MORPHINE SULFATE- morphine sulfate injection, solution MORPHINE SULFATE injection, solution
Morphine Sulfate by
Drug Labeling and Warnings
Morphine Sulfate by is a Prescription medication manufactured, distributed, or labeled by Fresenius Kabi USA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MORPHINE SULFATE INJECTION safely and effectively. See full prescribing information for MORPHINE SULFATE INJECTION.
Morphine Sulfate injection, for intravenous or intramuscular use, CII
Initial U.S. Approval: 1941WARNING: ADDICTION, ABUSE, AND MISUSE; LIFE-THREATENING RESPIRATORY DEPRESSION; NEONATAL OPIOID WITHDRAWAL SYNDROME; and RISKS FROM CONCOMITANT USE WITH BENZODIAZEPINES OR OTHER CNS DEPRESSANTS
See full prescribing information for complete boxed warning.
- Morphine Sulfate Injection exposes users to risks of addiction, abuse, and misuse, which can lead to overdose and death. Assess patient's risk before prescribing and monitor regularly for these behaviors and conditions. ( 5.1)
- Serious, life-threatening, or fatal respiratory depression may occur. Monitor closely, especially upon initiation or following a dose increase. ( 5.2)
- Prolonged use of Morphine Sulfate Injection during pregnancy can result in neonatal opioid withdrawal syndrome, which may be life-threatening if not recognized and treated. If prolonged opioid use is required in a pregnant woman, advise the patient of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available. ( 5.3)
- Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing for use in patients for whom alternative treatment options are inadequate; limit dosages and durations to the minimum required; and follow patients for signs and symptoms of respiratory depression and sedation. ( 5.4, 7)
RECENT MAJOR CHANGES
Boxed Warning
12/2016
Indications and Usage
12/2016
Dosage and Administration
12/2016
Contraindications
12/2016
Warnings and Precautions
12/2016
INDICATIONS AND USAGE
Morphine Sulfate Injection is an opioid agonist indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate. ( 1)
Limitations of Use ( 1)
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, reserve Morphine Sulfate Injection, for use in patients for whom alternative treatment options [e.g., non-opioid analgesics or opioid combination products]:
- Have not been tolerated, or are not expected to be tolerated,
- Have not provided adequate analgesia, or are not expected to provide adequate analgesia
DOSAGE AND ADMINISTRATION
- Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals. ( 2.1)
- Individualize dosing based on the severity of pain, patient response, prior analgesic experience, and risk factors for addiction, abuse, and misuse. ( 2.1)
- Direct Intravenous Injection: Initiate treatment with 0.1 mg to 0.2 mg per kg every 4 hours as needed to manage pain. ( 2.2)
- Intramuscular Injection: Initiate treatment with 10 mg, every 4 hours as needed to manage pain (based on a 70 kg adult). ( 2.2)
- Do not stop Morphine Sulfate Injection abruptly in a physically dependent patient. ( 2.4)
DOSAGE FORMS AND STRENGTHS
Injection, 2 mg/mL, 4 mg/mL, 5 mg/mL, 8 mg/mL, and 10 mg/mL in a pre-filled disposable syringe for intravenous or intramuscular use. ( 3)
CONTRAINDICATIONS
- Significant respiratory depression. ( 4)
- Acute or severe bronchial asthma in an unmonitored setting or in absence of resuscitative equipment. ( 4)
- Concurrent use of monoamine oxidase inhibitors (MAOIs) or use of MAOIs within the last 14 days ( 4)
- Known or suspected gastrointestinal obstruction, including paralytic ileus. ( 4)
- Hypersensitivity to morphine. ( 4)
WARNINGS AND PRECAUTIONS
- Cardiovascular Instability: High doses are excitatory. Have Naloxone Injection and resuscitative equipment immediately available. ( 5.5)
- Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients: Monitor closely, particularly during initiation and titration. ( 5.2)
- Adrenal Insufficiency: If diagnosed, treat with physiologic replacement of corticosteroids, and wean patient off of the opioid. ( 5.8)
- Severe Hypotension: Monitor during dosage initiation and titration. Avoid use of Morphine Sulfate Injection in patients with circulatory shock. ( 5.9)
- Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness: Monitor for sedation and respiratory depression. Avoid use of Morphine Sulfate Injection in patients with impaired consciousness or coma. ( 5.10)
ADVERSE REACTIONS
The most serious adverse reactions encountered are respiratory depression, apnea, circulatory depression, respiratory arrest, shock and cardiac arrest. Other common frequently observed adverse reactions include: sedation, lightheadedness, dizziness, nausea, vomiting, constipation and diaphoresis. ( 6)
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
DRUG INTERACTIONS
- Serotonergic Drugs: Concomitant use may result in serotonin syndrome. Discontinue Morphine Sulfate Injection if serotonin syndrome is suspected. ( 7)
- Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics: Avoid use with Morphine Sulfate Injection because they may reduce analgesic effect of Morphine Sulfate Injection or precipitate withdrawal symptoms. ( 7)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: ADDICTION, ABUSE, AND MISUSE; LIFE-THREATENING RESPIRATORY DEPRESSION; NEONATAL OPIOID WITHDRAWAL SYNDROME; and RISKS FROM CONCOMITANT USE WITH BENZODIAZEPINES OR OTHER CNS DEPRESSANTS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
2.2 Initial Dosage
2.3 Titration and Maintenance of Therapy
2.4 Discontinuation of Morphine Sulfate Injection
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Addiction, Abuse, and Misuse
5.2 Life-Threatening Respiratory Depression
5.3 Neonatal Opioid Withdrawal Syndrome
5.4 Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants
5.5 Cardiovascular Instability
5.6 Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients
5.7 Interactions with Monoamine Oxidase Inhibitors
5.8 Adrenal Insufficiency
5.9 Severe Hypotension
5.10 Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness
5.11 Risks of Use in Patients with Gastrointestinal Conditions
5.12 Increased Risk of Seizures in Patients with Seizure Disorders
5.13 Withdrawal
5.14 Central Nervous System Toxicity
5.15 Exposure, Hypothermia, Immersion and Shock
5.16 Risks of Driving and Operating Machinery
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: ADDICTION, ABUSE, AND MISUSE; LIFE-THREATENING RESPIRATORY DEPRESSION; NEONATAL OPIOID WITHDRAWAL SYNDROME; and RISKS FROM CONCOMITANT USE WITH BENZODIAZEPINES OR OTHER CNS DEPRESSANTS
Addiction, Abuse, and Misuse
Morphine Sulfate Injection exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death. Assess each patient's risk prior to prescribing Morphine Sulfate Injection, and monitor all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions ( 5.1)] .
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of Morphine Sulfate Injection. Monitor for respiratory depression, especially during initiation of Morphine Sulfate Injection, or following a dose increase. Because of delay in maximum CNS effect with intravenously administered morphine (30 min), rapid IV administration may result in overdosing[see Warnings and Precautions ( 5.2)].
Neonatal Opioid Withdrawal Syndrome
Prolonged use of Morphine Sulfate Injection during pregnancy can result in neonatal opioid withdrawal syndrome, which may be life-threatening if not recognized and treated, and requires management according to protocols developed by neonatology experts. If opioid use is required for a prolonged period in a pregnant woman, advise the patient of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available[see Warnings and Precautions ( 5.3)] .
