These highlights do not include all the information needed to use CIPROFLOXACIN EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for CIPROFLOXACIN EXTENDED-RELEASE TABLETS. CIPROFLOXACIN extended-release tablets, for oral useInitial U.S. Approval: 1987
Ciprofloxacin by
Drug Labeling and Warnings
Ciprofloxacin by is a Prescription medication manufactured, distributed, or labeled by Mylan Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CIPROFLOXACIN- ciprofloxacin tablet, film coated, extended release
Mylan Pharmaceuticals Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use CIPROFLOXACIN EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information for CIPROFLOXACIN EXTENDED-RELEASE TABLETS.
CIPROFLOXACIN extended-release tablets, for oral use Initial U.S. Approval: 1987 WARNING: SERIOUS ADVERSE REACTIONS INCLUDING TENDINITIS, TENDON RUPTURE, PERIPHERAL NEUROPATHY, CENTRAL NERVOUS SYSTEM EFFECTS AND EXACERBATION OF MYASTHENIA GRAVISSee full prescribing information for complete boxed warning.
Discontinue ciprofloxacin extended-release tablets immediately and avoid the use of fluoroquinolones, including ciprofloxacin extended-release tablets, in patients who experience any of these serious adverse reactions (5.1)
RECENT MAJOR CHANGESINDICATIONS AND USAGECiprofloxacin extended-release tablets are a fluoroquinolone antibacterial indicated in adults (≥ 18 years of age) with infections caused by designated, susceptible bacteria:
Usage To reduce the development of drug-resistant bacteria and maintain the effectiveness of ciprofloxacin extended-release tablets and other antibacterial drugs, ciprofloxacin extended-release tablets should be used only to treat infections that are proven or strongly suspected to be caused by bacteria. (1.4) DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions ≥ 2% were nausea, headache, dizziness, diarrhea, vomiting, and vaginal moniliasis. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONSSee 17 for PATIENT COUNSELING INFORMATION and Medication Guide. Revised: 5/2019 |
FULL PRESCRIBING INFORMATION
WARNING: SERIOUS ADVERSE REACTIONS INCLUDING TENDINITIS, TENDON RUPTURE, PERIPHERAL NEUROPATHY, CENTRAL NERVOUS SYSTEM EFFECTS AND EXACERBATION OF MYASTHENIA GRAVIS
-
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with disabling and potentially irreversible serious adverse reactions that have occurred together [see Warnings and Precautions (5.1)], including:
- o Tendinitis and tendon rupture [see Warnings and Precautions (5.2)]
- o Peripheral neuropathy [see Warnings and Precautions (5.3)]
- o Central nervous system effects [see Warnings and Precautions (5.4)]
Discontinue ciprofloxacin extended-release tablets immediately and avoid the use of fluoroquinolones, including ciprofloxacin extended-release tablets, in patients who experience any of these serious adverse reactions [see Warnings and Precautions (5.1)].
- Fluoroquinolones, including ciprofloxacin extended-release tablets, may exacerbate muscle weakness in patients with myasthenia gravis. Avoid ciprofloxacin extended-release tablets in patients with known history of myasthenia gravis [see Warnings and Precautions (5.5)].
- Because fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with serious adverse reactions [see Warnings and Precautions (5.1–5.16)], reserve ciprofloxacin extended-release tablets for use in patients who have no alternative treatment options for uncomplicated urinary tract infections [see Indications and Usage (1.1)].
1 INDICATIONS AND USAGE
Ciprofloxacin extended-release tablets are indicated for the treatment of infections caused by susceptible isolates of the designated microorganisms in the conditions and patient populations listed below.
1.1 Uncomplicated Urinary Tract Infections (Acute Cystitis)
Ciprofloxacin extended-release tablets are indicated for the treatment of uncomplicated urinary tract infections (UTIs) caused by Escherichia coli, Proteus mirabilis, Enterococcus faecalis, or Staphylococcus saprophyticus.
Because fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with serious adverse reactions [see Warnings and Precautions (5.1–5.16)] and for some patients uncomplicated UTI (acute cystitis) is self-limiting, reserve ciprofloxacin extended-release tablets for treatment of uncomplicated UTIs (acute cystitis) in patients who have no alternative treatment options.
1.2 Complicated Urinary Tract Infections, and Acute Uncomplicated Pyelonephritis
Ciprofloxacin extended-release tablets are indicated for the treatment of complicated urinary tract infections (cUTI) caused by Escherichia coli, Klebsiella pneumoniae, Enterococcus faecalis, Proteus mirabilis, or Pseudomonas aeruginosa and acute uncomplicated pyelonephritis (AUP) caused by Escherichia coli.
1.3 Limitations of Use
- The safety and efficacy of ciprofloxacin extended-release tablets in treating infections other than urinary tract infections has not been demonstrated.
- Ciprofloxacin extended-release tablets are not indicated for pediatric patients.
