SPOT CARE PINK ONE by Leejiham 80395-202
SPOT CARE PINK ONE by
Drug Labeling and Warnings
SPOT CARE PINK ONE by is a Otc medication manufactured, distributed, or labeled by Leejiham. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
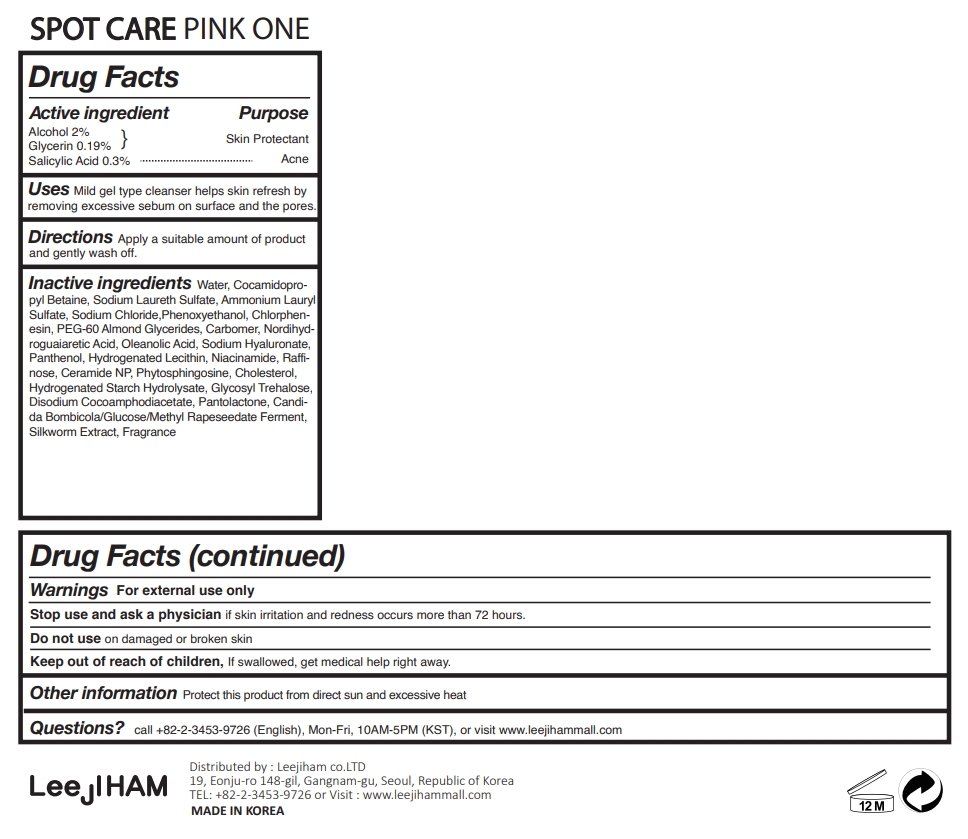
SPOT CARE PINK ONE- alcohol, salicylic acid, glycerin liquid
Leejiham
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
80395-202
Uses
Mild gel type cleanser helps skin refresh by removing excessive sebum on surface and the pores.
Inactive ingredients
Water, Cocamidopropyl Betaine, Sodium Laureth Sulfate, Ammonium Lauryl Sulfate, Sodium Chloride, Alcohol, Phenoxyethanol, Salicylic Acid, Chlorphenesin, PEG-60 Almond Glycerides, Glycerin, Carbomer, Nordihydroguaiaretic Acid, Oleanolic Acid, Sodium Hyaluronate, Panthenol, Hydrogenated Lecithin, Niacinamide, Raffinose, Ceramide NP, Phytosphingosine, Cholesterol, Hydrogenated Starch Hydrolysate, Glycosyl Trehalose, Disodium Cocoamphodiacetate, Pantolactone, Candida Bombicola/Glucose/Methyl Rapeseedate Ferment, Silkworm Extract, Fragrance
| SPOT CARE PINK ONE
alcohol, salicylic acid, glycerin liquid |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Leejiham (695004132) |
| Registrant - Leejiham (695004132) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Leejiham | 695004132 | manufacture(80395-202) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.