous tissue disorders: alopecia, pigmentation changes (including pigmented bands in nails, skin darkening and discoloration of tongue and teeth), pruritic erythematous maculopapular rash, pruritus, radiation recall dermatitis, rash, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN) and urticaria
Vascular disorders: hypotension following rapid intravenous administration [see Dosage and Administration (2.5)], perivasculitis
-
7 DRUG INTERACTIONS
7.1 Effect of Other Drugs on AVOPEF
Avoid concomitant use of strong CYP3A inhibitors.
Etoposide is a CYP3A4 substrate. Strong CYP3A inhibitors may increase etoposide exposure, which may increase the risk of AVOPEF-associated adverse reactions [see Clinical Pharmacology (12.3)].
CYP3A Inducers
Avoid concomitant use of strong CYP3A inducers.
Etoposide is a CYP3A4 substrate. Strong CYP3A inducers may reduce etoposide exposure, which may decrease the effectiveness of AVOPEF [see Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Based on findings in animal studies and its mechanism of action [see Clinical Pharmacology (12.1)], AVOPEF can cause fetal harm when administered to a pregnant woman. There are no available data on the use of AVOPEF in pregnant women to inform a drug-associated risk. AVOPEF contains alcohol which can interfere with neurobehavioral development [see Clinical Considerations]. In animal reproduction studies, intravenous or intraperitoneal administration of etoposide to pregnant animals during the period of organogenesis caused embryo-fetal mortality and structural abnormalities at doses below the recommended human dose of 50 mg/m2 based on body surface area (BSA). Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise female patients of reproductive potential to use effective contraception during treatment with AVOPEF and for 6 months after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)]. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and of miscarriage is 15% to 20%, respectively.
Clinical Considerations
AVOPEF contains alcohol [see Warnings and Precautions (5.6)]. Published studies have demonstrated that alcohol is associated with fetal harm including central nervous system abnormalities, behavioral disorders, and impaired intellectual development.
Data
Animal Data
In rats, an intravenous etoposide dose of 0.4 mg/kg/day (approximately 0.05 times the human dose of 50 mg/m2 based on BSA) during organogenesis caused maternal toxicity, embryotoxicity, and teratogenicity (skeletal abnormalities, exencephaly, encephalocele, and anophthalmia); higher doses of 1.2 and 3.6 mg/kg/day (approximately 0.14- and 0.5-times the human dose of 50 mg/m2 based on BSA) resulted in 90% and 100% embryonic resorptions, respectively. In mice, a single etoposide dose of 1.0 mg/kg (approximately 0.06 times the human dose of 50 mg/m2 based on BSA) administered intraperitoneally on Days 6, 7, or 8 of gestation caused embryotoxicity, cranial abnormalities, and major skeletal malformations. An intraperitoneal dose of 1.5 mg/kg (approximately 0.10-times the human dose 50 mg/m2 based on BSA) on Day 7 of gestation caused an increase in the incidence of intrauterine death, fetal malformations, and a significant decrease in the average fetal body weight.
8.2 Lactation
There is no information regarding the presence of etoposide or its metabolites in human milk or the effects on a breastfed child or on milk production. Because the potential for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment with AVOPEF and for 1 week after the last dose.
8.3 Females and Males of Reproductive Potential
Based on animal data and its mechanism of action, AVOPEF can cause fetal harm when administered to pregnant women [see Use in Specific Population (8.1)].
Pregnancy Testing
Verify the pregnancy status of female patients of reproductive potential prior to initiating AVOPEF.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with AVOPEF and for 6 months after the last dose [see Use in Specific Populations (8.1) and Nonclinical Toxicology (13.1)].
Males
Due to the potential for genotoxicity, advise males with female partners of reproductive potential to use effective contraception during treatment with AVOPEF and for 4 months after the last dose [see Nonclinical Toxicology (13.1)].
Infertility
Females
In females of reproductive potential, AVOPEF may cause infertility and result in amenorrhea. Premature menopause can occur with AVOPEF. Recovery of menses and ovulation is related to age at treatment.
Males
In male patients, AVOPEF may result in oligospermia, azoospermia, and permanent loss of fertility. Sperm counts have been reported to return to normal levels in some men, and in some cases, have occurred several years after the end of therapy [See Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of AVOPEF have not been established in pediatric patients.
