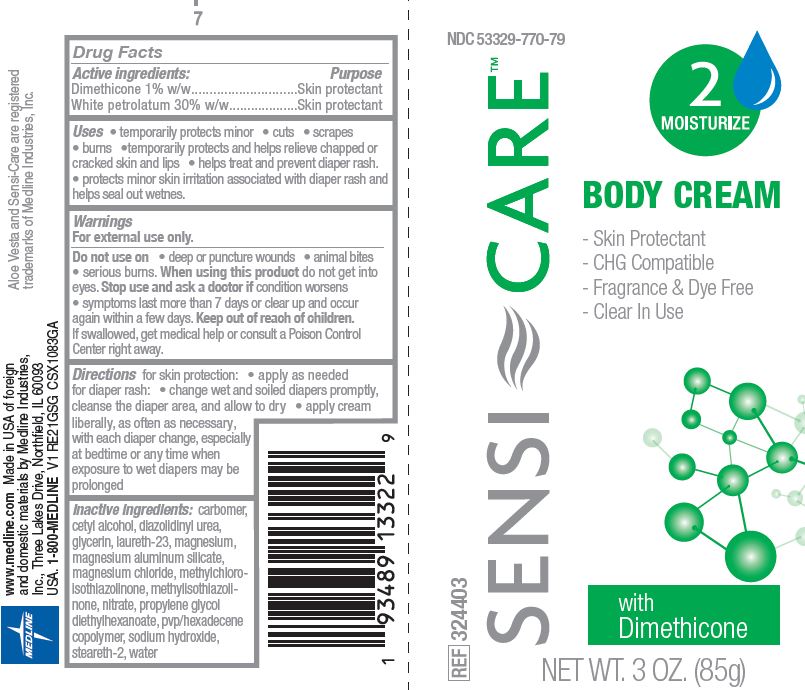
770 Sensi-Care Body Cream
Sensi-Care by
Drug Labeling and Warnings
Sensi-Care by is a Otc medication manufactured, distributed, or labeled by Medline Industries, LP. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SENSI-CARE- petrolatum, dimethicone cream
Medline Industries, LP
----------
770 Sensi-Care Body Cream
Uses
- temporarily protects minor cuts, scrapes, burns
- temporarily protects and helps relieve chapped or cracked skin and lips
- helps treat and prevent diaper rash
- protects minor skin irritation associated with diaper rash and helps seal out wetness
Warnings
For external use only.
Directions
for skin protection:
- apply as needed
for diaper rash:
- change wet and soiled diapers promptly, cleanse the diaper area, and allow to dry
- apply cream liberally, as often as necessary, with each diaper change, especially at bedtime or any time when exposure to wet diaper may be prolonged
Inactive ingredients
carbomer, cetyl alcohol, diazolidinyl urea, glycerin, laureth-23, magnesium, magnesium aluminum silicate, magnesium chloride, methylchloroisothiazolinone, methylisothiazolinone, nitrate, propylene glycol diethylhexanoate, pvp/hexadecene copolymer, sodium hydroxide, steareth-2, water
| SENSI-CARE
petrolatum, dimethicone cream |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Medline Industries, LP (025460908) |
| Registrant - Medline Industries, LP (025460908) |
Revised: 7/2024
Document Id: 1c480c5b-381f-6e5e-e063-6394a90a6fc1
Set id: af5fbed1-b639-66d4-e053-2a95a90a3026
Version: 5
Effective Time: 20240702
Trademark Results [Sensi-Care]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 SENSI-CARE 98606858 not registered Live/Pending |
Keyflow (UK) Ltd 2024-06-18 |
 SENSI-CARE 86803611 5448912 Live/Registered |
ConvaTec Inc. 2015-10-29 |
 SENSI-CARE 86123818 4672858 Live/Registered |
The Procter & Gamble Company 2013-11-20 |
 SENSI-CARE 75700756 2618533 Live/Registered |
CONVATEC INC. 1999-05-07 |
 SENSI-CARE 75700755 not registered Dead/Abandoned |
E.R. SQUIBB & SONS, INC. 1999-05-07 |
 SENSI-CARE 75371562 2322222 Live/Registered |
ConvaTec Inc. 1997-10-10 |
 SENSI-CARE 73551504 1387137 Dead/Cancelled |
SOUTHERN PLAINS MEDICAL CENTER, INC. 1985-08-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.