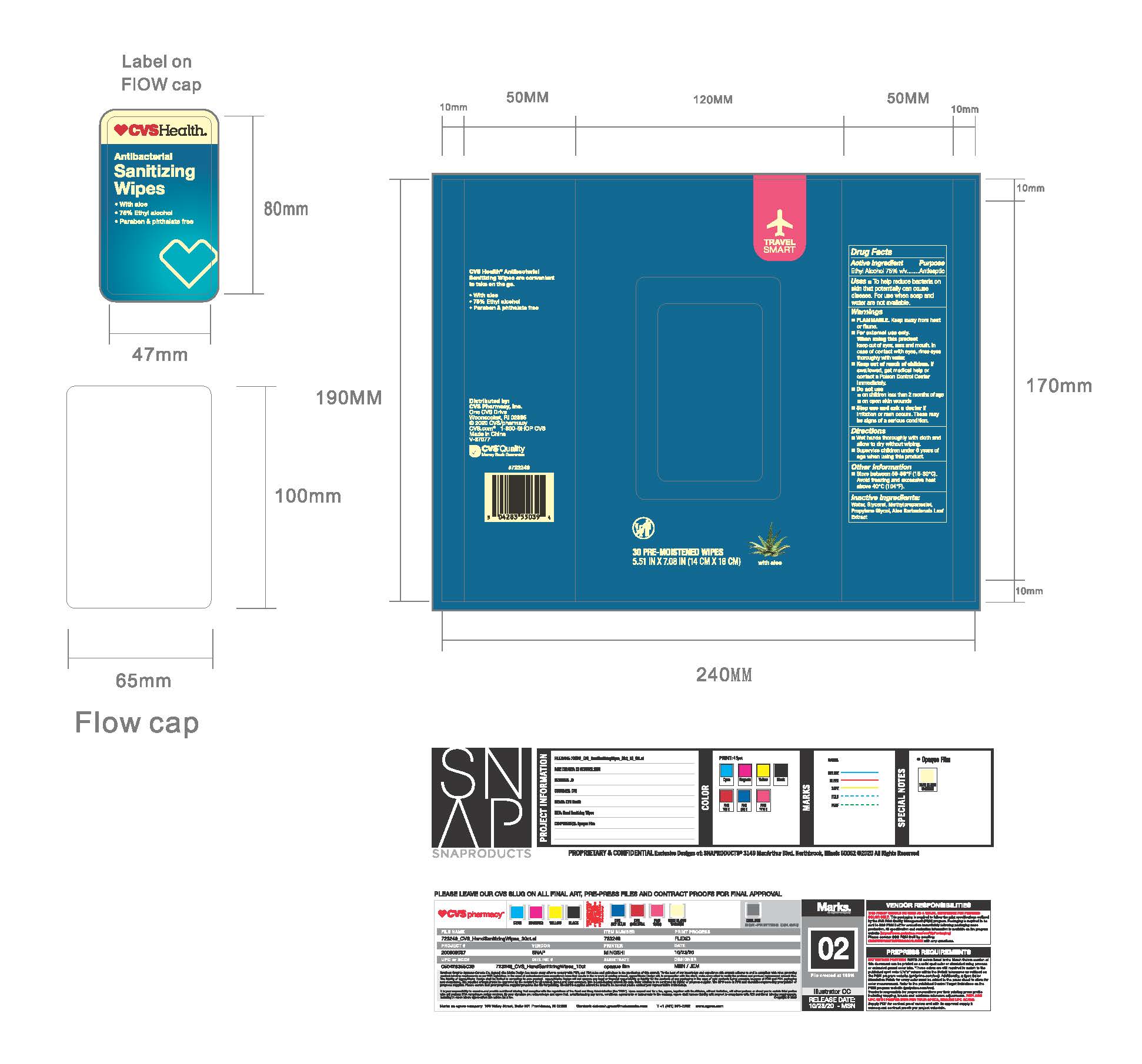
CVS Antibacterial Sanitizer Wipes
CVS Antibacterial Sanitizer Wipes by
Drug Labeling and Warnings
CVS Antibacterial Sanitizer Wipes by is a Otc medication manufactured, distributed, or labeled by SALES AND PRODUCT SOLUTIONS. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CVS ANTIBACTERIAL SANITIZER WIPES- ethanol 75% swab
SALES AND PRODUCT SOLUTIONS
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
CVS Antibacterial Sanitizer Wipes
When using this product
keep out of eyes, ears, and mouth, in case of contact with eyes, rinse eyes thoroughly with water
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately
Directions
Wet hands thoroughly with a cloth and a allow to dry without wiping.
Supervise children under 6 years of age when using this product.
| CVS ANTIBACTERIAL SANITIZER WIPES
ethanol 75% swab |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - SALES AND PRODUCT SOLUTIONS (037519852) |
| Registrant - SALES AND PRODUCT SOLUTIONS (037519852) |