ATROPINE SULFATE solution
Atropine Sulfate by
Drug Labeling and Warnings
Atropine Sulfate by is a Prescription medication manufactured, distributed, or labeled by Bausch & Lomb Incorporated, Unither Manufacturing LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ATROPINE SULFATE OPHTHALMIC SOLUTION, USP 1% safely and effectively. See full prescribing information for ATROPINE SULFATE OPHTHALMIC SOLUTION, USP 1%.
ATROPINE SULFATE OPHTHALMIC SOLUTION, USP 1%, for topical ophthalmic use
Initial U.S. Approval: 1960INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- In individuals from three (3) months of age or greater, 1 drop topically to the cul-de-sac of the conjunctiva, forty minutes prior to the intended maximal dilation time (2.1).
- In individuals 3 years of age or greater, doses may be repeated up to twice daily as needed (2.2).
DOSAGE FORMS AND STRENGTHS
Ophthalmic solution: 1% atropine sulfate (10 mg/mL) ( 3).
CONTRAINDICATIONS
Hypersensitivity or allergic reaction to any ingredient in the formulation ( 4).
WARNINGS AND PRECAUTIONS
- Photophobia and blurred vision due to pupil unresponsiveness and cycloplegia may last up to 2 weeks ( 5.1).
- Risk of blood pressure increase from systemic absorption ( 5.2).
- To avoid the potential for eye injury or contamination, care should be taken to avoid touching the single-dose vial to the eye or to any other surface ( 5.3).
ADVERSE REACTIONS
Most common adverse reactions that have been reported are eye pain and stinging on administration, blurred vision, photophobia, superficial keratitis, decreased lacrimation, drowsiness, increased heart rate and blood pressure ( 6).
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
The use of atropine and monoamine oxidase inhibitors (MAOI) is generally not recommended because of the potential to precipitate hypertensive crisis ( 7).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Photophobia and Blurred Vision
5.2 Elevation of Blood Pressure
5.3 Potential for Eye Injury or Contamination
6 ADVERSE REACTIONS
6.1 Ocular Adverse Reactions
6.2 Systemic Adverse Reactions
7 DRUG INTERACTIONS
7.1 Monoamine Oxidase Inhibitors (MAOI)
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
In individuals from three (3) months of age or greater, 1 drop topically to the cul-de-sac of the conjunctiva in one or both eyes as indicated, forty minutes prior to the intended maximal dilation time.
In individuals 3 years of age or greater, doses may be repeated up to twice daily as needed.
Discard the single-dose vial immediately after use in one or both eyes.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Photophobia and Blurred Vision
Photophobia and blurred vision due to pupil unresponsiveness and cycloplegia may last up to 2 weeks.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in the labeling:
- Photophobia and Blurred Vision [see Warnings and Precautions (5.1)]
- Elevation in Blood Pressure [see Warnings and Precautions (5.2)]
The following adverse reactions have been identified following use of atropine sulfate ophthalmic solution. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Ocular Adverse Reactions
Eye pain and stinging occurs upon instillation of atropine sulfate ophthalmic solution. Other commonly occurring adverse reactions include blurred vision, photophobia, superficial keratitis and decreased lacrimation. Allergic reactions such as papillary conjunctivitis, contact dermatitis, and eyelid edema may also occur less commonly.
6.2 Systemic Adverse Reactions
Systemic effects of atropine are related to its anti-muscarinic activity. Systemic adverse events reported include dryness of skin, mouth, and throat from decreased secretions from mucous membranes; drowsiness, restlessness, irritability or delirium from stimulation of the central nervous system; tachycardia; flushed skin of the face and neck.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of Atropine Sulfate Ophthalmic Solution, USP 1% administration in pregnant women to inform a drug-associated risk. Adequate animal development and reproduction studies have not been conducted with atropine sulfate. In humans, 1% atropine sulfate is systemically bioavailable following topical ocular administration [see Clinical Pharmacology (12.3)] . Atropine Sulfate Ophthalmic Solution, USP 1% should only be used during pregnancy if the potential benefit justifies the potential risk to the fetus.
8.2 Lactation
Risk Summary
There is no information to inform risk regarding the presence of atropine in human milk following ocular administrations of Atropine Sulfate Ophthalmic Solution, USP 1% to the mother. The effects on breastfed infants and the effects on milk production are also unknown. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Atropine Sulfate Ophthalmic Solution, USP 1% and any potential adverse effects on the breastfed child from Atropine Sulfate Ophthalmic Solution, USP 1%.
8.4 Pediatric Use
Due to the potential for systemic absorption, the use of Atropine Sulfate Ophthalmic Solution, USP 1% in children under the age of 3 months is not recommended and the use in children under 3 years of age should be limited to no more than one drop per eye per day. Safety and efficacy in children above the age of 3 months has been established in adequate and well controlled trials.
-
10 OVERDOSAGE
In the event of accidental ingestion or toxic overdosage with Atropine Sulfate Ophthalmic Solution, USP 1%, supportive care may include a short acting barbiturate or diazepam as needed to control marked excitement and convulsions. Large doses for sedation should be avoided because central depressant action may coincide with the depression occurring late in atropine poisoning. Central stimulants are not recommended.
Physostigmine, given by slow intravenous injection of 1 to 4 mg (0.5 to 1 mg in pediatric populations), rapidly abolishes delirium and coma caused by large doses of atropine. Since physostigmine is rapidly destroyed, the patient may again lapse into coma after one to two hours, and repeated doses may be required.