1.4 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ciprofloxacin extended-release tablets and other antibacterial drugs, ciprofloxacin extended-release tablets should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Appropriate culture and susceptibility tests should be performed before treatment in order to isolate and identify organisms causing infection and to determine their susceptibility to ciprofloxacin. Therapy with ciprofloxacin extended-release tablets may be initiated before results of these tests are known; once results become available appropriate therapy should be continued.
As with other drugs, some isolates of Pseudomonas aeruginosa may develop resistance fairly rapidly during treatment with ciprofloxacin. Culture and susceptibility testing performed periodically during therapy will provide information not only on the therapeutic effect of the antimicrobial agent but also on the possible emergence of bacterial resistance.
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
Ciprofloxacin extended-release tablets and ciprofloxacin immediate-release tablets are not interchangeable. Ciprofloxacin extended-release tablets should be administered orally once daily (Table 1).
|
Indication |
Dose |
Frequency |
Usual Duration |
|
Uncomplicated Urinary Tract Infection (Acute Cystitis) |
500 mg |
every 24 hours |
3 Days |
|
Complicated Urinary Tract Infection and Acute Uncomplicated Pyelonephritis |
1000 mg |
every 24 hours |
7–14 Days |
Patients whose therapy is started with ciprofloxacin IV for UTIs may be switched to ciprofloxacin extended-release tablets when clinically indicated at the discretion of the physician.
2.2 Administration
- Ciprofloxacin extended-release tablets should be taken whole and not split, crushed, or chewed.
- Ciprofloxacin extended-release tablets should be administered at least 2 hours before or 6 hours after antacids containing magnesium or aluminum, polymeric phosphate binders (for example, sevelamer, lanthanum carbonate), as well as sucralfate, VIDEX® (didanosine) chewable/buffered tablets or pediatric powder, other highly buffered drugs, metal cations such as iron, and multivitamin preparations with zinc [see Drug Interactions (7)].
- Concomitant administration of ciprofloxacin extended-release tablets with dairy products (like milk or yogurt) or with calcium-fortified products alone should be avoided since decreased absorption is possible. A 2-hour window between substantial calcium intake (greater than 800 mg) and dosing with ciprofloxacin extended-release tablets is recommended [see Patient Counseling Information (17)].
- Adequate hydration of patients receiving ciprofloxacin extended-release tablets should be maintained to prevent the formation of highly concentrated urine. Crystalluria has been reported with quinolones [see Warnings and Precautions (5.18), Adverse Reactions (6.1) and Patient Counseling Information (17)].
2.3 Impaired Renal Function
- In patients with cUTI and acute uncomplicated pyelonephritis with a creatinine clearance of ≤ 30 mL/min, the dose of ciprofloxacin extended-release tablets should be reduced from 1000 mg to 500 mg daily. The use of ciprofloxacin 1000 mg extended-release tablets is not recommended in this patient population.
- For patients on hemodialysis or peritoneal dialysis, administer ciprofloxacin extended-release tablets after the dialysis procedure is completed (maximum dose should be ciprofloxacin 500 mg extended-release tablets every 24 hours). The use of ciprofloxacin 1000 mg extended-release tablets is not recommended in this patient population [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
- For patients on continuous ambulatory peritoneal dialysis (CAPD), the maximum dose should be 500 mg every 24 hours.
3 DOSAGE FORMS AND STRENGTHS
Ciprofloxacin Extended-Release Tablets, USP are available containing ciprofloxacin, USP and ciprofloxacin hydrochloride, USP equivalent to 500 mg or 1000 mg of ciprofloxacin.
- The 500 mg tablets are orange, film-coated, modified capsule shaped, unscored tablets debossed with M 1743 on one side of the tablet and blank on the other side.
- The 1000 mg tablets are orange, film-coated, modified capsule shaped, unscored tablets debossed with M 1745 on one side of the tablet and blank on the other side.
4 CONTRAINDICATIONS
4.1 Hypersensitivity
Ciprofloxacin extended-release tablets are contraindicated in persons with a history of hypersensitivity to ciprofloxacin, any member of the quinolone class of antibacterials, or any of the product components [see Warnings and Precautions (5.7)].
4.2 Tizanidine
Concomitant administration with tizanidine is contraindicated [see Drug Interactions (7)].
5 WARNINGS AND PRECAUTIONS
5.1 Disabling and Potentially Irreversible Serious Adverse Reactions Including Tendinitis and Tendon Rupture, Peripheral Neuropathy, and Central Nervous System Effects
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with disabling and potentially irreversible serious adverse reactions from different body systems that can occur together in the same patient. Commonly seen adverse reactions include tendinitis, tendon rupture, arthralgia, myalgia, peripheral neuropathy, and central nervous system effects (hallucinations, anxiety, depression, insomnia, severe headaches, and confusion). These reactions can occur within hours to weeks after starting ciprofloxacin extended-release tablets. Patients of any age or without pre-existing risk factors have experienced these adverse reactions [see Warnings and Precautions (5.2, 5.3, 5.4)].
