ANDEXXA- andexanet alfa injection, powder, lyophilized, for solution
ANDEXXA by
Drug Labeling and Warnings
ANDEXXA by is a Prescription medication manufactured, distributed, or labeled by Alexion Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ANDEXXA safely and effectively. See Full Prescribing Information for ANDEXXA.
ANDEXXA® (coagulation factor Xa (recombinant), inactivated-zhzo)
Lyophilized powder for solution for intravenous injection
Initial U.S. Approval: 2018WARNING: THROMBOEMBOLIC RISKS, ISCHEMIC RISKS, CARDIAC ARREST, AND SUDDEN DEATHS
See full prescribing information for complete boxed warning
Treatment with ANDEXXA has been associated with serious and life-threatening adverse events, including: (5.1)
- Arterial and venous thromboembolic events
- Ischemic events, including myocardial infarction and ischemic stroke
- Cardiac arrest
- Sudden deaths
Monitor for thromboembolic events and initiate anticoagulation when medically appropriate. Monitor for symptoms and signs that precede cardiac arrest and provide treatment as needed.
INDICATIONS AND USAGE
ANDEXXA (coagulation factor Xa (recombinant), inactivated-zhzo) is a recombinant modified human factor Xa (FXa) protein indicated for patients treated with rivaroxaban or apixaban, when reversal of anticoagulation is needed due to life-threatening or uncontrolled bleeding. (1)
This indication is approved under accelerated approval based on the change from baseline in anti-FXa activity in healthy volunteers. An improvement in hemostasis has not been established. Continued approval for this indication may be contingent upon the results of studies that demonstrate an improvement in hemostasis in patients. (1, 14)
Limitations of Use
ANDEXXA has not been shown to be effective for, and is not indicated for, the treatment of bleeding related to any FXa inhibitors other than apixaban or rivaroxaban. (1)
DOSAGE AND ADMINISTRATION
For intravenous (IV) use only.
- Dose ANDEXXA based on the specific FXa inhibitor, dose of FXa inhibitor, and time since the patient's last dose of FXa inhibitor. (2)
- Administer as an IV bolus, with a target rate of 30 mg/min, followed by continuous infusion for up to 120 minutes. (2.3)
- There are two dosing regimens:
Dose* Initial IV Bolus Follow-On IV Infusion - * The safety and effectiveness of more than one dose have not been evaluated. (2.1)
Low Dose 400 mg at a target rate of 30 mg/min 4 mg/min for up to 120 minutes High Dose 800 mg at a target rate of 30 mg/min 8 mg/min for up to 120 minutes
DOSAGE FORMS AND STRENGTHS
ANDEXXA is available as a lyophilized powder in single-use vials of 200 mg of coagulation factor Xa (recombinant), inactivated-zhzo. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Arterial and venous thromboembolic events, ischemic events, and cardiac events, including sudden death, have occurred during treatment with ANDEXXA. Resume anticoagulant therapy as soon as medically appropriate following treatment with ANDEXXA. (5.1)
- Re-elevation or incomplete reversal of anticoagulant activity can occur. (5.2)
ADVERSE REACTIONS
The most common adverse reactions (≥ 5%) in bleeding subjects receiving ANDEXXA were urinary tract infections and pneumonia. (6.1)
The most common adverse reactions (≥ 3%) in healthy volunteers treated with ANDEXXA were infusion-related reactions.
To report SUSPECTED ADVERSE REACTIONS, contact Portola Pharmaceuticals, Inc. at 1-866-777-5947 or 650-822-8884 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: THROMBOEMBOLIC RISKS, ISCHEMIC RISKS, CARDIAC ARREST, AND SUDDEN DEATHS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dose
2.2 Reconstitution
2.3 Administration
2.4 Restarting Anticoagulant Therapy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Thromboembolic and Ischemic Risks
5.2 Re-elevation or Incomplete Reversal of Anti-FXa Activity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: THROMBOEMBOLIC RISKS, ISCHEMIC RISKS, CARDIAC ARREST, AND SUDDEN DEATHS
Treatment with ANDEXXA has been associated with serious and life-threatening adverse events, including: (5.1)
- Arterial and venous thromboembolic events
- Ischemic events, including myocardial infarction and ischemic stroke
- Cardiac arrest
- Sudden deaths
Monitor for thromboembolic events and initiate anticoagulation when medically appropriate. Monitor for symptoms and signs that precede cardiac arrest and provide treatment as needed.