Risks From Concomitant Use With Benzodiazepines Or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death[seeWarnings and Precautions ( 5.4), Drug Interactions ( 7)] .
- Reserve concomitant prescribing of Morphine Sulfate Injection and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
-
1 INDICATIONS AND USAGE
Morphine Sulfate Injection is indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate.
Limitations of Use
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses [see Warnings and Precautions ( 5.1)] , reserve Morphine Sulfate Injection for use in patients for whom alternative treatment options [e.g., non-opioid analgesics or opioid combination products]:
- Have not been tolerated, or are not expected to be tolerated,
- Have not provided adequate analgesia, or are not expected to provide adequate analgesia
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosage and Administration Instructions
Morphine Sulfate Injection is intended for intravenous and intramuscular administration.
Morphine Sulfate Injection is available in five concentrations for direct injection. Dosing errors can result in accidental overdose and death. Avoid dosing errors that may result from confusion between mg and mL and confusion with morphine injections of different concentrations when prescribing, dispensing, and administering Morphine Sulfate Injection. Ensure that the dose is communicated and dispensed accurately.
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions ( 5.1)].
Initiate the dosing regimen for each patient individually, taking into account the patient's severity of pain, patient response, prior analgesic treatment experience, and risk factors for addiction, abuse, and misuse [see Warnings and Precautions ( 5.1)] .
Monitor patients closely for respiratory depression, especially within the first 24-72 hours of initiating therapy and following dosage increases with Morphine Sulfate Injection and adjust the dosage accordingly [see Warnings and Precautions ( 5.2)].
Administration of Morphine Sulfate Injection should be limited to use by those familiar with the management of respiratory depression. Morphine must be injected slowly; rapid intravenous administration may result in chest wall rigidity.
Inspect Morphine Sulfate Injection for particulate matter and discoloration prior to administration.
2.2 Initial Dosage
2.3 Titration and Maintenance of Therapy
Individually titrate Morphine Sulfate Injection to a dose that provides adequate analgesia and minimizes adverse reactions. Continually reevaluate patients receiving Morphine Sulfate Injection to assess the maintenance of pain control and the relative incidence of adverse reactions, as well as monitoring for the development of addiction, abuse, or misuse [see Warnings and Precautions ( 5.1)] .
If unacceptable opioid-related adverse reactions are observed, consider reducing the dosage. Adjust the dosage to obtain an appropriate balance between management of pain and opioid-related adverse reactions.
2.4 Discontinuation of Morphine Sulfate Injection
When a patient who has been taking Morphine Sulfate Injection regularly and may be physically dependent or no longer requires therapy with Morphine Sulfate Injection, taper the dose gradually, by 25% to 50% every 2 to 4 days, while monitoring carefully for signs and symptoms of withdrawal. If the patient develops these signs or symptoms, raise the dose to the previous level and taper more slowly, either by increasing the interval between decreases, decreasing the amount of change in dose, or both. Do not abruptly discontinue Morphine Sulfate Injection in a physically dependent patient [see Warnings and Precautions ( 5.13), Drug Abuse and Dependence ( 9.3)] .
-
3 DOSAGE FORMS AND STRENGTHS
Morphine Sulfate Injection is available in the following strengths for intravenous (IV) and intramuscular (IM) administration.
- 2 mg/mL in 1 mL prefilled disposable syringe for IV or IM use.
- 4 mg/mL in 1 mL prefilled disposable syringe for IV or IM use.
- 5 mg/mL in 1 mL prefilled disposable syringe for IV or IM use.
- 8 mg/mL in 1 mL prefilled disposable syringe for IV or IM use.
- 10 mg/mL in 1 mL prefilled disposable syringe for IV or IM use.
-
4 CONTRAINDICATIONS
Morphine Sulfate Injection is contraindicated in patients with:
- Significant respiratory depression [see Warnings and Precautions ( 5.2)]
- Acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment [see Warnings and Precautions ( 5.6)]
- Concurrent use of monoamine oxidase inhibitors (MAOIs) or use of MAOIs within the last 14 days [see Warnings and Precautions ( 5.7)].
- Known or suspected gastrointestinal obstruction, including paralytic ileus [see Warnings and Precautions ( 5.11)]
- Hypersensitivity to morphine (e.g., anaphylaxis) [see Adverse Reactions ( 6)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Addiction, Abuse, and Misuse
Morphine Sulfate Injection contains morphine, a Schedule II controlled substance. As an opioid, Morphine Sulfate Injection exposes users to the risks of addiction, abuse, and misuse [see Drug Abuse and Dependence ( 9)] .
Although the risk of addiction in any individual is unknown, it can occur in patients appropriately prescribed Morphine Sulfate Injection. Addiction can occur at recommended dosages and if the drug is misused or abused.
Assess each patient's risk for opioid addiction, abuse, or misuse prior to prescribing Morphine Sulfate Injection, and monitor all patients receiving Morphine Sulfate Injection for the development of these behaviors and conditions. Risks are increased in patients with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). The potential for these risks should not, however, prevent the proper management of pain in any given patient. Patients at increased risk may be prescribed opioids such as Morphine Sulfate Injection, but use in such patients necessitates intensive counseling about the risks and proper use of Morphine Sulfate Injection, along with intensive monitoring for signs of addiction, abuse, and misuse.
Opioids are sought by drug abusers and people with addiction disorders and are subject to criminal diversion. Consider these risks when prescribing or dispensing Morphine Sulfate Injection. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity. Contact local state professional licensing board or state controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
5.2 Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death. Management of respiratory depression may include close observation, supportive measures, and use of opioid antagonists, depending on the patient's clinical status [see Overdosage ( 10)] . Carbon dioxide (CO 2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids.
While serious, life-threatening, or fatal respiratory depression can occur at any time during the use of Morphine Sulfate Injection, the risk is greatest during the initiation of therapy or following a dosage increase. Because of a delay in the maximum CNS effect with intravenously administered Morphine Sulfate Injection (30 min), rapid administration may result in overdosing. The respiratory depression may be severe and could require intervention [see OVERDOSAGE ( 10)] . Monitor patients closely for respiratory depression, especially within the first 24-72 hours of initiating therapy with and following dosage increases of Morphine Sulfate Injection.
To reduce the risk of respiratory depression, proper dosing and titration of Morphine Sulfate Injection, are essential [see Dosage and Administration ( 2.4)] . Overestimating the Morphine Sulfate Injection, dosage when converting patients from another opioid product can result in a fatal overdose with the first dose.
5.3 Neonatal Opioid Withdrawal Syndrome
Prolonged use of Morphine Sulfate Injection during pregnancy can result in withdrawal in the neonate. Neonatal opioid withdrawal syndrome, unlike opioid withdrawal syndrome in adults, may be life-threatening if not recognized and treated, and requires management according to protocols developed by neonatology experts. Observe newborns for signs of neonatal opioid withdrawal syndrome and manage accordingly. Advise pregnant women using opioids for a prolonged period of the risk of neonatal opioid withdrawal syndrome and ensure that appropriate treatment will be available [see Use in Specific Populations ( 8.1)] .