Discontinue ciprofloxacin extended-release tablets immediately at the first signs or symptoms of any serious adverse reaction. In addition, avoid the use of fluoroquinolones, including ciprofloxacin extended-release tablets, in patients who have experienced any of these serious adverse reactions associated with fluoroquinolones.
5.2 Tendinitis and Tendon Rupture
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with an increased risk of tendinitis and tendon rupture in all ages [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)]. This adverse reaction most frequently involves the Achilles tendon, and has also been reported in the rotator cuff (the shoulder), the hand, the biceps, the thumb, and other tendons. Tendinitis and tendon rupture can occur within hours or days of starting ciprofloxacin extended-release tablets, or as long as several months after completion of fluoroquinolone therapy. Tendinitis and tendon rupture can occur bilaterally.
The risk of developing fluoroquinolone-associated tendinitis and tendon rupture is further increased in patients over 60 years of age, in patients taking corticosteroid drugs, and in patients with kidney, heart or lung transplants. Other factors that may independently increase the risk of tendon rupture include strenuous physical activity, renal failure, and previous tendon disorders such as rheumatoid arthritis. Tendinitis and tendon rupture have also occurred in patients taking fluoroquinolones who do not have the above risk factors.
Discontinue ciprofloxacin extended-release tablets immediately if the patient experiences pain, swelling, inflammation or rupture of a tendon. Avoid fluoroquinolones, including ciprofloxacin extended-release tablets, in patients who have a history of tendon disorders or have experienced tendinitis or tendon rupture [see Adverse Reactions (6.2)].
5.3 Peripheral Neuropathy
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with an increased risk of peripheral neuropathy. Cases of sensory or sensorimotor axonal polyneuropathy affecting small and/or large axons resulting in paresthesias, hypoesthesias, dysesthesias and weakness have been reported in patients receiving fluoroquinolones, including ciprofloxacin extended-release tablets. Symptoms may occur soon after initiation of ciprofloxacin extended-release tablets and may be irreversible in some patients [see Warnings and Precautions (5.1) and Adverse Reactions (6.1, 6.2)].
Discontinue ciprofloxacin extended-release tablets immediately if the patient experiences symptoms of peripheral neuropathy including pain, burning, tingling, numbness, and/or weakness, or other alterations in sensations including light touch, pain, temperature, position sense and vibratory sensation and/or motor strength in order to minimize the development of an irreversible condition. Avoid fluoroquinolones, including ciprofloxacin extended-release tablets, in patients who have previously experienced peripheral neuropathy [see Adverse Reactions (6.1, 6.2)].
5.4 Central Nervous System Effects
Psychiatric Adverse Reactions
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with an increased risk of psychiatric adverse reactions, including: toxic psychosis, psychotic reactions progressing to suicidal ideations/thoughts, hallucinations, or paranoia; depression, or self-injurious behavior such as attempted or completed suicide; anxiety, agitation, or nervousness; confusion, delirium, disorientation, or disturbances in attention; insomnia or nightmares; memory impairment. These reactions may occur following the first dose. Advise patients receiving ciprofloxacin to inform their healthcare provider immediately if these reactions occur, discontinue the drug, and institute appropriate care [see Adverse Reactions (6.1)].
Central Nervous System Adverse Reactions
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with an increased risk of seizures (convulsions), increased intracranial pressure (pseudotumor cerebri), dizziness, and tremors. Ciprofloxacin extended-release tablets, like other fluoroquinolones, are known to trigger seizures or lower the seizure threshold. Cases of status epilepticus have been reported. As with all fluoroquinolones, use ciprofloxacin extended-release tablets with caution in epileptic patients and patients with known or suspected CNS disorders that may predispose to seizures or lower the seizure threshold (for example, severe cerebral arteriosclerosis, previous history of convulsion, reduced cerebral blood flow, altered brain structure, or stroke), or in the presence of other risk factors that may predispose to seizures or lower the seizure threshold (for example, certain drug therapy, renal dysfunction). If seizures occur, discontinue ciprofloxacin extended-release tablets, and institute appropriate care [see Adverse Reactions (6.1) and Drug Interactions (7)].
5.5 Exacerbation of Myasthenia Gravis
Fluoroquinolones, including ciprofloxacin extended-release tablets, have neuromuscular blocking activity and may exacerbate muscle weakness in patients with myasthenia gravis. Postmarketing serious adverse reactions, including deaths and requirement for ventilatory support, have been associated with fluoroquinolone use in patients with myasthenia gravis. Avoid ciprofloxacin extended-release tablets in patients with known history of myasthenia gravis [see Adverse Reactions (6.3) and Patient Counseling Information (17)].