-
1 INDICATIONS AND USAGE
ANDEXXA is indicated for patients treated with rivaroxaban or apixaban, when reversal of anticoagulation is needed due to life-threatening or uncontrolled bleeding.
This indication is approved under accelerated approval based on the change from baseline in anti-FXa activity in healthy volunteers [see Clinical Studies (14)]. An improvement in hemostasis has not been established. Continued approval for this indication may be contingent upon the results of studies that demonstrate an improvement in hemostasis in patients.
-
2 DOSAGE AND ADMINISTRATION
For intravenous (IV) use only.
2.1 Dose
There are two dosing regimens (see Table 1). The safety and efficacy of an additional dose have not been established.
Table 1: ANDEXXA Dosing Regimens Dose* Initial IV Bolus Follow-On IV Infusion Total Number of 200 mg Vials - * The safety and effectiveness of more than one dose have not been evaluated.
Low Dose 400 mg at a target rate of 30 mg/min 4 mg/min for up to 120 minutes (480 mg) 5
(2 vials bolus + 3 vials infusion)High Dose 800 mg at a target rate of 30 mg/min 8 mg/min for up to 120 minutes (960 mg) 9
(4 vials bolus + 5 vials infusion)The recommended dosing of ANDEXXA is based on the specific FXa inhibitor, dose of FXa inhibitor, and time since the patient's last dose of FXa inhibitor (see Table 2).
Table 2: ANDEXXA Dose Based on Rivaroxaban or Apixaban Dose (Timing of Last Dose of FXa Inhibitor before ANDEXXA Initiation) FXa Inhibitor FXa Inhibitor Last Dose < 8 Hours or Unknown ≥ 8 Hours Rivaroxaban ≤ 10 mg Low Dose Low Dose > 10 mg or Unknown High Dose Apixaban ≤ 5 mg Low Dose > 5 mg or Unknown High Dose 2.2 Reconstitution
- The reconstituted solution contains coagulation factor Xa (recombinant), inactivated-zhzo at a concentration of 10 mg/mL.
- Reconstituted ANDEXXA in vials is stable at room temperature for up to eight hours, or may be stored for up to 24 hours at 2°C to 8°C.
- Reconstituted ANDEXXA in IV bags is stable at room temperature for up to eight hours.
IV Bolus Preparation
Determine total number of vials required (see Table 1).
200 mg vials:
Reconstitute the 200 mg vial of ANDEXXA with 20 mL of Sterile Water for Injection (SWFI).
Use a 20-mL (or larger) syringe and 20-gauge (or higher) needle.
Slowly inject the SWFI, directing the solution onto the inside wall of the vial to minimize foaming.
To reduce the total reconstitution time needed during preparation, reconstitute all required vials in succession.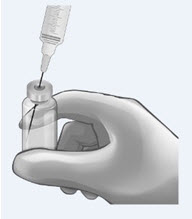
To ensure dissolution of the cake or powder, gently swirl each vial until complete dissolution of powder occurs (A). Do not shake (B); shaking could lead to foaming. Typical dissolution time for each vial is approximately three to five minutes. If dissolution is incomplete, discard the vial, and do not use the product.
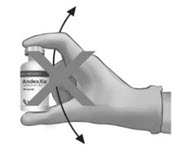
Upon reconstitution, the parenteral drug product should be inspected visually for particulate matter and discoloration prior to administration.(A) (B) 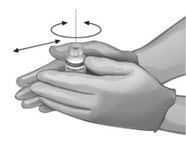
Use 60-mL (or larger) syringe with a 20-gauge (or higher) needle to withdraw the reconstituted ANDEXXA solution from each of the vials until the required dosing volume is achieved. Note the total volume withdrawn into the syringe.