5.4 Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants
Profound sedation, respiratory depression, coma, and death may result from the concomitant use of Morphine Sulfate Injection with benzodiazepines or other CNS depressants (e.g., non-benzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol). Because of these risks, reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions ( 7)] .
If the decision is made to prescribe a benzodiazepine or other CNS depressant concomitantly with an opioid analgesic, prescribe the lowest effective dosages and minimum durations of concomitant use. In patients already receiving an opioid analgesic, prescribe a lower initial dose of the benzodiazepine or other CNS depressant than indicated in the absence of an opioid, and titrate based on clinical response. If an opioid analgesic is initiated in a patient already taking a benzodiazepine or other CNS depressant, prescribe a lower initial dose of the opioid analgesic, and titrate based on clinical response. Follow patients closely for signs and symptoms of respiratory depression and sedation.
Advise both patients and caregivers about the risks of respiratory depression and sedation when Morphine Sulfate Injection is used with benzodiazepines or other CNS depressants (including alcohol and illicit drugs). Advise patients not to drive or operate heavy machinery until the effects of concomitant use of the benzodiazepine or other CNS depressant have been determined. Screen patients for risk of substance use disorders, including opioid abuse and misuse, and warn them of the risk for overdose and death associated with the use of additional CNS depressants including alcohol and illicit drugs [see Drug Interactions ( 7)] .
5.5 Cardiovascular Instability
While low doses of intravenously administered morphine have little effect on cardiovascular stability, high doses are excitatory, resulting from sympathetic hyperactivity and increase in circulatory catecholamines. Have Naloxone Injection and resuscitative equipment immediately available for use in case of life-threatening or intolerable side effects and whenever morphine therapy is being initiated.
5.6 Life-Threatening Respiratory Depression in Patients with Chronic Pulmonary Disease or in Elderly, Cachectic, or Debilitated Patients
The use of Morphine Sulfate Injection in patients with acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment is contraindicated.
Patients with Chronic Pulmonary Disease: Morphine Sulfate Injection-treated patients with significant chronic obstructive pulmonary disease or cor pulmonale, and those with a substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression are at increased risk of decreased respiratory drive including apnea, even at recommended dosages of Morphine Sulfate Injection [see Warnings and Precautions ( 5.2)]
Elderly, Cachectic, or Debilitated Patients: Life-threatening respiratory depression is more likely to occur in elderly, cachectic, or debilitated patients because they may have altered pharmacokinetics or altered clearance compared to younger, healthier patients [see Warnings and Precautions ( 5.2)] .
Monitor such patients closely, particularly when initiating and titrating Morphine Sulfate Injection and when Morphine Sulfate Injection is given concomitantly with other drugs that depress respiration [see Warnings and Precautions ( 5.2 )] . Alternatively, consider the use of non-opioid analgesics in these patients.
5.7 Interactions with Monoamine Oxidase Inhibitors
Monoamine oxidase inhibitors (MAOIs) may potentiate the effects of morphine, including respiratory depression, coma, and confusion. Morphine Sulfate Injection should not be used in patients taking MAOIs or within 14 days of stopping such treatment.
5.8 Adrenal Insufficiency
Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use. Presentation of adrenal insufficiency may include non-specific symptoms and signs including nausea, vomiting, anorexia, fatigue, weakness, dizziness, and low blood pressure. If adrenal insufficiency is suspected, confirm the diagnosis with diagnostic testing as soon as possible. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off of the opioid to allow adrenal function to recover and continue corticosteroid treatment until adrenal function recovers. Other opioids may be tried as some cases reported use of a different opioid without recurrence of adrenal insufficiency. The information available does not identify any particular opioids as being more likely to be associated with adrenal insufficiency.
5.9 Severe Hypotension
Morphine Sulfate Injection may cause severe hypotension including orthostatic hypotension and syncope in ambulatory patients. There is increased risk in patients whose ability to maintain blood pressure has already been compromised by a reduced blood volume or concurrent administration of certain CNS depressant drugs (e.g., phenothiazines or general anesthetics) [see Drug Interactions ( 7)]. Monitor these patients for signs of hypotension after initiating or titrating the dosage of Morphine Sulfate Injection. In patients with circulatory shock, Morphine Sulfate Injection may cause vasodilation that can further reduce cardiac output and blood pressure. Avoid the use of Morphine Sulfate Injection in patients with circulatory shock.
5.10 Risks of Use in Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness
In patients who may be susceptible to the intracranial effects of CO 2 retention (e.g., those with evidence of increased intracranial pressure or brain tumors), Morphine Sulfate Injection may reduce respiratory drive, and the resultant CO 2 retention can further increase intracranial pressure. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with Morphine Sulfate Injection
Opioids may also obscure the clinical course in a patient with a head injury. Avoid the use of Morphine Sulfate Injection in patients with impaired consciousness or coma.
5.11 Risks of Use in Patients with Gastrointestinal Conditions
Morphine Sulfate Injection is contraindicated in patients with known or suspected gastrointestinal obstruction, including paralytic ileus.
The morphine in Morphine Sulfate Injection may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis for worsening symptoms.
5.12 Increased Risk of Seizures in Patients with Seizure Disorders
The morphine in Morphine Sulfate Injection, may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Monitor patients with a history of seizure disorders for worsened seizure control during Morphine Sulfate Injection therapy.
5.13 Withdrawal
Avoid the use of mixed agonist/antagonist (e.g, pentazocine, nalbuphine, and butorphanol) or partial agonist (e.g., buprenorphine) analgesics in patients who are receiving a full opioid agonist analgesic, including Morphine Sulfate Injection. In these patients, mixed agonist/antagonist and partial agonist analgesics may reduce the analgesic effect and/or precipitate withdrawal symptoms.
When discontinuing Morphine Sulfate Injection in a physically-dependent patient, gradually taper the dosage [see Dosage and Administration ( 2.4)] . Do not abruptly discontinue Morphine Sulfate Injection in these patients [see Drug Abuse and Dependence ( 9.3)] .
5.14 Central Nervous System Toxicity
Dysphoric reactions may occur after any size dose and toxic psychoses have been reported.
5.15 Exposure, Hypothermia, Immersion and Shock
Caution must be used when injecting any opioid intramuscularly into chilled areas or in patients with hypotension or shock, since impaired perfusion may prevent complete absorption; if repeated injections are administered, an excessive amount may be suddenly absorbed if normal circulation is re-established.