8.5 Geriatric Use
Clinical studies of etoposide for the treatment of refractory testicular tumors did not include sufficient numbers of patients aged 65 years and older to determine whether they respond differently from younger patients. Other reported clinical experience demonstrated that patients 65 years and older experienced more myelosuppression, anorexia, mucositis, dehydration, somnolence, elevated blood urea nitrogen (BUN), infectious complications and alopecia compared to younger patients.
In general, dosage selection for an older patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
Reduce the dose of AVOPEF in patients with creatinine clearance (CLcr) of 15 to 50 mL/min [see Dosage and Administration (2.4)]. No dosage modification is recommended for patients with CLcr > 50 mL/min.
-
11 DESCRIPTION
AVOPEF injection contains etoposide, a topoisomerase inhibitor. The chemical name for etoposide is 4'-Demethylepipodophyllotoxin 9-[4,6-O-(R)-ethylidene-β-D glucopyranoside] and has the following structural formula:
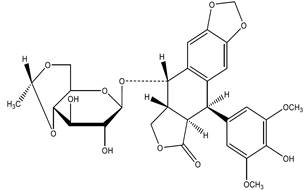
Etoposide is a white or off-white crystalline powder with the molecular formula C29H32O13 and a molecular weight of 588.56. It is sparingly soluble in acetone, slightly soluble in methanol or in chloroform, very slightly soluble in ethanol and practically insoluble in water.
AVOPEF (etoposide) Injection is a sterile, clear, colorless to light yellow liquid available in 100 mg/5 mL multiple-dose vials containing 20 mg/mL of etoposide for intravenous administration. Each mL contains 20 mg etoposide, 37.8% v/v (1490 mg) dehydrated alcohol, 600 mg polyethylene glycol 400, and 80 mg polysorbate 80. The pH is adjusted with anhydrous citric acid and is between 3 to 4. Vial headspace contains nitrogen.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Etoposide causes the induction of DNA strand breaks by an interaction with DNA-topoisomerase II or the formation of free radicals, leading to cell cycle arrest, primarily at the G2 stage of the cell cycle, and cell death.
12.2 Pharmacodynamics
Etoposide exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
Etoposide total systemic exposure (AUC) and the maximum plasma concentration (Cmax) increases in dose proportional manner over dosage range of 100 to 600 mg/m2 (6 times the maximum approved recommended dose). Etoposide does not accumulate following daily administration of 100 mg/m2 for 4 to 5 days.
Distribution
Etoposide mean volume of distribution at steady state is 7 L/m2 to 17 L/m2.
No clinically significant amount of etoposide distributes into the CSF.
Etoposide plasma protein binding is 97% in vitro (primarily albumin).
Elimination
Etoposide elimination half-life is 4 hours to 11 hours. Total body clearance is 16 mL/min/m2 to 36 mL/min/m2 over a range of 100 mg/m2 to 600 mg/m2 (6 times the maximum approved recommended dose). The mean renal clearance of etoposide is 7 mL/min/m2 to 10 mL/min/m2 over a dose range of 80 mg/m2 to 600 mg/m2 (6 times the maximum approved recommended dose).
Metabolism
Etoposide is metabolized by opening of the lactone ring, O-demethylation and conjugation (i.e., glucuronidation and sulfation. O-demethylation occurs through CY3A to the active catechol metabolite.
Excretion
After intravenous administration of 14C-etoposide [100 to 124 mg/m2 (1.2 times the maximum approved recommended dose)], mean recovery of radioactivity in the urine was 56% of the dose at 120 hours, 45% of which was excreted as etoposide; fecal recovery of radioactivity was 44% of the dose at 120 hours.
Specific Populations
No clinically significant differences in pharmacokinetic parameters of etoposide were observed based on age and sex.
Patients with Renal Impairment
Patients with impaired renal function have exhibited reduced total body clearance, increased AUC, and a lower volume of distribution at steady state.
Drug Interactions
In vitro studies
Phenylbutazone, sodium salicylate, and aspirin displaced protein-bound etoposide in vitro.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A single intravenous dose of etoposide at ≥ 4 times the human dose of 50 mg/m2 based on BSA caused benign and malignant breast tumors in female rats and mice.
Mutagenesis
Etoposide was mutagenic in an in vitro bacterial reverse mutation (Ames) assay.
