Topical Anti-Fungal Solution by Wintermute Biomedical, Inc. / Wintermute Biomedical, Inc / Mayne Pharma International Pty Ltd Drug Facts
Topical Anti-Fungal Solution by
Drug Labeling and Warnings
Topical Anti-Fungal Solution by is a Otc medication manufactured, distributed, or labeled by Wintermute Biomedical, Inc., Wintermute Biomedical, Inc, Mayne Pharma International Pty Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
TOPICAL ANTI-FUNGAL SOLUTION- undecylenic acid liquid
Wintermute Biomedical, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
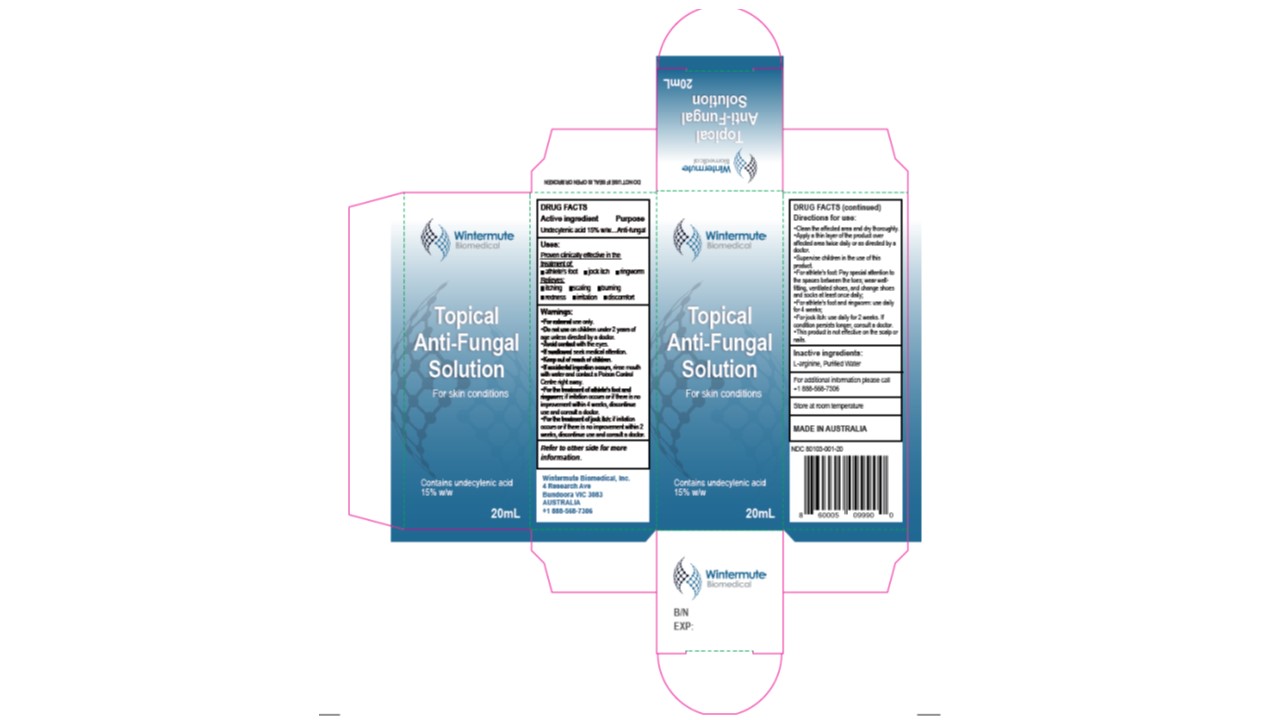
Drug Facts
Uses
Proven effective in the treatment of:
* Athlete's foot
* Jock itch
* Ringworm
Relieves:
* Itchine
*Scaling
* Burning
* Redness
* Irritation
* Discomfort
Warnings
* For external use only
* Do not use on children under 2 years of age unless directed by a doctor
* Avoid contact with the eyes
* If swallowed seek medical attention
* Keep out of reach of children
* If accidental ingestion occurs, rinse mouth with water and contact a Poison Control Center right away
* For the treatment of athlete's foot and ringworm; if irritation offurs or if there is no improvement within 4 weeks, discontinue use and consult a doctor.
* For the treatment of jock itch: if irritation occurs of it there is no improvement within 2 weeks, discontinue use and consult a doctor.
Ask doctor section
* For treatment of athlete's foot and ringworm; if irritation occurs or if there is no improvement within 4 weeks, discontinue use and consult a doctor.
* For the treatment of jock itch: if irritation occurs or it there is no improvement within 2 weeks, discontinue use and consult a doctor.
Directions
Directions of Use:
* Clean the affected area and dry thoroughly
* Apply a thin layer or the product over affected area twise daily or as directed by a doctor.
* Supervise children in the use of this product
* For athlete's foot: Pay special attention to the spaces between the toes; wear well-fitting, ventilated shoes, and change shoes and socks at least once daily.
* For athlete's foot and ringworm: use daily for 4 weeks
* For Jock itch: use daily for 2 weeks. If conditions persists longer, consult a doctor.
* This product is not effective on the scalp or nails.
Prinicipal Display Panel
Wintermute Biomedical
Topical Anti-Fungal Solution
For skin conditions
Contains Undecylenic acid 15%W/W
Store at room temperature
Wintermute Biomedical Inc.
4 Research Ave
Bundroora VIC 3083
Australia
+1-888-568-7306
Made in Australia
NDC: 80103-001-xx
| TOPICAL ANTI-FUNGAL SOLUTION
undecylenic acid liquid |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Wintermute Biomedical, Inc. (078573246) |
| Registrant - Wintermute Biomedical, Inc. (078573246) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Wintermute Biomedical, Inc. | 078573246 | pack(80103-001) , label(80103-001) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Mayne Pharma International Pty Ltd | 756003745 | manufacture(80103-001) | |