5.16 Risks of Driving and Operating Machinery
Morphine Sulfate Injection may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery. Warn patients not to drive or operate dangerous machinery unless they are tolerant to the effects of Morphine Sulfate Injection and know how they will react to the medication.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described, or described in greater detail, in other sections:
- Addiction, Abuse, and Misuse [see Warnings and Precautions ( 5.1)]
- Life-Threatening Respiratory Depression [see Warnings and Precautions ( 5.2)]
- Neonatal Opioid Withdrawal Syndrome [see Warnings and Precautions ( 5.3)]
- Interactions with Benzodiazepine or Other CNS Depressants [see Warnings and Precautions ( 5.4)]
- Cardiovascular Instability [see Warnings and Precautions ( 5.5)]
- Adrenal Insufficiency [see Warnings and Precautions ( 5.8)]
- Severe Hypotension [see Warnings and Precautions ( 5.9)]
- Gastrointestinal Adverse Reactions [see Warnings and Precautions ( 5.11)]
- Seizures [see Warnings and Precautions ( 5.12)]
- Withdrawal [see Warnings and Precautions ( 5.13)]
The following adverse reactions associated with the use of morphine were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Serious adverse reactions associated with Morphine Sulfate Injection included, respiratory depression, apnea, and to a lesser degree, circulatory depression, respiratory arrest, shock and cardiac arrest. Rarely, anaphylactoid reactions have been reported when morphine or other phenanthrene alkaloids of opium are administered intravenously.
The most frequently observed adverse reactions included sedation, lightheadedness, dizziness, nausea, vomiting, constipation and diaphoresis.
Other possible adverse reactions include:
Central Nervous System – Euphoria, dysphoria, weakness, headache, agitation, tremor, uncoordinated muscle movements, visual disturbances, transient hallucinations and disorientation.
Gastrointestinal – Constipation, biliary tract spasm.
Cardiovascular – Tachycardia, bradycardia, palpitation, faintness, syncope and orthostatic hypotension.
Genitourinary – Oliguria and urinary retention; an antidiuretic effect has been reported.
Allergic – Pruritus, urticaria and skin rashes. Anaphylactoid reactions have been reported following intravenous administration.
Other – Opioid-induced histamine release may be responsible for the flushing of the face, diaphoresis and pruritus often seen with these drugs. Wheals and urticaria at the site of injection are probably related to histamine release. Local tissue irritation, pain and induration have been reported following repeated subcutaneous injection.
Androgen deficiency: Cases of androgen deficiency have occurred with chronic use of opioids [see Clinical Pharmacology ( 12.2)] .
Anaphylaxis: Anaphylaxis has been reported with ingredients contained in Morphine Sulfate Injection.
Serotonin syndrome: Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal insufficiency: Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
-
7 DRUG INTERACTIONS
Table 1 includes clinically significant drug interactions with Morphine Sulfate Injection.
Table 1: Clinically Significant Drug Interactions with Morphine Sulfate Injection Benzodiazepines and Other Central Nervous System (CNS) Depressants
Clinical Impact:
Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death.
Intervention:
Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients closely for signs of respiratory depression and sedation [see Warnings and Precautions ( 5.4)] .
Examples:
Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol.
Serotonergic Drugs
Clinical Impact:
The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome.
Intervention:
If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Morphine Sulfate Injection if serotonin syndrome is suspected.
Examples:
Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue).
Monoamine Oxidase Inhibitors (MAOIs)
Clinical Impact:
MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions ( 5.7)].
Intervention:
Do not use Morphine Sulfate Injection in patients taking MAOIs or within 14 days of stopping such treatment.
If urgent use of an opioid is necessary, use test doses and frequent titration of small doses of other opioids (such as oxycodone, hydrocodone, oxymorphone, or buprenorphine) to treat pain while closely monitoring blood pressure and signs and symptoms of CNS and respiratory depression.
Examples:
phenelzine, tranylcypromine, linezolid
Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics
Clinical Impact:
May reduce the analgesic effect of Morphine Sulfate Injection and/or precipitate withdrawal symptoms.
Intervention:
Avoid concomitant use.
Examples:
butorphanol, nalbuphine, pentazocine, buprenorphine.
Muscle Relaxants
Clinical Impact:
Morphine may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression.
Intervention:
Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Morphine Sulfate Injection and/or the muscle relaxant as necessary.
Cimetidine
Clinical Impact:
The concomitant administration of morphine sulfate and cimetidine has been reported to precipitate apnea, confusion and muscle twitching in an isolated report.
Intervention:
Monitor patients for increased respiratory and CNS depression when receiving cimetidine concomitantly with Morphine Sulfate Injection.
Diuretics
Clinical Impact:
Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone.
Intervention:
Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed.
Anticholinergic Drugs
Clinical Impact:
The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus.
Intervention:
Monitor patients for signs of urinary retention or reduced gastric motility when Morphine Sulfate Injection is used concomitantly with anticholinergic drugs.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Prolonged use of opioid analgesics during pregnancy can cause neonatal opioid withdrawal syndrome [see Warnings and Precautions ( 5.3)]. There are no available data with Morphine Sulfate Injection in pregnant women to inform a drug-associated risk for major birth defects and miscarriage. Published studies with morphine use during pregnancy have not reported a clear association with morphine and major birth defects [see Human Data]. In published animal reproduction studies, morphine administered subcutaneously during the early gestational period produced neural tube defects (i.e., exencephaly and cranioschisis) at 5 and 16 times the human daily dose of 60 mg based on body surface area (HDD) in hamsters and mice, respectively, lower fetal body weight and increased incidence of abortion at 0.4 times the HDD in the rabbit, growth retardation at 6 times the HDD in the rat, and axial skeletal fusion and cryptorchidism at 16 times the HDD in the mouse. Administration of morphine sulfate to pregnant rats during organogenesis and through lactation resulted in cyanosis, hypothermia, decreased brain weights, pup mortality, decreased pup body weights, and adverse effects on reproductive tissues at 3-4 times the HDD; and long-term neurochemical changes in the brain of offspring which correlate with altered behavioral responses that persist through adulthood at exposures comparable to and less than the HDD [see Animal Data] . Based on animal data, advise pregnant women of the potential risk to a fetus.
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Prolonged use of opioid analgesics during pregnancy for medical or nonmedical purposes can result in physical dependence in the neonate and neonatal opioid withdrawal syndrome shortly after birth. Neonatal opioid withdrawal syndrome presents as irritability, hyperactivity and abnormal sleep pattern, high pitched cry, tremor, vomiting, diarrhea, and failure to gain weight. The onset, duration, and severity of neonatal opioid withdrawal syndrome vary based on the specific opioid used, duration of use, timing and amount of last maternal use, and rate of elimination of the drug by the newborn. Observe newborns for symptoms of neonatal opioid withdrawal syndrome and manage accordingly [see Warnings and Precautions ( 5.3)] .
Labor or Delivery
Opioids cross the placenta and may produce respiratory depression and psycho-physiologic effects in neonates. An opioid antagonist, such as naloxone, must be available for reversal of opioid-induced respiratory depression in the neonate. Morphine Sulfate Injection is not recommended for use in pregnant women during or immediately prior to labor, when other analgesic techniques are more appropriate. Opioid analgesics, including Morphine Sulfate Injection, can prolong labor through actions which temporarily reduce the strength, duration, and frequency of uterine contractions. However, this effect is not consistent and may be offset by an increased rate of cervical dilation, which tends to shorten labor. Monitor neonates exposed to opioid analgesics during labor for signs of excess sedation and respiratory depression.
Data
Human Data
The results from a population-based prospective cohort, including 70 women exposed to morphine during the first trimester of pregnancy and 448 women exposed to morphine at any time during pregnancy, indicate no increased risk for congenital malformations. However, these studies cannot definitely establish the absence of any risk because of methodological limitations, including small sample size and non-randomized study design.