Transfer the ANDEXXA solution from the syringe into an empty polyolefin or polyvinyl chloride IV bag with a volume of 250 mL or less.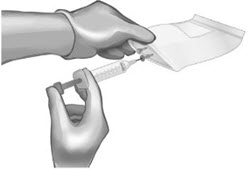
Discard the syringe and needle.
Discard the vials, including any unused portion.
Continuous IV Infusion Preparation
- Follow the same procedure outlined above for IV bolus preparation. Reconstitute the total number of vials needed based on the dose requirements. More than one 40 to 60-mL syringe, or an equivalent 100-mL syringe, may be used for transfer of reconstituted solution to the IV bag.
- Infusion will require a 0.2 or 0.22 micron in-line polyethersulfone or equivalent low protein-binding filter.
2.3 Administration
- Upon reconstitution, the parenteral drug product should be inspected visually for particulate matter and discoloration prior to administration.
- Administer ANDEXXA intravenously, using a 0.2 or 0.22 micron in-line polyethersulfone or equivalent low protein-binding filter.
- Start the bolus at a target rate of approximately 30 mg/min.
- Within two minutes following the bolus dose, administer the continuous IV infusion for up to 120 minutes.
2.4 Restarting Anticoagulant Therapy
Patients treated with FXa inhibitor therapy have underlying disease states that predispose them to thromboembolic events. Reversing FXa inhibitor therapy exposes patients to the thrombotic risk of their underlying disease. To reduce the risk of thrombosis, resume anticoagulant therapy as soon as medically appropriate following treatment with ANDEXXA.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Thromboembolic and Ischemic Risks
The thromboembolic and ischemic risks were assessed in 352 bleeding subjects who received ANDEXXA. Of the 63 subjects who experienced a thrombotic event, the median time to first event was 7 days, and 21 subjects experienced the event within the first three days. A total of 63 (18%) experienced 88 thromboembolic or ischemic events. Of the 352 subjects who received ANDEXXA, 223 received at least one anticoagulation dose within 30 days after treatment. Of these 223, 18 subjects (8%) had a thrombotic event and/or ischemic event after resumption.
Monitor subjects treated with ANDEXXA for signs and symptoms of arterial and venous thromboembolic events, ischemic events, and cardiac arrest. To reduce thromboembolic risk, resume anticoagulant therapy as soon as medically appropriate following treatment with ANDEXXA.
The safety of ANDEXXA has not been evaluated in subjects who experienced thromboembolic events or disseminated intravascular coagulation within two weeks prior to the life-threatening bleeding event requiring treatment with ANDEXXA. Safety of ANDEXXA also has not been evaluated in subjects who received prothrombin complex concentrates, recombinant factor VIIa, or whole blood products within seven days prior to the bleeding event.
5.2 Re-elevation or Incomplete Reversal of Anti-FXa Activity
The time course of anti-FXa activity following ANDEXXA administration was consistent among the healthy volunteer studies and the ANNEXA-4 study in bleeding subjects [see Clinical Studies (14)]. Compared to baseline, there was a rapid and substantial decrease in anti-FXa activity corresponding to the ANDEXXA bolus. This decrease was sustained through the end of the ANDEXXA continuous infusion. The anti-FXa activity returned to the placebo levels approximately two hours after completion of a bolus or continuous infusion. Subsequently, the anti-FXa activity decreased at a rate similar to the clearance of the FXa inhibitors.
Seventy-one subjects were anticoagulated with apixaban and had baseline levels of anti-FXa activity > 150 ng/mL. Nineteen subjects who were anticoagulated with rivaroxaban had elevated baseline anti-FXa activity levels >300 ng/mL. Forty-eight of the 71 apixaban-treated subjects (68%) experienced a > 90% decrease from baseline anti-FXa activity after administration of ANDEXXA. Ten of the 19 rivaroxaban subjects (53%) experienced a > 90% decrease from baseline anti-FXa activity after administration of ANDEXXA.
-
6 ADVERSE REACTIONS
The most common adverse reactions (≥ 5%) in bleeding subjects receiving ANDEXXA were urinary tract infections and pneumonia.
