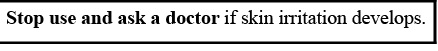SafetyPlus Hand Sanitizer with Aloe 70% 32oz
70% SafetyPlus Hand Sanitizer 32oz by
Drug Labeling and Warnings
70% SafetyPlus Hand Sanitizer 32oz by is a Otc medication manufactured, distributed, or labeled by SafetyPlus, Inc., Orange Lab, Inc., Orange Lab, Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
70% SAFETYPLUS HAND SANITIZER 32OZ- ethyl alcohol gel
SafetyPlus, Inc.
----------
SafetyPlus Hand Sanitizer with Aloe 70% 32oz
| 70% SAFETYPLUS HAND SANITIZER 32OZ
ethyl alcohol gel |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - SafetyPlus, Inc. (122313405) |
| Registrant - Orange Lab, Inc. (004862271) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Orange Lab, Inc | 004862271 | manufacture(80174-109) | |
Revised: 4/2026
Document Id: 4f10aaab-4854-44ad-e063-6394a90a274c
Set id: ad40ee47-3810-9fa4-e053-2995a90a6b8e
Version: 5
Effective Time: 20260409
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.