Impairment of Fertility
Irreversible testicular atrophy was present in rats treated with etoposide intravenously for 30 days at 0.5 mg/kg/day (approximately 0.06 times the human dose of 50 mg/m2 based on BSA).
A single intravenous dose of etoposide at approximately 5 times the human dose of 50 mg/m2 based on BSA caused accelerated atrophic processes in the ovaries of rats, 3 months post-treatment. In vitro, etoposide treatment resulted in damage of pre-follicular fetal ovaries.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
AVOPEF (etoposide) Injection, 100 mg/5 mL (20 mg/mL) is a sterile, clear, colorless to light yellow liquid available in multiple-dose glass vials as follows:
NDC Number
Strength
Package
83831-144-05
100 mg/5 mL (20 mg/mL)
1 multiple-dose vial in 1 carton.
This container closure is not made with natural rubber latex.
Storage and Handling
Store at 20°C to 25° C (68°F to 77° F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature]. After first use, store the partially used multiple-dose vial in the original carton at controlled room temperature, 20°C to 25°C (68°F to 77°F) for up to 28 days [see Dosage and Administration (2.5)].
AVOPEF is a hazardous drug. Follow applicable special handling and disposal procedures [see References (15)].
-
17 PATIENT COUNSELING INFORMATION
Inform patients that AVOPEF can cause severe myelosuppression. Advise patients that periodic monitoring of their blood counts is required during treatment with AVOPEF. Advise patients to contact their healthcare provider for any signs or symptoms of myelosuppression [see Warnings and Precautions (5.1)].
Hypersensitivity and Infusion-Related Reactions
Inform patients that AVOPEF can cause severe infusion-related reactions, allergic reactions and anaphylaxis. Advise patients to immediately call their healthcare provider to report any signs or symptoms of an infusion-related reaction, allergic reaction or anaphylaxis [see Warnings and Precautions (5.2)].
Risk of Increased AVOPEF Toxicity with Low Serum Albumin
Inform patients that the risk of AVOPEF toxicity is increased with low serum albumin. Advise patients that periodic monitoring of their serum albumin is required during treatment with AVOPEF. Advise patients to contact their healthcare provider if they experience signs or symptoms of low serum albumin or AVOPEF toxicity [see Warnings and Precautions (5.5)].
Secondary Leukemia
Inform patients that AVOPEF can cause secondary leukemia. Advise patients that periodic monitoring is recommended during and after treatment with AVOPEF and to contact their healthcare provider if they experience signs or symptoms of leukemia [see Warnings and Precautions (5.4)].
Alcohol Content
Advise patients of the effects of alcohol in AVOPEF, including possible effects on the central nervous system. Advise patients in whom alcohol should be avoided or minimized to consider the alcohol content of AVOPEF. AVOPEF at 100 mg/m2 delivers 1.5 g/m2 of ethanol. For a patient with a BSA of 2.0 m2 this delivers 3.0 grams of ethanol [see Description (11)]. Alcohol impairs the ability to drive or use machines [see Warnings and Precautions (5.6)].
Embryo-Fetal Toxicity
- Advise pregnant women and females of reproductive potential of the potential risk to a fetus and to inform their healthcare provider with a known or suspected pregnancy [see Warnings and Precautions (5.7) and Use in Specific Populations (8.1)].
- Advise females of reproductive potential to use effective contraception during treatment with AVOPEF and for 6 months after the last dose [see Use in Specific Populations (8.3)].
- Advise males with female partners of reproductive potential to use effective contraception during treatment with AVOPEF and for 4 months after the last dose [see Nonclinical Toxicology (13.1)].
Advise women not to breastfeed during treatment with AVOPEF and for 1 week after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise males and females of reproductive potential that AVOPEF may impair fertility [see Use in Specific Populations (8.3) and Nonclinical Toxicology (13.1)].
Manufactured for:
Avyxa Pharma, LLC
New Jersey 07054, USA
Made in China

- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AVOPEF
etoposide injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 83831-144 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ETOPOSIDE (UNII: 6PLQ3CP4P3) (ETOPOSIDE - UNII:6PLQ3CP4P3) ETOPOSIDE 100 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) DEHYDRATED ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 83831-144-05 1 in 1 CARTON 04/01/2026 1 5 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220200 04/01/2026 Labeler - Avyxa Pharma, LLC (128918748) Registrant - Avyxa Holdings, LLC. (119187191)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