The most common adverse reactions (≥ 3%) in healthy subjects treated with ANDEXXA were infusion-related reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared directly to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In the pooled safety analysis of clinical trials of ANDEXXA, 223 healthy volunteers received FXa inhibitors, followed by treatment with ANDEXXA. The frequency of adverse reactions was similar in the ANDEXXA-treated group (120/223; 54%) and in the placebo-treated group (54/94; 57%). Infusion-related adverse reactions occurred in 18% (39/223) of the ANDEXXA-treated group and were the only type of adverse reaction that occurred more frequently than in the placebo group. No serious or severe adverse reactions were reported.
The ANNEXA-4 study is an ongoing multinational, prospective, open-label study using ANDEXXA in subjects presenting with acute major bleeding and who have recently received an FXa inhibitor. To date, safety data are available for 352 subjects. Sixty-three percent of the 352 subjects were 75 years or older. Subjects had received either apixaban (194/352; 55%) or rivaroxaban (128/352; 36%) as anticoagulation treatment for atrial fibrillation (286/352; 81%) or venous thromboembolism (87/352; 25%). In the majority of subjects, ANDEXXA was used to reverse anticoagulant therapy following either an intracranial hemorrhage (227; 64%) or a gastrointestinal bleed (90; 26%), with the remaining 35 subjects (10%) experiencing bleeding at other sites. Subjects were assessed at a Day 30 follow-up visit following infusion with ANDEXXA.
Deaths
In the ongoing ANNEXA-4 study, of the 352 subjects completing 30-day safety follow-up, there were 54 deaths (15%) occurring prior to the Day 30 visit. The number of cardiovascular deaths, including three with unknown causes and two that were unadjudicated, was 42 of 352 (12%), and the number of non-cardiovascular deaths was 12 (3%). Twenty (37%) subjects died within ten days after the ANDEXXA infusion. All subjects died prior to Day 45. Of the 54 subjects who died, the bleeding type was intracranial bleeding in 37 (69%), gastrointestinal bleeding in 12 (22%), and other bleeding types in 5 (9%) subjects.
Thromboembolic and Ischemic Events
In the ANNEXA-4 study, 63/352 (18%) subjects experienced one or more of the following overall thromboembolic events: cerebrovascular accident (CVA) (16/63; 25%), deep venous thrombosis (16/63; 25%), acute myocardial infarction (10/63; 16%), pulmonary embolism (5/63; 8%), and transient ischemic attack (1/63; 2%). The median time to event was seven days. A total of 33% of subjects with thromboembolic events (21/63) experienced the thromboembolic event during the first three days. Of the 352 subjects who received ANDEXXA, 223 received at least one anticoagulation dose within 30 days after treatment. Of these 223, 18 subjects (8%) had a thrombotic event and/or ischemic event after resumption [see Warnings and Precautions (5.1)].
No thromboembolic events were observed in 223 healthy volunteers who received FXa inhibitors and were treated with ANDEXXA.
Infusion-Related Reactions
Infusion-related reactions occurred in 18% (39/223) of ANDEXXA-treated healthy volunteers vs. 6% (6/94) of placebo-treated subjects. These reactions were characterized by a range of symptoms, including flushing, feeling hot, cough, dysgeusia, and dyspnea. Symptoms were mild to moderate in severity, and 90% (35/39) did not require treatment. One subject with a history of hives prematurely discontinued ANDEXXA after developing mild hives. Two of 352 (0.6%) subjects in the ANNEXA-4 study experienced an infusion-related reaction.
6.2 Immunogenicity
As with all therapeutic proteins, there is the potential for immunogenicity. Using an electrochemiluminescence (ECL)-based assay, 145 ANDEXXA-treated healthy subjects were tested for antibodies to ANDEXXA as well as for antibodies cross-reacting with factor X (FX) and FXa. Low titers of anti-ANDEXXA antibodies were observed in 26/145 healthy subjects (17%); 6% (9/145) were first observed at Day 30, with 20 subjects (14%) still having titers at the last time point (Days 44 to 48). To date, the pattern of antibody response in subjects in the ongoing ANNEXA-4 study has been similar to that observed in healthy volunteers. Of the 236 subjects with available samples, 6.8% (16/236) had antibodies against ANDEXXA. None of these anti-ANDEXXA antibodies were neutralizing. No neutralizing antibodies cross-reacting with FX or FXa were detected in healthy subjects (0/145) or in bleeding subjects (0/209) to date.
Detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to ANDEXXA with the incidence of antibodies to other products may be misleading.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of ANDEXXA in pregnant women to inform patients of associated risks. Animal reproductive and developmental studies have not been conducted with ANDEXXA.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There is no information regarding the presence of ANDEXXA in human milk, the effects on the breastfed child, or the effects on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ANDEXXA and any potential adverse effects on the breastfed child from ANDEXXA or from the underlying maternal condition.
8.4 Pediatric Use
The safety and efficacy of ANDEXXA in the pediatric population have not been studied.
8.5 Geriatric Use
Of the 352 subjects in the ANNEXA-4 study of ANDEXXA, 314 were 65 years of age or older, and 231 were 75 years of age or older. No overall differences in safety or efficacy were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between elderly and younger subjects; however, greater sensitivity of some older individuals cannot be ruled out.
The pharmacokinetics of ANDEXXA in healthy older (≥ 65 years; n=10) subjects were not different compared to younger (18-45 years; n=10) subjects.
-
11 DESCRIPTION
ANDEXXA (coagulation factor Xa (recombinant), inactivated-zhzo) is a sterile, white to off-white lyophilized powder available in single-use vials.
Each 200 mg vial delivers 200 mg of coagulation factor Xa formulated with the inactive ingredients tromethamine (Tris base), Tris hydrochloride, L-arginine hydrochloride, sucrose (1% w/v), mannitol (2.5% w/v), and polysorbate 80 (0.01% w/v) at pH 7.8.
After reconstitution of the lyophilized powder with SWFI for IV administration, the product is a clear, colorless to slightly yellow solution. ANDEXXA contains no preservatives.
The active ingredient in ANDEXXA is a genetically modified variant of human FXa. The active site serine was substituted with alanine, rendering the molecule unable to cleave and activate prothrombin. The gamma-carboxyglutamic acid (Gla) domain was removed to eliminate the protein's ability to assemble into the prothrombinase complex, thus removing the potential anticoagulant effects.
No additives of human or animal origin are used in the manufacture of ANDEXXA. The recombinant protein is produced in a genetically engineered Chinese Hamster Ovary (CHO) cell expression system and has a molecular weight of approximately 41 kDa. The manufacturing process incorporates two validated virus clearance steps.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Coagulation factor Xa (recombinant), inactivated-zhzo exerts its procoagulant effect by binding and sequestering the FXa inhibitors, rivaroxaban and apixaban. Another observed procoagulant effect of the ANDEXXA protein is its ability to bind to, and inhibit the activity of, Tissue Factor Pathway Inhibitor (TFPI). Inhibition of TFPI activity can increase tissue factor (TF)-initiated thrombin generation.
12.2 Pharmacodynamics
The effects of ANDEXXA can be measured using assays for its anti-FXa activity, free fraction of FXa inhibitor, and thrombin generation. In addition to its ability to sequester the FXa inhibitors, rivaroxaban and apixaban, ANDEXXA has been shown to inhibit TFPI activity.
The dose and dosing regimen of ANDEXXA that are required to reverse anti-FXa activity and to restore thrombin generation were determined in dose-ranging studies on healthy volunteers. Dosing of ANDEXXA, as a bolus followed by a two-hour continuous infusion, resulted in a rapid decrease in anti-FXa activity (within two minutes after the completion of the bolus administration) followed by reduced anti-FXa activity that was maintained throughout the duration of the continuous infusion [see Clinical Studies (14)]. The anti-FXa activity returned to the placebo levels approximately two hours after completion of a bolus or continuous infusion, whereas TFPI activity in plasma returned to the pretreatment levels approximately 96 hours following ANDEXXA administration.
Elevation of TF-initiated thrombin generation above the baseline range (prior to anticoagulation) occurred within two minutes following a bolus administration of ANDEXXA and was maintained throughout the duration of the continuous infusion. The TF-initiated thrombin generation was elevated above placebo for at least 22 hours. The sustained elevation of thrombin generation over the baseline range and the sustained elevation over placebo were not observed in a contact-activated thrombin generation assay (an assay that is not affected by TF-TFPI interaction).