Animal Data
Formal reproductive and developmental toxicology studies for morphine have not been conducted. Exposure margins for the following published study reports are based on human daily dose of 60 mg morphine using a body surface area comparison (HDD).
Neural tube defects (exencephaly and cranioschisis) were noted following subcutaneous administration of morphine sulfate (35-322 mg/kg) on Gestation Day 8 to pregnant hamsters (4.7 to 43.5 times the HDD). A no adverse effect level was not defined in this study and the findings cannot be clearly attributed to maternal toxicity. Neural tube defects (exencephaly), axial skeletal fusions, and cryptorchidism were reported following a single subcutaneous (SC) injection of morphine sulfate to pregnant mice (100-500 mg/kg) on Gestation Day 8 or 9 at 200 mg/kg or greater (16 times the HDD) and fetal resorption at 400 mg/kg or higher (32 times the HDD). No adverse effects were noted following 100 mg/kg morphine in this model (8 times the HDD). In one study, following continuous subcutaneous infusion of doses greater than or equal to 2.72 mg/kg to mice (0.2 times the HDD), exencephaly, hydronephrosis, intestinal hemorrhage, split supraoccipital, malformed sternebrae, and malformed xiphoid were noted. The effects were reduced with increasing daily dose; possibly due to rapid induction of tolerance under these infusion conditions. The clinical significance of this report is not clear.
Decreased fetal weights were observed in pregnant rats treated with 20 mg/kg/day morphine sulfate (3.2 times the HDD) from Gestation Day 7 to 9. There was no evidence of malformations despite maternal toxicity (10% mortality). In a second rat study, decreased fetal weight and increased incidences of growth retardation were noted at 35 mg/kg/day (5.7 times the HDD) and there was a reduced number of fetuses at 70 mg/kg/day (11.4 times the HDD) when pregnant rats were treated with 10, 35, or 70 mg/kg/day morphine sulfate via continuous infusion from Gestation Day 5 to 20. There was no evidence of fetal malformations or maternal toxicity.
An increased incidence of abortion was noted in a study in which pregnant rabbits were treated with 2.5 (0.8 times the HDD) to 10 mg/kg morphine sulfate via subcutaneous injection from Gestation Day 6 to 10. In a second study, decreased fetal body weights were reported following treatment of pregnant rabbits with increasing doses of morphine (10-50 mg/kg/day) during the pre-mating period and 50 mg/kg/day (16 times the HDD) throughout the gestation period. No overt malformations were reported in either publication; although only limited endpoints were evaluated.
In published studies in rats, exposure to morphine during gestation and/or lactation periods is associated with: decreased pup viability at 12.5 mg/kg/day or greater (2 times the HDD); decreased pup body weights at 15 mg/kg/day or greater (2.4 times the HDD); decreased litter size, decreased absolute brain and cerebellar weights, cyanosis, and hypothermia at 20 mg/kg/day (3.2 times the HDD); alteration of behavioral responses (play, social-interaction) at 1 mg/kg/day or greater (0.2 times the HDD); alteration of maternal behaviors (e.g., decreased nursing and pup retrievals) in mice at 1 mg/kg or higher (0.08 times the HDD) and rats at 1.5 mg/kg/day or higher (0.2 times the HDD); and a host of behavioral abnormalities in the offspring of rats, including altered responsiveness to opioids at 4 mg/kg/day (0.7 times the HDD) or greater.
Fetal and/or postnatal exposure to morphine in mice and rats has been shown to result in morphological changes in fetal and neonatal brain and neuronal cell loss, alteration of a number of neurotransmitter and neuromodulator systems, including opioid and non-opioid systems, and impairment in various learning and memory tests that appear to persist into adulthood. These studies were conducted with morphine treatment usually in the range of 4 to 20 mg/kg/day (0.7 to 3.2 times the HDD).
Additionally, delayed sexual maturation and decreased sexual behaviors in female offspring at 20 mg/kg/day (3.2 times the HDD), and decreased plasma and testicular levels of luteinizing hormone and testosterone, decreased testes weights, seminiferous tubule shrinkage, germinal cell aplasia, and decreased spermatogenesis in male offspring were also observed at 20 mg/kg/day (3.2 times the HDD). Decreased litter size and viability were observed in the offspring of male rats that were intraperitoneally administered morphine sulfate for 1 day prior to mating at 25 mg/kg/day (4.1 times the HDD) and mated to untreated females. Decreased viability and body weight and/or movement deficits in both first and second generation offspring were reported when male mice were treated for 5 days with escalating doses of 120 to 240 mg/kg/day morphine sulfate (9.7 to 19.5 times the HDD) or when female mice treated with escalating doses of 60 to 240 mg/kg/day (4.9 to 19.5 times the HDD) followed by a 5-day treatment-free recovery period prior to mating. Similar multigenerational findings were also seen in female rats pre-gestationally treated with escalating doses of 10 to 22 mg/kg/day morphine (1.6 to 3.6 times the HDD).
8.2 Lactation
Risk Summary
Morphine is present in breast milk. Published lactation studies report variable concentrations of morphine in breast milk with administration of immediate-release morphine to nursing mothers in the early postpartum period with a milk-to-plasma morphine AUC ratio of 2.5:1 measured in one lactation study. However, there is insufficient information to determine the effects of morphine on the breastfed infant and the effects of morphine on milk production. Lactation studies have not been conducted with Morphine Sulfate Injection and no information is available on the effects of the drug on the breastfed infant or the effects of the drug on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Morphine Sulfate Injection and any potential adverse effects on the breastfed infant from Morphine Sulfate Injection or from the underlying maternal condition.
Clinical Considerations
Infants exposed to Morphine Sulfate Injection through breast milk should be monitored for excess sedation and respiratory depression. Withdrawal symptoms can occur in breastfed infants when maternal administration of an opioid analgesic is stopped, or when breast-feeding is stopped.
8.3 Females and Males of Reproductive Potential
Infertility
Chronic use of opioids may cause reduced fertility in females and males of reproductive potential. It is not known whether these effects on fertility are reversible [see Adverse Reactions ( 6)].
In published animal studies, morphine administration adversely effected fertility and reproductive endpoints in male rats and prolonged estrus cycle in female rats [see Nonclinical Toxicology ( 13)] .
8.4 Pediatric Use
The safety and effectiveness of Morphine Sulfate Injection in pediatric patients below the age of 18 have not been established.
8.5 Geriatric Use
The pharmacodynamic effects of morphine in the elderly are more variable than in the younger population. Older patients will vary widely in the effective initial dose, rate of development of tolerance and the frequency and magnitude of associated adverse effects as the dose is increased.
Elderly patients (aged 65 years or older) may have increased sensitivity to morphine. In general, use caution when selecting a dosage for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
Respiratory depression is the chief risk for elderly patients treated with opioids, and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of Morphine Sulfate Injection slowly in geriatric patients and monitor for signs of central nervous system and respiratory depression [see Warnings and Precautions ( 5.6)] .
Morphine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Hepatic Impairment
Morphine sulfate pharmacokinetics have been reported to be significantly altered in patients with cirrhosis. Start these patients with a lower than normal dosage of Morphine Sulfate Injection and titrate slowly while monitoring for signs of respiratory depression, sedation, and hypotension [see Clinical Pharmacology ( 12.3)].
