Acetaminophen by Preferred Pharmaceuticals Inc. ACETAMINOPHEN tablet
Acetaminophen by
Drug Labeling and Warnings
Acetaminophen by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
- Uses
-
Warnings
Liver warning:This product contains acetaminophen.
Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert:acetaminophen may cause severe skin reactions.
Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- do not take more than directed (see overdose warning)
adults and children 12 years and over
- take 2 caplets every 6 hours while symptoms last
- do not take more than 6 caplets in 24 hours, unless directed by a doctor
- do not use for more than 10 days unless directed by a doctor
children under 12 years
ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
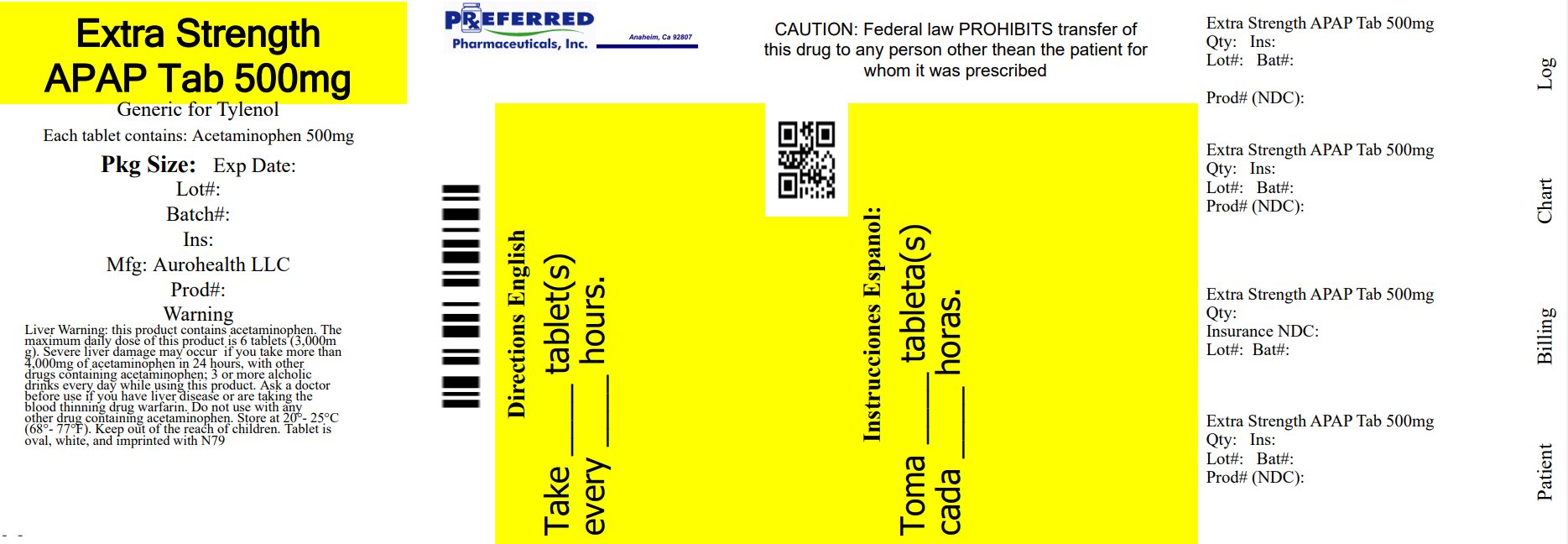
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL 500 mg
AUROHEALTH
NDC: 68788-8559-1
Compare to the active ingredient
in Tylenol ® Extra strength Caplets *
See New Warnings Information & Directions
EXTRA STRENGTH
For Adults
Pain Relief
Acetaminophen
Caplets 500 mg
Pain reliever/Fever reducer
Extra Strength APAP Tablets 500mg aurohealth
500 mg each
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN
acetaminophen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68788-8559(NDC:58602-773) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength POVIDONE K30 (UNII: U725QWY32X) STARCH, CORN (UNII: O8232NY3SJ) PALMITOSTEARIC ACID (UNII: Q8Y7S3B85M) Product Characteristics Color white (White to off White) Score no score Shape CAPSULE (Biconvex) Size 18mm Flavor Imprint Code N79 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68788-8559-1 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/08/2024 2 NDC: 68788-8559-2 20 in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2025 3 NDC: 68788-8559-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2025 4 NDC: 68788-8559-5 50 in 1 BOTTLE; Type 0: Not a Combination Product 05/06/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 01/08/2024 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-8559)
Trademark Results [Acetaminophen]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ACETAMINOPHEN 85615223 not registered Dead/Abandoned |
General Merchandise importers and Expoters 2012-05-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.