Laboratory assessment of coagulation does not necessarily correlate with or predict the hemostatic effectiveness of ANDEXXA.
12.3 Pharmacokinetics
A summary of the pharmacokinetic (PK) properties of ANDEXXA in healthy subjects is shown in the table below (see Table 3).
Table 3: Summary of PK Parameters with High and Low Doses Low Dose High Dose n 11 10 From Table 14.2.1.2A of Clinical Study Report 16-512.
Data presented are geometric mean (Geometric Mean % Coefficient of Variation), [range].AUC0-∞ (hr*µg/mL) 200.5 (16.3)
[153.4; 255.6]572.9 (16.0)
[467.1; 783.9]Cmax (µg/mL) 76.6 (17.5)
[61.1; 100.1]206.6 (18.8)
[158.9; 280.5]Clearance (L/hr) 4.4 (16.3)
[3.4; 5.7]3.1 (16.0)
[2.3; 3.8]T1/2 (hr) 3.3 (15.0)
[2.3; 4.0]2.7 (20.0)
[1.9; 3.4]Vss (L) 4.4 (17.6)
[3.3; 5.7]3.0 (23.3)
[2.2; 5.0] - 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
The safety and efficacy of ANDEXXA were evaluated in two prospective, randomized, placebo-controlled studies, conducted in healthy volunteers (Study 1 ANNEXA-A; Study 2 ANNEXA-R). Both studies examined the percent change in anti-FXa activity, from baseline to nadir, for the low-dose and high-dose regimens of bolus followed by continuous infusion. Nadir is defined as the smallest value measured within five minutes after the end of the continuous infusion.
The safety and efficacy of ANDEXXA were evaluated in an ongoing, prospective, single-arm, open-label study (Study 3 ANNEXA-4) in subjects presenting with acute major bleeding and who have recently received an FXa inhibitor. This study examined the percent change in anti-FXa activity from baseline to the nadir between five minutes after the end of the bolus up until the end of the infusion and the rate of effective hemostasis within 12 hours after infusion, as rated by an independent endpoint adjudication committee.
Study 1 ANNEXA-A (NCT02207725) – apixaban reversal
In Study 1, healthy subjects (median age: 57 years; range: 50 to 73 years) received apixaban 5 mg twice daily for three and a half days to achieve steady-state. At three hours after the last apixaban dose (~ Cmax), ANDEXXA or placebo was administered. Eight subjects received placebo, and 24 received ANDEXXA, administered as a 400 mg IV bolus followed by a 4 mg per minute continuous infusion for 120 minutes (total 480 mg).
Study 2 ANNEXA-R (NCT02220725) – rivaroxaban reversal
In Study 2, healthy subjects (median age: 57 years; range: 50 to 68 years) received rivaroxaban 20 mg once per day for four days to achieve steady-state. At four hours after the last rivaroxaban dose (~ Cmax), ANDEXXA or placebo was administered. Thirteen subjects received placebo, and 26 received ANDEXXA, administered as an 800 mg IV bolus followed by an 8 mg per minute continuous infusion for 120 minutes (total 960 mg).
Reduction in Anti-FXa Activity
In Study 1 and Study 2, the percent change from baseline in anti-FXa activity at its nadir was statistically significant (p < 0.0001) in favor of the ANDEXXA groups compared to placebo in both Studies 1 and 2. The results of Study 1 and Study 2 are provided below (see Table 4).
The time courses of anti-FXa activity before and after ANDEXXA administration are shown in Figure 1.
Table 4 - A: Change in Anti-FXa Activity/Study 1 (apixaban) Anti-FXa Activity ANDEXXA
n=23Placebo
n=8Mean baseline ng/mL (± SD) 173.0
(50.5)191.7
(34.4)Mean ng/mL (± SD) change from baseline at the nadir* -160.6
(49.3)-63.2
(18.1)Mean % (± SD) change from baseline at the nadir* -92.3
(2.8)-32.7
(5.6)95% confidence interval (CI)† -59.5 (-64.1; -55.2) not applicable p-value < 0.0001‡ not applicable Table 4 - B: Change in Anti-FXa Activity/Study 2 (rivaroxaban) Anti-FXa Activity ANDEXXA
n=26Placebo
n=13SD = Standard deviation.