Artificial respiration with oxygen may be necessary. Cooling measures may be needed to help to reduce fever, especially in pediatric populations.
The fatal adult dose of atropine is not known. In pediatric populations, 10 mg or less may be fatal.
-
11 DESCRIPTION
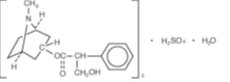
Atropine Sulfate Ophthalmic Solution, USP 1% is an aseptically prepared, sterile solution for topical ophthalmic use. The product does not contain an antimicrobial preservative. The active ingredient is represented by the chemical structure:
Chemical Name: Benzeneacetic acid, α-(hydroxymethyl)-, 8-methyl-8-azabicyclo[3.2.1.]oct-3-yl ester, endo –(±)-, sulfate (2:1) (salt), monohydrate.
Molecular Formula: (C 17H 23NO 3) 2 H 2SO 4 H 2O
Molecular Weight: 694.84 g/mol
Each mL of Atropine Sulfate Ophthalmic Solution, USP 1% contains: Active:atropine sulfate 10 mg equivalent to 8.3 mg of atropine.
Inactives:boric acid, hydroxypropyl methylcellulose, hydrochloric acid and/or sodium hydroxide may be added to adjust pH (3.5 to 6.0), and water for injection USP.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Atropine is a reversible antagonist of muscarine-like actions of acetylcholine and is therefore classified as an anti-muscarinic agent. Atropine is relatively selective for muscarinic receptors. Its potency at nicotinic receptors is much lower, and actions at non-muscarinic receptors are generally undetectable clinically. Atropine does not distinguish among the M1, M2, and M3 subgroups of muscarinic receptors.
The pupillary constrictor muscle depends on muscarinic cholinoceptor activation. This activation is blocked by topical atropine resulting in unopposed sympathetic dilator activity and mydriasis. Atropine also weakens the contraction of the ciliary muscle, or cycloplegia. Cycloplegia results in loss of the ability to accommodate such that the eye cannot focus for near vision.
12.2 Pharmacodynamics
The onset of action after administration of Atropine Sulfate Ophthalmic Solution, USP 1% generally occurs within minutes with maximal effect seen in hours and the effect can last multiple days [see Clinical Studies (14)] .
12.3 Pharmacokinetics
In a study of healthy subjects, after topical ocular administration of 30 μL of atropine sulfate ophthalmic solution 1%, the mean (± SD) systemic bioavailability of l-hyoscyamine was reported to be approximately 64 ± 29% (range 19% to 95%) as compared to intravenous administration of atropine sulfate. The median (range) time to maximum plasma concentration (Tmax) was 19 minutes (range 3 to 60 minutes), and the mean (±SD) peak plasma concentration (Cmax) of l-hyoscyamine was 288 ± 73 pg/mL. The mean (±SD) plasma half-life was reported to be approximately 2.5 ± 0.8 hours.
In a separate study of patients undergoing ocular surgery, after topical ocular administration of 40 μL of atropine sulfate ophthalmic solution, 1%, the mean (± SD) plasma Cmax of l-hyoscyamine was 860 ± 402 pg/mL, which was observed within 8 minutes following administration.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
Topical administration of Atropine Sulfate Ophthalmic Solution, USP 1% results in mydriasis and/or cycloplegia with efficacy demonstrated in both adults and children. The maximum effect for mydriasis is achieved in about 30–40 minutes after administration, with recovery after approximately 7–10 days. The maximum effect for cycloplegia is achieved within 60–180 minutes after administration, with recovery after approximately 7–12 days.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Atropine Sulfate Ophthalmic Solution, USP 1% is supplied as an aseptically prepared, sterile solution for topical ophthalmic use supplied as a 0.4 mL fill in a translucent, low-density polyethylene, single-dose vial. One (1) strip of 5 single-dose vials is packaged into a foil pouch.
- NDC 24208-965-01 10 single-dose vials. 2 foil pouches each containing one strip of 5 single-dose vials.
Storage and Handling:
Store at 20°C to 25°C (68°F to 77°F).
Store single-dose vials in the foil pouches. Opened vials cannot be resealed and should be discarded immediately after use.
-
17 PATIENT COUNSELING INFORMATION
- Advise patients that drops will sting upon instillation that they may experience blurry vision and sensitivity to light and should protect their eyes in bright illumination during dilation. These effects may last up to a couple weeks.
- Advise patients to keep the single-dose vials in the foil pouches until ready to use. The solution from one single-dose vial is to be used immediately after opening to dose one or both eye(s) of a single patient. The single-dose vial, including any remaining contents, should be discarded immediately after administration [see Dosage and Administration (2)] .
- Advise patients not to touch the tip of the single-dose vial to their eye or to any surface, in order to avoid eye injury or contamination of the solution.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA© 2022 Bausch & Lomb Incorporated or its affiliates
9775700
- Package/Label Display Panel
-
INGREDIENTS AND APPEARANCE
ATROPINE SULFATE
atropine sulfate solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 24208-965 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ATROPINE SULFATE (UNII: 03J5ZE7KA5) (ATROPINE - UNII:7C0697DR9I) ATROPINE SULFATE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 24208-965-01 10 in 1 CARTON 07/13/2022 1 0.4 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA213581 07/13/2022 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Unither Manufacturing LLC 079176615 manufacture(24208-965)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.