5.6 Other Serious and Sometimes Fatal Adverse Reactions
Other serious and sometimes fatal adverse reactions, some due to hypersensitivity, and some due to uncertain etiology, have been reported in patients receiving therapy with quinolones, including ciprofloxacin. These events may be severe and generally occur following the administration of multiple doses. Clinical manifestations may include one or more of the following:
- Fever, rash, or severe dermatologic reactions (for example, toxic epidermal necrolysis, Stevens-Johnson syndrome);
- Vasculitis; arthralgia; myalgia; serum sickness;
- Allergic pneumonitis;
- Interstitial nephritis; acute renal insufficiency or failure;
- Hepatitis; jaundice; acute hepatic necrosis or failure;
- Anemia, including hemolytic and aplastic; thrombocytopenia, including thrombotic thrombocytopenic purpura; leukopenia; agranulocytosis; pancytopenia; and/or other hematologic abnormalities.
Discontinue ciprofloxacin extended-release tablets immediately at the first appearance of a skin rash, jaundice, or any other sign of hypersensitivity and supportive measures instituted [see Adverse Reactions (6.1, 6.2)].
5.7 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions, some following the first dose, have been reported in patients receiving quinolone therapy, including ciprofloxacin extended-release tablets. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, tingling, pharyngeal or facial edema, dyspnea, urticaria, and itching. Only a few patients had a history of hypersensitivity reactions. Serious anaphylactic reactions require immediate emergency treatment with epinephrine and other resuscitation measures, including oxygen, intravenous fluids, intravenous antihistamines, corticosteroids, pressor amines, and airway management, including intubation, as indicated [see Contraindications (4.1), Adverse Reactions (6.1) and Patient Counseling Information (17)].
5.8 Hepatotoxicity
Cases of severe hepatotoxicity, including hepatic necrosis, life-threatening hepatic failure, and fatal events, have been reported with ciprofloxacin extended-release tablets. Acute liver injury is rapid in onset (range 1–39 days), and is often associated with hypersensitivity. The pattern of injury can be hepatocellular, cholestatic or mixed. Most patients with fatal outcomes were older than 55 years old. In the event of any signs and symptoms of hepatitis (such as anorexia, jaundice, dark urine, pruritus, or tender abdomen), discontinue treatment immediately.
There can be a temporary increase in transaminases, alkaline phosphatase, or cholestatic jaundice, especially in patients with previous liver damage, who are treated with ciprofloxacin extended-release tablets [see Adverse Reactions (6.1, 6.2, 6.3)].
5.9 Risk of Aortic Aneurysm and Dissection
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients. The cause for the increased risk has not been identified. In patients with a known aortic aneurysm or patients who are at greater risk for aortic aneurysms, reserve ciprofloxacin extended-release tablets for use only when there are no alternative antibacterial treatments available.
5.10 Serious Adverse Reactions with Concomitant Theophylline Use
Serious and fatal reactions have been reported in patients receiving concurrent administration of ciprofloxacin extended-release tablets and theophylline. These reactions have included cardiac arrest, seizure, status epilepticus, and respiratory failure. Instances of nausea, vomiting, tremor, irritability, or palpitation have also occurred.
Although similar serious adverse reactions have been reported in patients receiving theophylline alone, the possibility that these reactions may be potentiated by ciprofloxacin extended-release tablets cannot be eliminated. If concomitant use cannot be avoided, monitor serum levels of theophylline and adjust dosage as appropriate [see Drug Interactions (7)].
5.11 Clostridium Difficile-Associated Diarrhea
Clostridium difficile (C. difficile)-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including ciprofloxacin extended-release tablets, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and institute surgical evaluation as clinically indicated [see Adverse Reactions (6.2)].
5.12 Prolongation of the QT Interval
Some fluoroquinolones, including ciprofloxacin extended-release tablets have been associated with prolongation of the QT interval on the electrocardiogram and cases of arrhythmia. Cases of torsade de pointes have been reported during postmarketing surveillance in patients receiving fluoroquinolones, including ciprofloxacin extended-release tablets.
Avoid ciprofloxacin extended-release tablets in patients with known prolongation of the QT interval, risk factors for QT prolongation or torsade de pointes (for example, congenital long QT syndrome, uncorrected electrolyte imbalance, such as hypokalemia or hypomagnesemia and cardiac disease, such as heart failure, myocardial infarction, or bradycardia), and patients receiving Class IA antiarrhythmic agents (quinidine, procainamide), or Class III antiarrhythmic agents (amiodarone, sotalol), tricyclic antidepressants, macrolides, and antipsychotics. Elderly patients may also be more susceptible to drug-associated effects on the QT interval [see Adverse Reactions (6.2) and Use in Specific Populations (8.5)].