8.7 Renal Impairment
Morphine sulfate pharmacokinetics are altered in patients with renal failure. Start these patients with a lower than normal dosage of Morphine Sulfate Injection and titrate slowly while monitoring for signs of respiratory depression, sedation, and hypotension [see Clinical Pharmacology ( 12.3)].
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Morphine Sulfate Injection contains morphine, a Schedule II controlled substance.
9.2 Abuse
Morphine Sulfate Injection contains morphine, a substance with a high potential for abuse similar to other opioids including fentanyl, hydrocodone, hydromorphone, methadone, oxycodone, oxymorphone, and tapentadol. Morphine Sulfate Injection can be abused and is subject to misuse, addiction, and criminal diversion [see Warnings and Precautions ( 5.1)].
All patients treated with opioids require careful monitoring for signs of abuse and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use.
Prescription drug abuse is the intentional non-therapeutic use of a prescription drug, even once, for its rewarding psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that develop after repeated substance use and includes: a strong desire to take the drug, difficulties in controlling its use, persisting in its use despite harmful consequences, a higher priority given to drug use than to other activities and obligations, increased tolerance, and sometimes a physical withdrawal.
“Drug-seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among drug abusers and people suffering from untreated addiction. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with poor pain control.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Health care providers should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence in all addicts. In addition, abuse of opioids can occur in the absence of true addiction.
Morphine Sulfate Injection, like other opioids, can be diverted for non-medical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
9.3 Dependence
Both tolerance and physical dependence can develop during chronic opioid therapy. Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Tolerance may occur to both the desired and undesired effects of drugs, and may develop at different rates for different effects.
Physical dependence results in withdrawal symptoms after abrupt discontinuation or a significant dosage reduction of a drug. Withdrawal also may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone, nalmefene), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued opioid usage.
Morphine Sulfate Injection should not be abruptly discontinued in a physically-dependent patient [see Dosage and Administration ( 2.4)]. If Morphine Sulfate Injection is abruptly discontinued in a physically-dependent patient, a withdrawal syndrome may occur. Some or all of the following can characterize this syndrome: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations ( 8.1)] .
-
10 OVERDOSAGE
Clinical Presentation
Acute overdose with Morphine Sulfate Injection can be manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, in some cases, pulmonary edema, bradycardia, hypotension, partial or complete airway obstruction, snoring, and death. Marked mydriasis rather than miosis may be seen with hypoxia in overdose [see Clinical Pharmacology ( 12.2)] .
Treatment of Overdose
In case of overdose, priorities are the reestablishment of a patent and protected airway and institution of assisted or controlled ventilation, if needed. Employ other supportive measures (including oxygen and vasopressors) in the management of circulatory shock and pulmonary edema as indicated. Cardiac arrest or arrhythmias will require advanced life-support techniques.
The opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression resulting from opioid overdose. For clinically significant respiratory or circulatory depression secondary to morphine overdose, administer an opioid antagonist. Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to morphine overdose.
Because the duration of opioid reversal is expected to be less than the duration of action of morphine in Morphine Sulfate Injection, carefully monitor the patient until spontaneous respiration is reliably reestablished. If the response to an opioid antagonist is suboptimal or only brief in nature, administer additional antagonist as directed by the product's prescribing information.
In an individual physically dependent on opioids, administration of the recommended usual dosage of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal symptoms experienced will depend on the degree of physical dependence and the dose of the antagonist administered. If a decision is made to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be begun with care and by titration with smaller than usual doses of the antagonist.
-
11 DESCRIPTION
Morphine sulfate is an opioid agonist. Morphine Sulfate Injection USP is available as a sterile, nonpyrogenic solution of morphine sulfate, free of antioxidants and preservatives in pre-filled syringes for intravenous and intramuscular administration. Each 1 mL pre-filled syringe contains 2 mg, 4 mg, 5 mg, 8 mg or 10 mg of Morphine Sulfate USP in 1 mL total volume.
The chemical name is 7,8-Didehydro-4,5-epoxy-17-methyl-(5α,6α)-morphinan-3,6diol sulfate (2: 1) (salt), pentahydrate. The molecular weight is 758.83. Its molecular formula is (C 17H 19NO 3) 2 H 2SO 4 5H 2O and it has the following chemical structure.
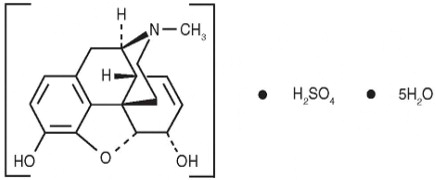
Morphine sulfate is a fine white powder. When exposed to air it gradually loses water of hydration, and darkens on prolonged exposure to light. It is soluble in water and ethanol at room temperature.
The inactive ingredients in Morphine Sulfate Injection, USP include:
- For the 2 mg/mL and 4 mg/mL products: 8.4 mg sodium chloride, 2.3 mg of sodium citrate, 0.74 mg of citric acid, 0.111 mg of edetate disodium, 0.053 mg of calcium chloride and water for injection.
- For the 5 mg/mL, 8 mg/mL and 10 mg/mL products: 7.5 mg sodium chloride, 3.45 mg of sodium citrate, 1.11 mg of citric acid, 0.111 mg of edetate disodium, 0.053 mg of calcium chloride and water for injection.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Morphine is a full opioid agonist and is relatively selective for the mu-opioid receptor, although it can bind to other opioid receptors at higher doses. The principal therapeutic action of morphine is analgesia. Like all full opioid agonists, there is no ceiling effect for analgesia with morphine. Clinically, dosage is titrated to provide adequate analgesia and may be limited by adverse reactions, including respiratory and CNS depression.
The precise mechanism of the analgesic action is unknown. However, specific CNS opioid receptors for endogenous compounds with opioid-like activity have been identified throughout the brain and spinal cord and are thought to play a role in the analgesic effects of this drug.
12.2 Pharmacodynamics
Effects on the Central Nervous System
Morphine produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves a reduction in the responsiveness of the brain stem respiratory centers to both increases in carbon dioxide tension and electrical stimulation.
Morphine causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen due to hypoxia in overdose situations.
Effects on the Gastrointestinal Tract and Other Smooth Muscle
Morphine causes a reduction in motility associated with an increase in smooth muscle tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm, resulting in constipation. Other opioid-induced effects may include a reduction in biliary and pancreatic secretions, spasm of sphincter of Oddi, and transient elevations in serum amylase. Morphine may also cause spasm of the sphincter of the urinary bladder.
Effects on the Cardiovascular System
In therapeutic doses, morphine does not usually exert major effects on the cardiovascular system. Morphine produces peripheral vasodilation, which may result in orthostatic hypotension or syncope. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes sweating and/or orthostatic hypotension.
Effects on the Endocrine System
Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans. [see Adverse Reactions ( 6)]. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon .
Chronic use of opioids may influence the hypothalamic-pituitary-gonadal axis, leading to androgen deficiency that may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility. The causal role of opioids in the clinical syndrome of hypogonadism is unknown because the various medical, physical, lifestyle, and psychological stressors that may influence gonadal hormone levels have not been adequately controlled for in studies conducted to date [see Adverse Reactions ( 6)] .
