79740-004 下架 Accel Hand Sanitizer
Accel Hand Sanitizer by
Drug Labeling and Warnings
Accel Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by ANTHOS GROUP, INC, THE. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
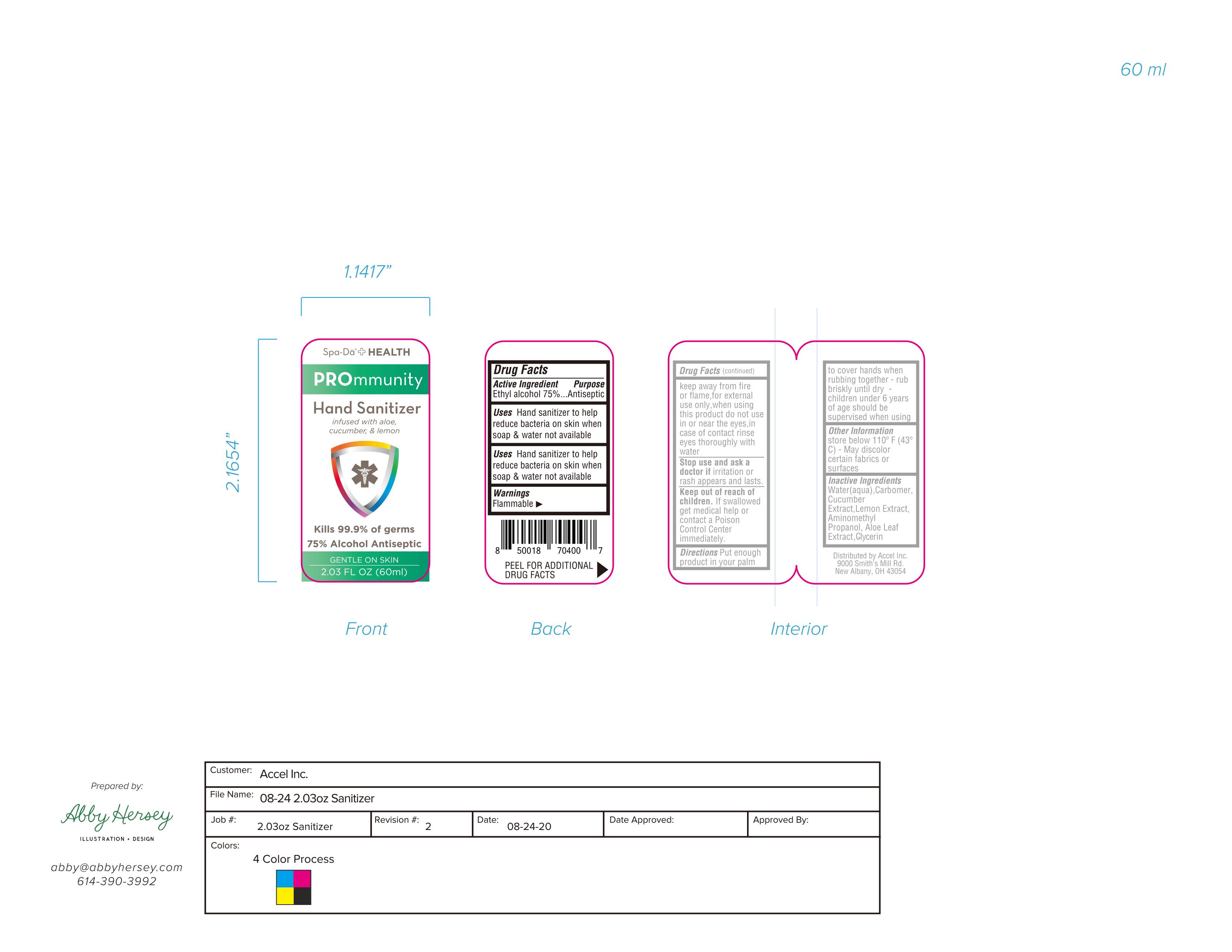
ACCEL HAND SANITIZER- hand sanitizer liquid
ANTHOS GROUP, INC, THE
----------
79740-004 下架
Accel Hand Sanitizer
Warnings
Flammable.keep away from fire or flame.for external use only.when using this product do not use in or near the eyes.in case of contact rinse eyes thoroughly with water
Stop use and ask a doctoir ii irritation or rash appears andi lasts.
Keep out of reach Orf children. If swallowed get medical help or contact a Poison Control Center immediatelly.
when using this product do not use in or near the eyes.in case of contact rinse eyes thoroughly with water
Stop use and ask a doctoir ii irritation or rash appears andi lasts.
Keep out of reach Orf children. If swallowed get medical help or contact a Poison Control Center immediatelly.
Keep out of reach Orf children. If swallowed get medical help or contact a Poison Control Center immediatelly.
Directions
Put enough product in your palm to cover hands when rubibing together rubbriskly until dry children under 6 years of age should be supervised when using
| ACCEL HAND SANITIZER
hand sanitizer liquid |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - ANTHOS GROUP, INC, THE (117511051) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ANTHOS GROUP, INC, THE | 117511051 | manufacture(79740-004) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.