5.13 Musculoskeletal Disorders in Pediatric Patients and Arthropathic Effects in Animals
An increased incidence of adverse reactions compared to controls, including reactions related to joints and/or surrounding tissues, has been observed [see Adverse Reactions (6.2)].
In pre-clinical studies, oral administration of ciprofloxacin extended-release tablets caused lameness in immature dogs. Histopathological examination of the weight-bearing joints of these dogs revealed permanent lesions of the cartilage. Related quinolone-class drugs also produce erosions of cartilage of weight-bearing joints and other signs of arthropathy in immature animals of various species [see Use in Specific Populations (8.4) and Nonclinical Toxicology (13.2)].
5.14 Photosensitivity/Phototoxicity
Moderate to severe photosensitivity/phototoxicity reactions, the latter of which may manifest as exaggerated sunburn reactions (for example, burning, erythema, exudation, vesicles, blistering, edema) involving areas exposed to light (typically the face, “V” area of the neck, extensor surfaces of the forearms, dorsa of the hands), can be associated with the use of quinolones including ciprofloxacin extended-release tablets after sun or UV light exposure. Therefore, avoid excessive exposure to these sources of light. Discontinue ciprofloxacin extended-release tablets if phototoxicity occurs [see Adverse Reactions (6.1)].
5.15 Development of Drug Resistant Bacteria
Prescribing ciprofloxacin extended-release tablets in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.16 Potential Risks with Concomitant Use of Drugs Metabolized by Cytochrome P450 1A2 Enzymes
Ciprofloxacin is an inhibitor of the hepatic CYP1A2 enzyme pathway. Co-administration of ciprofloxacin and other drugs primarily metabolized by CYP1A2 (for example, theophylline, methylxanthines, caffeine, tizanidine, ropinirole, clozapine, olanzapine and zolpidem) results in increased plasma concentrations of the co-administered drug and could lead to clinically significant pharmacodynamic adverse reactions of the co-administered drug [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
5.17 Interference with Timely Diagnosis of Syphilis
Ciprofloxacin has not been shown to be effective in the treatment of syphilis. Antimicrobial agents used in high dose for short periods of time to treat gonorrhea may mask or delay the symptoms of incubating syphilis. Perform a serologic test for syphilis in all patients with gonorrhea at the time of diagnosis. Perform follow-up serologic test for syphilis three months after ciprofloxacin extended-release tablets treatment.
5.18 Crystalluria
Crystals of ciprofloxacin have been observed rarely in the urine of human subjects but more frequently in the urine of laboratory animals, which is usually alkaline [see Nonclinical Toxicology (13.2)]. Crystalluria related to ciprofloxacin has been reported only rarely in humans because human urine is usually acidic. Avoid alkalinity of the urine in patients receiving ciprofloxacin extended-release tablets. Hydrate patients well to prevent the formation of highly concentrated urine [see Dosage and Administration (2.2)].
5.19 Blood Glucose Disturbances
Fluoroquinolones, including ciprofloxacin extended-release tablets, have been associated with disturbances of blood glucose, including symptomatic hyperglycemia and hypoglycemia, usually in diabetic patients receiving concomitant treatment with an oral hypoglycemic agent (e.g., glyburide) or with insulin. In these patients, careful monitoring of blood glucose is recommended. Severe cases of hypoglycemia resulting in coma or death have been reported. If a hypoglycemic reaction occurs in a patient being treated with ciprofloxacin extended-release tablets, discontinue ciprofloxacin extended-release tablets and initiate appropriate therapy immediately [see Adverse Reactions (6.1), Drug Interactions (7)].
6 ADVERSE REACTIONS
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling:
- Disabling and Potentially Irreversible Serious Adverse Reactions [see Warnings and Precautions (5.1)]
- Tendinitis and Tendon Rupture [see Warnings and Precautions (5.2)]
- Peripheral Neuropathy [see Warnings and Precautions (5.3)]
- Central Nervous System Effects [see Warnings and Precautions (5.4)]
- Exacerbation of Myasthenia Gravis [see Warnings and Precautions (5.5)]
- Other Serious and Sometimes Fatal Adverse Reactions [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Hepatotoxicity [see Warnings and Precautions (5.8)]
- Risk of Aortic Aneurysm and Dissection [see Warnings and Precautions (5.9)]
- Serious Adverse Reactions with Concomitant Theophylline Use [see Warnings and Precautions (5.10)]
- Clostridium Difficile-Associated Diarrhea [see Warnings and Precautions (5.11)]
- Prolongation of the QT Interval [see Warnings and Precautions (5.12)]
- Musculoskeletal Disorders in Pediatric Patients [see Warnings and Precautions (5.13)]
- Photosensitivity/Phototoxicity [see Warnings and Precautions (5.14)]
- Development of Drug Resistant Bacteria [see Warnings and Precautions (5.15)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical trials in patients with urinary tract infections enrolled 961 patients treated with 500 mg or 1000 mg ciprofloxacin extended-release tablets. The overall incidence, type and distribution of adverse reactions were similar in patients receiving both 500 mg and 1000 mg of ciprofloxacin extended-release tablets. The adverse reaction information from clinical studies does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
In the clinical trial of uncomplicated UTIs, ciprofloxacin extended-release tablets (500 mg once daily) in 444 patients was compared to ciprofloxacin immediate-release tablets (250 mg twice daily) in 447 patients for 3 days. Discontinuations due to adverse reactions thought to be drug-related occurred in 0.2% (1/444) of patients in the ciprofloxacin extended-release tablets arm and in 0% (0/447) of patients in the control arm.