Note: Baseline is the last assessment obtained prior to the first dose of ANDEXXA or placebo.- * Nadir is the smallest value for anti-FXa activity at the 110-minute (ten minutes prior to the end of the infusion) time point, 2-minute time point before completion of the infusion, or the 5-minute time point after the completion of the infusion for each subject.
- † The CI is for the Hodges-Lehman estimate of shift.
- ‡ p-value obtained from a 2-sided exact Wilcoxon rank-sum test.
Mean baseline ng/mL (± SD) 335.3
(91.0)317.2
(91.0)Mean ng/mL (± SD) change from baseline at the nadir* -324.5
(89.2)-14.4
(58.8)Mean % (± SD) change from baseline at the nadir* -96.7
(1.8)-44.6
(11.8)95% confidence interval (CI)† -51.9 (-58.0; -47.0) not applicable p-value < 0.0001‡ not applicable Figure 1: Change in Anti-FXa Activity (ng/mL) in Subjects Anticoagulated with Apixaban (A – Study 1) and Rivaroxaban (B – Study 2)
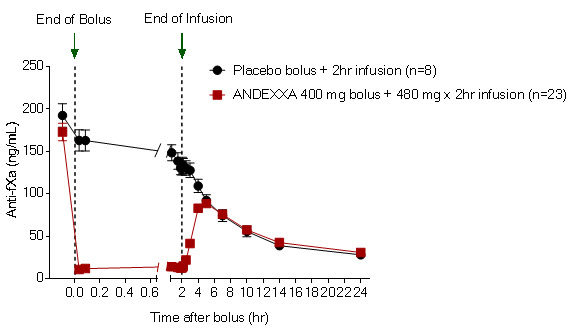
(A) 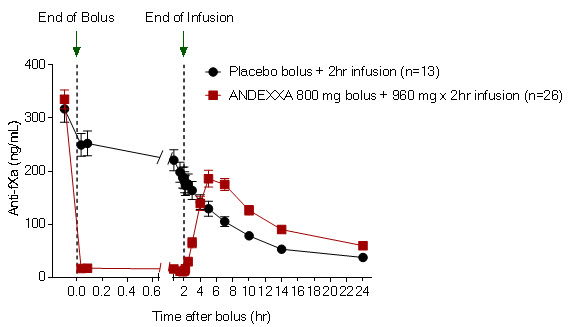
(B) Anti-FXa activity was measured prior to and after ANDEXXA or placebo administration.
Dashed lines indicate the end of the bolus or infusion. A break in the x-axis is added to better visualize the immediate, short-term dynamics of anti-FXa activity following ANDEXXA treatment. The points on the graph represent the mean anti-FXa activity level; error bars illustrate standard error. There was a statistically significant difference (p < 0.05) in the percent change of anti-FXa activity normalized to pre-bolus between ANDEXXA and placebo until two hours after administration of infusion.
A. Apixaban – with ANDEXXA 400 mg IV bolus plus 4 mg/min infusion for 120 minutes.
B. Rivaroxaban – with ANDEXXA 800 mg IV bolus plus 8 mg/min infusion for 120 minutes.Study 3 ANNEXA-4 (NCT02329327)
In an ongoing multinational, prospective, single-arm, open-label study, ANDEXXA was administered to 352 subjects taking FXa inhibitors who presented with acute major bleeding. The co-primary endpoints are: (a) percent change in anti-FXa activity from baseline to the nadir between five minutes after the end of the bolus up until the end of the infusion; and (b) rate of effective hemostasis within 12 hours after infusion, as rated by an independent endpoint adjudication committee.
Interim results of the study include data for 352 subjects dosed with ANDEXXA, of which 234 were efficacy-evaluable defined as subjects (1) on treatment with apixaban or rivaroxaban; (2) who had a baseline anti-FXa activity above 75 ng/mL; and (3) were adjudicated as meeting eligibility criteria for acute major bleeding [also see Adverse Reactions (6)].