Effects on the Immune System
Opioids have been shown to have a variety of effects on components of the immune system in in vitro and animal models. The clinical significance of these findings is unknown. Overall, the effects of opioids appear to be modestly immunosuppressive.
Concentration–Efficacy Relationships
The minimum effective analgesic concentration will vary widely among patients, especially among patients who have been previously treated with potent agonist opioids. The minimum effective analgesic concentration of morphine for any individual patient may increase over time due to an increase in pain, the development of a new pain syndrome, and/or the development of analgesic tolerance. [see Dosage and Administration ( 2.1, 2.3)]
Onset of analgesia occurs with 5-20 minutes following intramuscular administration of morphine, rising to peak analgesia sixty minutes after a single intramuscular injection. The duration of analgesia after a single injection is usually three to four hours.
Concentration–Adverse Reaction Relationships
There is a relationship between increasing morphine plasma concentration and increasing frequency of dose-related opioid adverse reactions such as nausea, vomiting, CNS effects, and respiratory depression. In opioid-tolerant patients, the situation may be altered by the development of tolerance to opioid-related adverse reactions [see Dosage and Administration ( 2.1, 2.2)] .
12.3 Pharmacokinetics
Absorption: Average peak morphine plasma levels of 67.4 ± 22.5 ng/mL were noted around 5 to 30 minutes following intramuscular injection of 10 mg morphine sulfate from a prefilled syringe.
Distribution: Morphine has an apparent volume of distribution ranging from 1.0 to 4.7 L/kg after intravenous dosage. Protein binding is low, about 36%, and muscle tissue binding is reported as 54%. A blood-brain barrier exists, and when morphine is introduced outside of the CNS (e.g., intravenously), plasma concentrations of morphine remain higher than the corresponding CSF morphine sulfate levels.
Elimination:
Morphine has a total plasma clearance which ranges from 0.9 to 1.2 L/kg/h (liters/kilogram/hour) in postoperative patients, but shows considerable interindividual variation. Terminal half-life is commonly reported to vary from 1.5 to 4.5 hours, although the longer half-lives were obtained when morphine levels were monitored over protracted periods with very sensitive radioimmunoassay methods. The accepted elimination half-life in normal subjects is 1.5 to 2 hours.
Specific Populations
Sex
While evidence of greater post-operative Morphine Sulfate Injection consumption in men compared to women is present in the literature, clinically significant differences in analgesic outcomes and pharmacokinetic parameters have not been consistently demonstrated. Some studies have shown an increased sensitivity to the adverse effects of Morphine Sulfate Injection USP, including respiratory depression, in women compared to men.
Hepatic Impairment
Morphine pharmacokinetics are altered in patients with cirrhosis. Clearance was found to decrease with a corresponding increase in half-life. The M3G and M6G to morphine sulfate AUC ratio is also decreased in these subjects, indicating diminished metabolic activity. Adequate studies of the pharmacokinetics of morphine in patients with severe hepatic impairment have not been conducted.
Renal Impairment
Morphine pharmacokinetics are altered in patients with renal failure. AUC is increased and clearance is decreased and the metabolites, M3G and M6G, may accumulate to much higher plasma levels in patients with renal failure as compared to patients with normal renal function. Adequate studies of the pharmacokinetics of morphine in patients with severe renal impairment have not been conducted.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term studies in animals to evaluate the carcinogenic potential of morphine have not been conducted.
Mutagenesis
No formal studies to assess the mutagenic potential of morphine have been conducted. In the published literature, morphine was found to be mutagenic in vitro increasing DNA fragmentation in human T-cells. Morphine was also reported to be mutagenic in the in vivo mouse micronucleus assay and positive for the induction of chromosomal aberrations in mouse spermatids and murine lymphocytes. Mechanistic studies suggest that the in vivo clastogenic effects reported with morphine in mice may be related to increases in glucocorticoid levels produced by morphine in these species. In contrast to the above positive findings, in vitro studies in the literature have also shown that morphine did not induce chromosomal aberrations in human leukocytes or translocations or lethal mutations in Drosophila.
Impairment of Fertility
No formal nonclinical studies to assess the potential of morphine to impair fertility have been conducted.
Several nonclinical studies from the literature have demonstrated adverse effects on male fertility in the rat from exposure to morphine. One study in which male rats were administered morphine sulfate subcutaneously prior to mating (up to 30 mg/kg twice daily) and during mating (20 mg/kg twice daily) with untreated females, a number of adverse reproductive effects including reduction in total pregnancies and higher incidence of pseudopregnancies at 20 mg/kg/day (3.2 times the HDD) were reported.
Studies from the literature have also reported changes in hormonal levels in male rats (i.e. testosterone, luteinizing hormone) following treatment with morphine at 10 mg/kg/day or greater (1.6 times the HDD).
Female rats that were administered morphine sulfate intraperitoneally prior to mating exhibited prolonged estrous cycles at 10 mg/kg/day (1.6 times the HDD).
Exposure of adolescent male rats to morphine has been associated with delayed sexual maturation and following mating to untreated females, smaller litters, increased pup mortality, and/or changes in reproductive endocrine status in adult male offspring have been reported (estimated 5 times the plasma levels at the HDD).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Morphine Sulfate Injection is available for intravenous (IV) or intramuscular (IM) use as:
2 mg/mL in 1 mL pre-filled disposable syringe, NDC: 76045-004-10
4 mg/mL in 1 mL pre-filled disposable syringe, NDC: 76045-005-10
5 mg/mL in 1 mL pre-filled disposable syringe, NDC: 76045-006-10
8 mg/mL in 1 mL pre-filled disposable syringe, NDC: 76045-007-10
10 mg/mL in 1 mL pre-filled disposable syringe, NDC: 76045-008-10
Available in a carton of twenty-four (24) syringes for each strength.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature.]
PROTECT FROM LIGHT. DO NOT FREEZE.
This product is for single dose only.
Contains no preservative or antioxidant.
DISCARD ANY UNUSED PORTION.
DO NOT HEAT-STERILIZE.
DO NOT place syringe on a sterile field.DO NOT autoclave syringe.
DO NOT introduce any other fluid into the syringe at any time.
All steps must be done sequentially.
-
17 PATIENT COUNSELING INFORMATION
Serotonin Syndrome
Opioids can cause a rare but potentially life-threatening condition resulting from concomitant administration of serotonergic drugs. Instruct patients to inform their healthcare providers if they are taking, or plan to take serotonergic medications [see Drug Interactions ( 7) ].
Constipation
Advise patients of the potential for severe constipation [see Clinical Pharmacology ( 12.2)] .
-
INSTRUCTIONS FOR USE
INSTRUCTIONS FOR USE
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if color is darker than pale yellow, if it is discolored in any other way or if it contains a precipitate.