In the clinical trial of cUTI and acute uncomplicated pyelonephritis (AUP) defined as infections occurring in premenopausal, non-pregnant women with no known urological abnormalities or comorbidities, ciprofloxacin extended-release tablets (1000 mg once daily) in 517 patients was compared to ciprofloxacin immediate-release tablets (500 mg twice daily) in 518 patients for 7 to 14 days. Discontinuations due to adverse reactions thought to be drug-related occurred in 3.1% (16/517) of patients in the ciprofloxacin extended-release tablets arm and in 2.3% (12/518) of patients in the control arm. The most common reasons for discontinuation in the ciprofloxacin extended-release tablets arm were nausea/vomiting (4 patients) and dizziness (3 patients). In the control arm the most common reason for discontinuation was nausea/vomiting (3 patients).
In these clinical trials, the following events occurred in ≥ 2% of all ciprofloxacin extended-release tablets patients: nausea (4%), headache (3%), dizziness (2%), diarrhea (2%), vomiting (2%) and vaginal moniliasis (2%).
Adverse reactions, judged by investigators to be at least possibly drug-related, occurring in greater than or equal to 1% of all ciprofloxacin extended-release tablets treated patients were: nausea (3%), diarrhea (2%), headache (1%), dyspepsia (1%), dizziness (1%), and vaginal moniliasis (1%). Vomiting (1%) occurred in the 1000 mg group.
|
System Organ Class |
Adverse Reactions |
|
Body as a Whole |
Abdominal pain Asthenia Malaise |
|
Cardiovascular |
Bradycardia Migraine Syncope |
|
Central Nervous System |
Abnormal dreams Convulsive seizures (including status epilepticus) Depersonalization Depression (potentially culminating in self-injurious behavior, such as suicidal ideations/thoughts and attempted or completed suicide) Hypertonia Incoordination Insomnia Somnolence Tremor Vertigo |
|
Gastrointestinal |
Constipation Dry mouth Flatulence Thirst |
|
Hepatobiliary Disorders |
Liver function tests abnormal |
|
Investigations |
Prothrombin decrease |
|
Metabolic |
Hyperglycemia Hypoglycemia |
|
Psychiatric Disorders |
Anorexia |
|
Skin/Hypersensitivity |
Dry skin Photosensitivity/phototoxicity reactions Pruritus Rash Skin disorder Urticarial Vesiculobullous rash |
|
Special Senses |
Diplopia Taste perversion |
|
Urogenital |
Dysmenorrhea Hematuria Kidney function abnormal Vaginitis |
6.2 Postmarketing Experience
The following adverse reactions have been reported from worldwide marketing experience with fluoroquinolones, including ciprofloxacin extended-release tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure (Table 3).
|
System Organ Class |
Adverse Reactions |
|
Cardiovascular |
QT prolongation Torsade de Pointes Vasculitis and ventricular arrhythmia |
|
Central Nervous System |
Hypertonia Myasthenia Exacerbation of myasthenia gravis Peripheral neuropathy Polyneuropathy Twitching |
|
Eye Disorders |
Nystagmus |
|
Gastrointestinal |
Pseudomembranous colitis |
|
Hemic/Lymphatic |
Pancytopenia (life threatening or fatal outcome) Methemoglobinemia |
|
Hepatobiliary |
Hepatic failure (including fatal cases) |
|
Infections and Infestations |
Candidiasis (oral, gastrointestinal, vaginal) |
|
Investigations |
Prothrombin time prolongation or decrease Cholesterol elevation (serum) Potassium elevation (serum) |
|
Musculoskeletal |
Myalgia Myoclonus Tendinitis Tendon rupture |
|
Psychiatric Disorders |
Agitation Confusion Delirium Psychosis (toxic) |
|
Skin/Hypersensitivity |
Acute generalized exanthematous pustulosis (AGEP) Fixed eruption Serum sickness-like reaction |
|
Special Senses |
Anosmia Hyperesthesia Hypesthesia Taste loss |
6.3 Adverse Laboratory Changes
Changes in laboratory parameters while on ciprofloxacin are listed below:
Hepatic: Elevations of ALT (SGPT), AST (SGOT), alkaline phosphatase, LDH, serum bilirubin.