For anti-FXa activity, the median decrease from baseline to nadir in anti-FXa activity for apixaban was -93% with a 95% CI of (-94%; -92%) and for rivaroxaban was -93% with a 95% CI of (-94%; -90%).
An improvement in hemostasis has not been established. ANDEXXA has not been shown to be effective for bleeding related to any FXa inhibitors other than apixaban or rivaroxaban.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
ANDEXXA (coagulation factor Xa (recombinant), inactivated-zhzo) is a white to off-white lyophilized cake or powder supplied as 4 single-use vials in a carton. ANDEXXA is not made with natural rubber latex.
ANDEXXA vials are provided as follows (see Table 6):
Table 6: Presentation of ANDEXXA NDC Carton Configuration Vial Cap Color Packaging Color NDC: 69853-0102-1 4 single use vials in a carton, each vial containing 200 mg of ANDEXXA Vials have a red flip-off cap. - Carton and vial label have a red to blue transition colored stripe across the front.
- Carton and label have "200 mg/vial" in a blue graphic on the front panel.
-
17 PATIENT COUNSELING INFORMATION
Inform patients that reversing FXa inhibitor therapy increases the risk of thromboembolic events. Arterial and venous thromboembolic events, ischemic events, cardiac events, and sudden death were observed within 30 days following ANDEXXA administration [see Warnings and Precautions (5.1)].
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton
NDC: 69853-0101-1
ANDEXXA®
coagulation factor Xa
(recombinant), inactivated-zhzoLyophilized powder for solution. For intravenous use only.
100
mg/vialFour 100 mg single-dose vials. Contains no preservative.
Rx Only

-
PRINCIPAL DISPLAY PANEL - 200 mg Vial Carton
NDC: 69853-0102-1
ANDEXXA®
coagulation factor Xa
(recombinant), inactivated-zhzoLyophilized powder for solution. For intravenous use only.
200
mg/vialFour 200 mg single-dose vials. Contains no preservative.
Rx Only

-
INGREDIENTS AND APPEARANCE
ANDEXXA
andexanet alfa injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69853-0101 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength andexanet alfa (UNII: BI009E452R) (andexanet alfa - UNII:BI009E452R) andexanet alfa 100 mg in 10 mL Inactive Ingredients Ingredient Name Strength tromethamine (UNII: 023C2WHX2V) ARGININE HYDROCHLORIDE (UNII: F7LTH1E20Y) sucrose (UNII: C151H8M554) mannitol (UNII: 3OWL53L36A) polysorbate 80 (UNII: 6OZP39ZG8H) hydrochloric acid (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69853-0101-1 4 in 1 CARTON 05/14/2018 10/31/2020 1 10 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125586 05/14/2018 10/31/2020 ANDEXXA
andexanet alfa injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69853-0102 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength andexanet alfa (UNII: BI009E452R) (andexanet alfa - UNII:BI009E452R) andexanet alfa 200 mg in 20 mL Inactive Ingredients Ingredient Name Strength tromethamine (UNII: 023C2WHX2V) ARGININE HYDROCHLORIDE (UNII: F7LTH1E20Y) sucrose (UNII: C151H8M554) mannitol (UNII: 3OWL53L36A) polysorbate 80 (UNII: 6OZP39ZG8H) TROMETHAMINE HYDROCHLORIDE (UNII: 383V75M34E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69853-0102-1 4 in 1 CARTON 01/08/2019 1 20 mL in 1 VIAL, SINGLE-USE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125586 01/08/2019 Labeler - Portola Pharmaceuticals (142296016) Establishment Name Address ID/FEI Business Operations Baxter Pharmaceutical Solutions, LLC 604719430 MANUFACTURE(69853-0101, 69853-0102) Establishment Name Address ID/FEI Business Operations Packaging Coordinators, Inc., AndersonBrecon, Inc. 053217022 LABEL(69853-0101, 69853-0102) , PACK(69853-0101, 69853-0102)
Trademark Results [ANDEXXA]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ANDEXXA 86776730 5251742 Live/Registered |
Portola Pharmaceuticals, Inc. 2015-10-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.