Colgate Anticavity Toothpaste
Colgate Anticavity by
Drug Labeling and Warnings
Colgate Anticavity by is a Otc medication manufactured, distributed, or labeled by Luoding Quality Amenities Supply Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
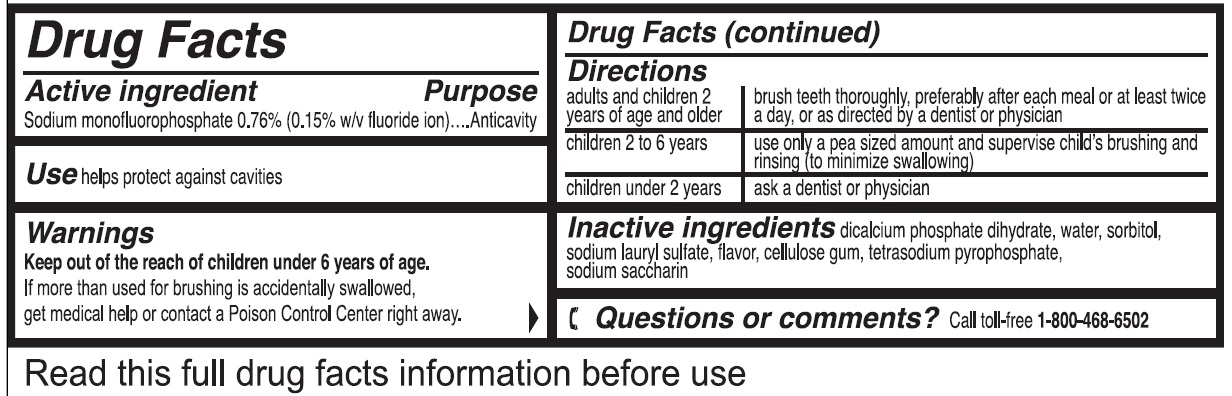
COLGATE ANTICAVITY- sodium monofluorophosphate paste
Luoding Quality Amenities Supply Limited
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Colgate Anticavity Toothpaste
Directions
| adults and children 2 years of age and older | brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or physician |
| children 2 to 6 years | use only a pea sized amount and supervise child's brushing and rinsing (to minimize swallowing) |
| children under 2 years | ask a dentist or physician |
| COLGATE ANTICAVITY
sodium monofluorophosphate paste |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Luoding Quality Amenities Supply Limited (528126807) |
Revised: 9/2023
Document Id: 0685f812-fa03-815a-e063-6294a90acbae
Set id: b0779880-b705-1197-e053-2995a90a2f45
Version: 2
Effective Time: 20230929
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.