Loratadine by BluePoint Laboratories / Aurobindo Pharma Limited LORATADINE tablet
Loratadine by
Drug Labeling and Warnings
Loratadine by is a Otc medication manufactured, distributed, or labeled by BluePoint Laboratories, Aurobindo Pharma Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Active ingredient (in each tablet)
- Purpose
- Uses
- Warnings
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding
- Keep out of reach of children.
- Directions
- Other Information
- Inactive ingredients
- Questions or comments?
-
PACKAGE LABEL- PRINCIPAL DISPLAY PANEL - 10mg (100 Tablets Bottle)
NDC: 68001-438-00
Non-Drowsy*
Loratadine
Tablets USP 10mg
Antihistamine
24 Hour
Relief of:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
Indoor & Outdoor
Allergies
# When taken as directed.
See Drug Facts Panel. 100 Tablets

-
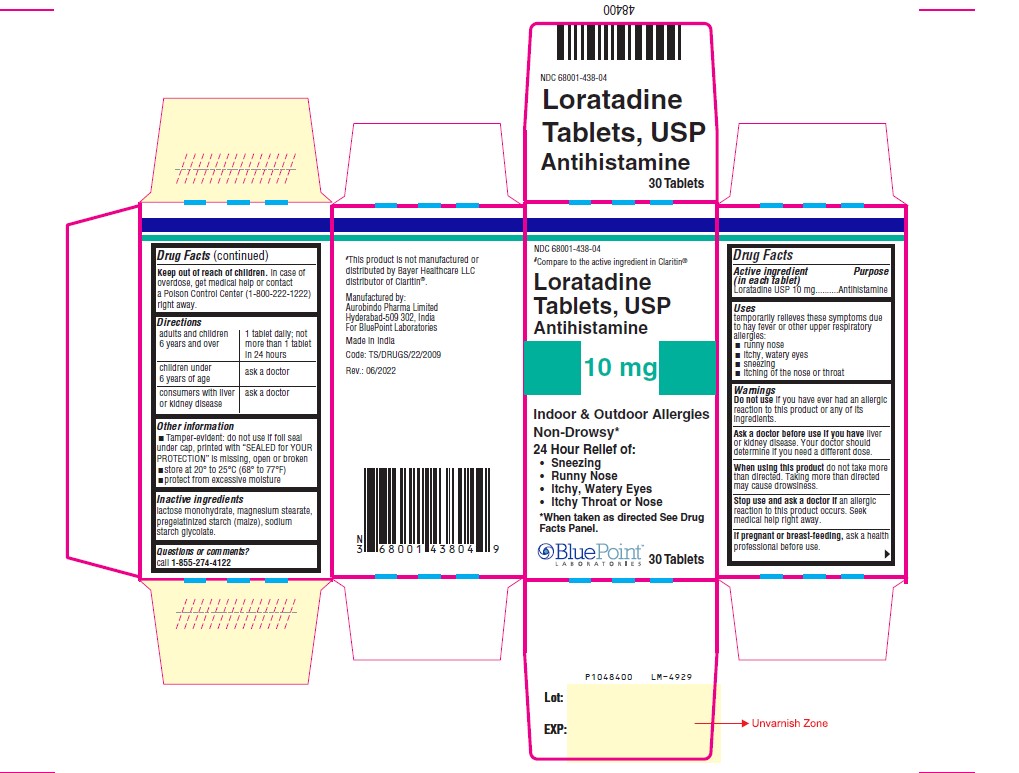
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 10 mg Container Carton (30 Tablets)
NDC: 68001-438-04
#Compare to the active ingredient in claritin®
Non-Drowsy*
Loratadine
Tablets USP 10mg
Antihisamine
Indoor and outdoor allergies
24 Hour
Relief of:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
*When taken as directed.
See Drug Facts Panel. 30 Tablets
-
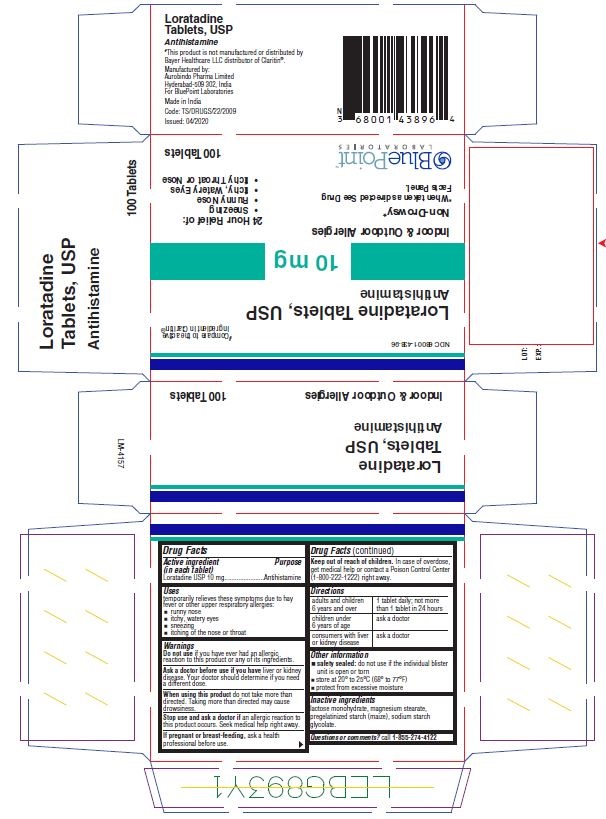
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL- 10 mg Blister Carton (100 Tablets)
NDC: 68001-438-96
#Compare to the active
ingredient in claritin®
Non-Drowsy*
Loratadine
Tablets USP 10mg
Antihisamine
Indoor and outdoor allergies
24 Hour
Relief of:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
*When taken as directed.
See Drug Facts Panel. 100 Tablets
-
INGREDIENTS AND APPEARANCE
LORATADINE
loratadine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68001-438 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color white (White to off-white) Score no score Shape ROUND Size 6mm Flavor Imprint Code 39;L Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68001-438-00 1 in 1 CARTON 08/26/2020 1 100 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC: 68001-438-96 10 in 1 CARTON 08/26/2020 2 NDC: 68001-438-16 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 68001-438-04 1 in 1 CARTON 08/26/2020 3 30 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC: 68001-438-97 1 in 1 CARTON 08/26/2020 4 300 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA208314 08/26/2020 Labeler - BluePoint Laboratories (985523874) Establishment Name Address ID/FEI Business Operations Aurobindo Pharma Limited 650381903 analysis(68001-438) , manufacture(68001-438)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.