Hematologic: Eosinophilia, leukopenia, decreased blood platelets, elevated blood platelets, pancytopenia.
Renal: Elevations of serum creatinine, BUN, crystalluria, cylindruria, and hematuria have been reported.
Other changes occurring were: elevation of serum gammaglutamyl transferase, elevation of serum amylase, reduction in blood glucose, elevated uric acid, decrease in hemoglobin, anemia, bleeding diathesis, increase in blood monocytes, and leukocytosis.
7 DRUG INTERACTIONS
Ciprofloxacin is an inhibitor of human cytochrome P450 1A2 (CYP1A2) mediated metabolism. Co-administration of ciprofloxacin extended-release tablets with other drugs primarily metabolized by CYP1A2 results in increased plasma concentrations of these drugs and could lead to clinically significant adverse events of the co-administered drug.
|
Drugs That are Affected by Ciprofloxacin |
||
|
Drug(s) |
Recommendation |
Comments |
|
Tizanidine |
Contraindicated |
Concomitant administration of tizanidine and ciprofloxacin extended-release tablets is contraindicated due to the potentiation of hypotensive and sedative effects of tizanidine [see Contraindications (4.2)]. |
|
Theophylline |
Avoid Use (Plasma Exposure Likely to be Increased and Prolonged) |
Concurrent administration of ciprofloxacin extended-release tablets with theophylline may result in increased risk of a patient developing central nervous system (CNS) or other adverse reactions. If concomitant use cannot be avoided, monitor serum levels of theophylline and adjust dosage as appropriate [see Warnings and Precautions (5.10)]. |
|
Drugs Known to Prolong QT Interval |
Avoid Use |
Ciprofloxacin extended-release tablets may further prolong the QT interval in patients receiving drugs known to prolong the QT interval (for example, class IA or III antiarrhythmics, tricyclic antidepressants, macrolides, antipsychotics) [see Warnings and Precautions (5.12) and Use in Specific Populations (8.5)]. |
|
Oral antidiabetic drugs |
Use with caution Glucose-lowering effect potentiated |
Hypoglycemia sometimes severe has been reported when ciprofloxacin extended-release tablets and oral antidiabetic agents, mainly sulfonylureas (for example, glyburide, glimepiride), were co-administered, presumably by intensifying the action of the oral antidiabetic agent. Fatalities have been reported. Monitor blood glucose when ciprofloxacin extended-release tablets are co-administered with oral antidiabetic drugs [see Adverse Reactions (6.1)]. |
|
Phenytoin |
Use with caution Altered serum levels of phenytoin (increased and decreased) |
To avoid the loss of seizure control associated with decreased phenytoin levels and to prevent phenytoin overdose-related adverse reactions upon ciprofloxacin extended-release tablets discontinuation in patients receiving both agents, monitor phenytoin therapy, including phenytoin serum concentration during and shortly after co-administration of ciprofloxacin extended-release tablets with phenytoin. |
|
Cyclosporine |
Use with caution (transient elevations in serum creatinine) |
Monitor renal function (in particular serum creatinine) when ciprofloxacin extended-release tablets are co-administered with cyclosporine. |
|
Anti-coagulant drugs |
Use with caution (Increase in anticoagulant effect) |
The risk may vary with the underlying infection, age and general status of the patient so that the contribution of ciprofloxacin extended-release tablets to the increase in INR (international normalized ratio) is difficult to assess. Monitor prothrombin time and INR frequently during and shortly after co-administration of ciprofloxacin extended-release tablets with an oral anti-coagulant (for example, warfarin). |
|
Methotrexate |
Use with caution Inhibition of methotrexate renal tubular transport potentially leading to increased methotrexate plasma levels |
Potential increase in the risk of methotrexate associated toxic reactions. Therefore, carefully monitor patients under methotrexate therapy when concomitant ciprofloxacin extended-release tablets therapy is indicated. |
|
Ropinirole |
Use with caution |
Monitoring for ropinirole-related adverse reactions and appropriate dose adjustment of ropinirole is recommended during and shortly after co-administration with ciprofloxacin extended-release tablets [see Warnings and Precautions (5.16)]. |
|
Clozapine |
Use with caution |
Careful monitoring of clozapine associated adverse reactions and appropriate adjustment of clozapine dosage during and shortly after co-administration with ciprofloxacin extended-release tablets is advised. |
|
NSAIDs |
Use with caution |
Non-steroidal anti-inflammatory drugs (but not acetyl salicylic acid) in combination of very high doses of quinolones have been shown to provoke convulsions in pre-clinical studies and in postmarketing. |
|
Sildenafil |
Use with caution Two-fold increase in exposure |
Monitor for sildenafil toxicity [see Clinical Pharmacology (12.3)]. |
|
Duloxetine |
Avoid Use Five-fold increase in duloxetine exposure |
If unavoidable monitor, for duloxetine toxicity |
|
Caffeine/Xanthine Derivatives |
Use with caution Reduced clearance resulting in elevated levels and prolongation of serum half-life |
