CT SPF 20 by Meiyume CT Magic Lotion SPF20
CT SPF 20 by
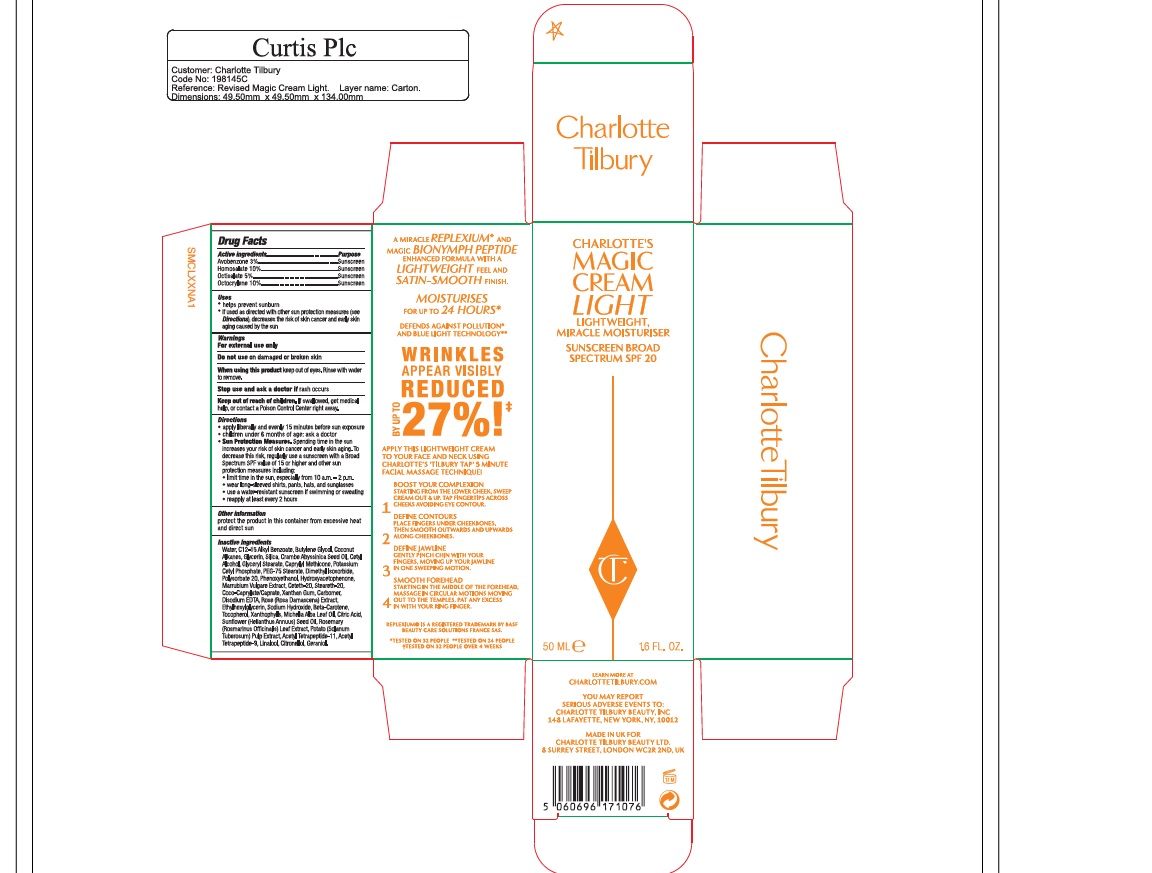
Drug Labeling and Warnings
CT SPF 20 by is a Otc medication manufactured, distributed, or labeled by Meiyume. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CT SPF 20- avobenzone lotion
Meiyume
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
CT Magic Lotion SPF20
helps prevent sunburn
if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If swallowed, get medical help, or contact a Poison Control Center right away.
apply liberally and evenly 15 minutes before sun exposure
children under 6 months of age: ask a doctor
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including
*limit time in the sun, especially from 10 a.m. - 2 p.m.
*wear long-sleeved shirts, pants, hats, and sunglasses
*use a water-resistant sunscreen if swimming or sweating
*reapply at least every 2 hours
Water, C12-15 Alkyl Benzoate Butylene Glycol, Coconut Alkanes, Glycerin, Silica, Crambe Abyssinica Seed Oil, Cetyl Alcohol, Glyceryl Stearate, Caprylyl Methicone, Potassium Cetyl Phosphate, PEG-75 Stearate, Dimethyl Isosorbide, Polysorbate 20, Phenoxyethanol, Hydroxyacetophenone, Marrubium Vulgare Extract, Ceteth-20, Steareth-20, Coco-Caprylate/Caprate, Xanthan Gum, Carbomer, Disodium EDTA, Rose (Rosa Damascera) Extract, Ethylhexylglycerin, Sodium Hydroxide, Beta-Carotene, Tocopherol, Xanthophylls, Michelia Alba Leaf Oil, Citric Acid, Sunflower (Helianthus Annuus) Seed Oil, Rosemary (Rosmarinus Officinalis) Leaf Extract, Potato (Solanum Tuberosum) Pulp Extract, Acetyl Tetrapeptide-11, Acetyl Tetrapeptide-9, Linalool, Citronellol, Geraniol
| CT SPF 20
avobenzone lotion |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Meiyume (225596238) |
| Registrant - Meiyume (225596238) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Meiyume | 225596238 | pack(51280-005) , manufacture(51280-005) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.