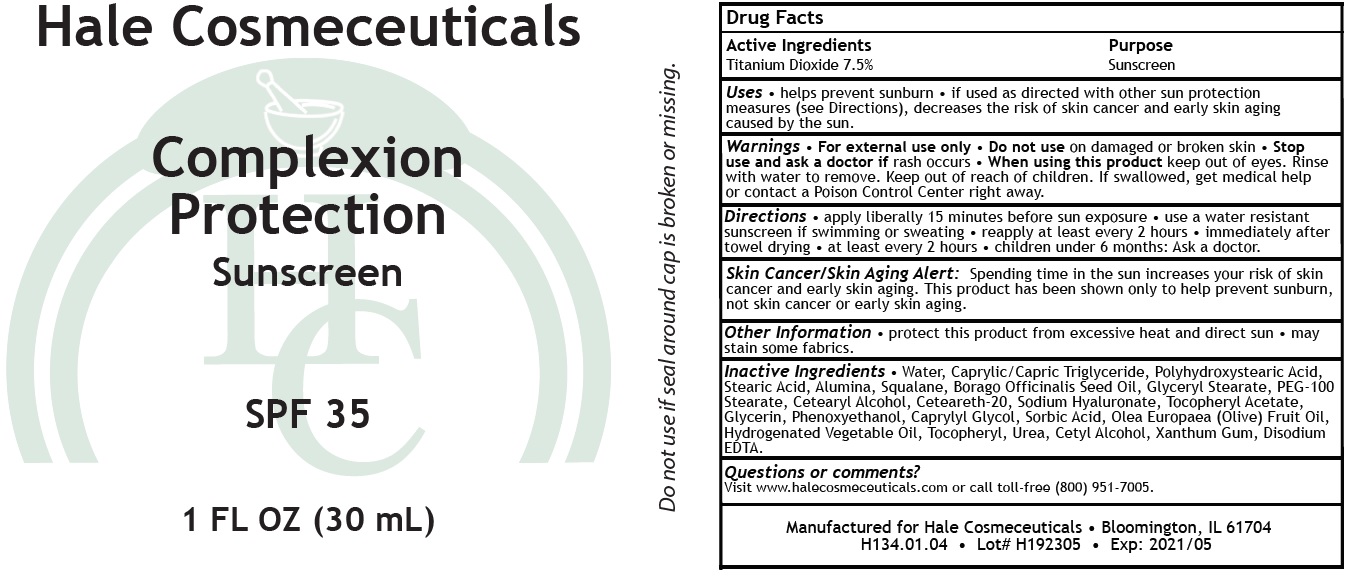
Hale Cosmeceuticals Complexion Protection Sunscreen SPF 35
Hale Cosmeceuticals Complexion Protection Sunscreen SPF 35 by
Drug Labeling and Warnings
Hale Cosmeceuticals Complexion Protection Sunscreen SPF 35 by is a Otc medication manufactured, distributed, or labeled by Hale Cosmeceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HALE COSMECEUTICALS COMPLEXION PROTECTION SUNSCREEN SPF 35- titanium dioxide cream
Hale Cosmeceuticals, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Hale Cosmeceuticals Complexion Protection Sunscreen SPF 35
Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- immediately after towel drying
- at least every 2 hours
- children under 6 months: Ask a doctor
Skin Cancer/Skin Aging Alert:Spending time in the sun increases your risk of skin cancer and early skin aging. This product has been shown only to help prevent sunburn, not skin cancer or early skin aging.
Inactive Ingredients
- Water, Caprylic/Capric Triglyceride, Polyhydroxystearic Acid, Stearic Acid, Alumina, Squalane, Borago Officinalis Seed Oil, Glyceryl Stearate, PEG-100 Stearate, Cetearyl Alcohol, Ceteareth-20, Sodium Hyaluronate, Tocopheryl Acetate, Glycerin, Phenoxyethanol, Caprylyl Glycol, Sorbic Acid, Olea Europaea (Olive) Fruit Oil, Hydrogenated Vegetable Oil, Tocopheryl, Urea, Cetyl Alcohol, Xanthum Gum, Disodium EDTA.
| HALE COSMECEUTICALS COMPLEXION PROTECTION SUNSCREEN SPF 35
titanium dioxide cream |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Hale Cosmeceuticals, Inc. (168405822) |
Revised: 12/2022
Document Id: efcf8feb-77cf-389d-e053-2a95a90a7232
Set id: b5a7c5f4-9219-2ea4-e053-2a95a90a1f97
Version: 3
Effective Time: 20221214