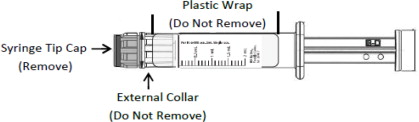
CAUTION: Certain glass syringes may malfunction, break or clog when connected to some Needleless Luer Access Devices (NLADs) and needles. This syringe has a larger internal syringe tip and an external collar (luer collar). The external collar must remain attached to the syringe. Data show that the syringe achieves acceptable connections with the BD Eclipse™ Needle and the Terumo SurGuard2™ Safety Needle and with the following non-center post NLADs: Alaris SMARTSITE™, B-Braun ULTRASITE™, BD-Q SYTE™, Maximum MAX PLUS™, and B-Braun SAFSITE™. The data also show acceptable connections are achieved to the center post ICU Medical CLAVE™. However, spontaneous disconnection of this glass syringe from needles and NLADs with leakage of drug product may occur. Assure that the needle or NLAD is securely attached before beginning the injection. Visually inspect the glass syringe-needle or glass syringe –NLAD connection before and during drug administration. Do not remove the clear plastic wrap around the external collar. (See Figure 1)
Figure 1
1. Inspect the outer packaging (blister pack) by verifying:
- blister integrity
- drug name
- drug strength
- dose volume
- route of administration
- expiration date to be sure that the drug has not expired
- sterile field applicability
Do not use if package has been damaged.
2. Peel open the paper (top web) of the outer packaging that displays the product information to access the syringe. Do not pop syringe through.
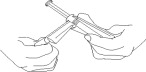
3. Bend the plastic part of the outer packaging (thermoform) so as to present the plunger rod for syringe removal. (See Figure 2)
Figure 2
4. Perform visual inspection on the syringe by verifying:
- absence of syringe damage
- absence of external particles
- absence of internal particles
- proper drug color
- expiration date to be sure that the drug has not expired
- drug name
- drug strength
- dose volume
- route of administration
- sterile field applicability
- integrity of the plastic wrap around the external collar
5. Do not remove plastic wrap around the external collar. Push plunger rod slightly to break the stopper loose while tip cap is still on.
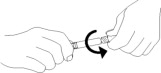
6. Do not remove plastic wrap around the external collar. Remove tip cap by twisting it off. (See Figure 3)
Figure 3
7. Discard the tip cap.
8. Expel air bubble.
9. Adjust dose into sterile material (if applicable).
10. Connect the syringe to appropriate injection connection depending on route of administration. Before injection, ensure that the syringe is securely attached to the needle or needleless luer access device (NLAD).
11. Depress plunger rod to deliver medication. Ensure that pressure is maintained on the plunger rod during the entire administration.
12. Remove syringe from NLAD (if applicable) and discard into appropriate receptacle. If delivering medication via intramuscular (IM) route, do not recap needle. To prevent needle-stick injuries, needles should not be recapped.
For more information concerning this drug, please call Fresenius Kabi USA, LLC at 1-800-551-7176.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
The brand names mentioned in this document are the trademarks of their respective owners.

Lake Zurich, IL 60047
www.fresenius-kabi.us
451509C
Rev. 01/2018
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY – Morphine 1 mL Carton Panel
Rx only NDC: 76045-004-10
Morphine Sulfate Injection, USP CII
2 mg/mL
For Intramuscular or Intravenous use.Do NOT place syringe on a Sterile Field.
24 X 1 mL Prefilled single-use syringes
Discard unused portion
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY - Morphine 1 mL Blister Pack Label
Rx only NDC: 76045-004-10
Morphine Sulfate Injection, USP CII
2 mg/mL
For IM or IV use.1 mL Prefilled single use syringe

- PRINCIPAL DISPLAY PANEL
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY – Morphine 1 mL Carton Panel
Rx only NDC: 76045-005-10
Morphine Sulfate Injection, USP CII
4 mg/mL
For Intramuscular or Intravenous use.Do NOT place syringe on a Sterile Field.
24 X 1 mL Prefilled single-use syringes
Discard unused portion
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY - Morphine 1 mL Blister Pack Label
Rx only NDC: 76045-005-10
Morphine Sulfate Injection, USP CII
4 mg/mL
For IM or IV use.1 mL Prefilled single use syringe

- PRINCIPAL DISPLAY PANEL
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY – Morphine 1 mL Carton Panel
Rx only NDC: 76045-006-10
Morphine Sulfate Injection, USP CII
5 mg/mL
For Intramuscular or Intravenous use.Do NOT place syringe on a Sterile Field.
24 X 1 mL Prefilled single-use syringes
Discard unused portion
-
PRINCIPAL DISPLAY PANEL
PACKAGE LABEL - PRINCIPAL DISPLAY - Morphine 1 mL Blister Pack Label
Rx only NDC: 76045-006-10
Morphine Sulfate Injection, USP CII
5 mg/mL
For IM or IV use.1 mL Prefilled single use syringe

- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MORPHINE SULFATE
morphine sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76045-004 Route of Administration INTRAVENOUS, INTRAMUSCULAR DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MORPHINE SULFATE (UNII: X3P646A2J0) (MORPHINE - UNII:76I7G6D29C) MORPHINE SULFATE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76045-004-10 24 in 1 CARTON 10/30/2013 1 1 in 1 BLISTER PACK 1 1 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204223 10/30/2013 MORPHINE SULFATE
morphine sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76045-005 Route of Administration INTRAVENOUS, INTRAMUSCULAR DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MORPHINE SULFATE (UNII: X3P646A2J0) (MORPHINE - UNII:76I7G6D29C) MORPHINE SULFATE 4 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76045-005-10 24 in 1 CARTON 10/30/2013 1 1 in 1 BLISTER PACK 1 1 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204223 10/30/2013 MORPHINE SULFATE
morphine sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76045-006 Route of Administration INTRAVENOUS, INTRAMUSCULAR DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MORPHINE SULFATE (UNII: X3P646A2J0) (MORPHINE - UNII:76I7G6D29C) MORPHINE SULFATE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76045-006-10 24 in 1 CARTON 10/30/2013 1 1 in 1 BLISTER PACK 1 1 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204223 10/30/2013 MORPHINE SULFATE
morphine sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76045-007 Route of Administration INTRAVENOUS, INTRAMUSCULAR DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MORPHINE SULFATE (UNII: X3P646A2J0) (MORPHINE - UNII:76I7G6D29C) MORPHINE SULFATE 8 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76045-007-10 24 in 1 CARTON 10/30/2013 11/30/2018 1 1 in 1 BLISTER PACK 1 1 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204223 10/30/2013 MORPHINE SULFATE
morphine sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 76045-008 Route of Administration INTRAVENOUS, INTRAMUSCULAR DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MORPHINE SULFATE (UNII: X3P646A2J0) (MORPHINE - UNII:76I7G6D29C) MORPHINE SULFATE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) EDETATE DISODIUM (UNII: 7FLD91C86K) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76045-008-10 24 in 1 CARTON 10/30/2013 11/30/2018 1 1 in 1 BLISTER PACK 1 1 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204223 10/30/2013 Labeler - Fresenius Kabi USA, LLC (608775388) Establishment Name Address ID/FEI Business Operations Fresenius Kabi USA, LLC 080381675 analysis(76045-004, 76045-005, 76045-006, 76045-007, 76045-008) , manufacture(76045-004, 76045-005, 76045-006, 76045-007, 76045-008)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.