Ciprofloxacin extended-release tablets inhibit the formation of paraxanthine after caffeine administration (or pentoxifylline containing products). Monitor for xanthine toxicity and adjust dose as necessary. |
|
Zolpidem |
Avoid Use |
Co-administration with ciprofloxacin may increase blood levels of zolpidem, concurrent use is not recommended |
|
Drug(s) Affecting Pharmacokinetics of Ciprofloxacin Extended-Release Tablets |
||
|
Antacids, Sucralfate, Multivitamins and Other Products Containing Multivalent Cations (magnesium/aluminum antacids; polymeric phosphate binders (for example, sevelamer, lanthanum carbonate); sucralfate; Videx (didanosine) chewable/buffered tablets or pediatric powder; other highly buffered drugs; or products containing calcium, iron, or zinc and dairy products) |
Ciprofloxacin extended-release tablets should be taken at least two hours before or six hours after Multivalent cation-containing products administration [see Dosage and Administration (2)]. |
Decrease ciprofloxacin extended-release tablets absorption, resulting in lower serum and urine levels considerably lower than desired for concurrent administration of these agents with ciprofloxacin extended-release. |
|
Probenecid |
Use with caution (interferes with renal tubular secretion of ciprofloxacin extended-release tablets and increases ciprofloxacin extended-release tablets serum levels) |
Potentiation of ciprofloxacin extended-release tablets toxicity may occur. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. Ciprofloxacin extended-release tablets should not be used during pregnancy unless the potential benefit justifies the potential risk to both fetus and mother. An expert review of published data on experiences with ciprofloxacin use during pregnancy by TERIS–the Teratogen Information System concluded that therapeutic doses during pregnancy are unlikely to pose a substantial teratogenic risk (quantity and quality of data = fair), but the data are insufficient to state that there is no risk.
A controlled prospective observational study followed 200 women exposed to fluoroquinolones (52.5% exposed to ciprofloxacin and 68% first trimester exposures) during gestation. In utero exposure to fluoroquinolones during embryogenesis was not associated with increased risk of major malformations. The reported rates of major congenital malformations were 2.2% for the fluoroquinolone group and 2.6% for the control group (background incidence of major malformations is 1–5%). Rates of spontaneous abortions, prematurity and low birth weight did not differ between the groups and there were no clinically significant musculoskeletal dysfunctions up to one year of age in the ciprofloxacin exposed children.
Another prospective follow-up study reported on 549 pregnancies with fluoroquinolone exposure (93% first trimester exposures). There were 70 ciprofloxacin exposures, all within the first trimester. The malformation rates among live-born babies exposed to ciprofloxacin and to fluoroquinolones overall were both within background incidence ranges. No specific patterns of congenital abnormalities were found. The study did not reveal any clear adverse reactions due to in utero exposure to ciprofloxacin.
No differences in the rates of prematurity, spontaneous abortions, or birth weight were seen in women exposed to ciprofloxacin during pregnancy. However, these small postmarketing epidemiology studies, of which most experience is from short term, first trimester exposure, are insufficient to evaluate the risk for the less common defects or to permit reliable and definitive conclusions regarding the safety of ciprofloxacin in pregnant women and their developing fetuses.
Reproduction studies have been performed in rats and mice using oral doses up to 100 mg/kg (0.6 and 0.3 times the maximum daily human dose of 1000 mg based upon body surface area, respectively) and have revealed no evidence of harm to the fetus due to ciprofloxacin. In rabbits, oral ciprofloxacin dose levels of 30 and 100 mg/kg (approximately 0.4- and 1.3-times the highest recommended therapeutic dose based upon body surface area) produced gastrointestinal toxicity resulting in maternal weight loss and an increased incidence of abortion, but no teratogenicity was observed at either dose level. After intravenous administration of doses up to 20 mg/kg (approximately 0.3-times the highest recommended therapeutic dose based upon body surface area), no maternal toxicity was produced in the rabbit, and no embryotoxicity or teratogenicity was observed.
8.3 Nursing Mothers
Ciprofloxacin is excreted in human milk. The amount of ciprofloxacin absorbed by the nursing infant is unknown. Because of the potential risk of serious adverse reactions (including articular damage) in infants nursing from mothers taking ciprofloxacin extended-release tablets, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Safety and effectiveness of ciprofloxacin extended-release tablets in pediatric patients and adolescents less than 18 years of age have not been established. Ciprofloxacin causes arthropathy (arthralgia, arthritis) in juvenile animals [see Nonclinical Toxicology (13.2)]. Ciprofloxacin extended-release tablets are not indicated for pediatric patients [see Indications and Usage (1.3)].
8.5 Geriatric Use
Geriatric patients are at increased risk for developing severe tendon disorders including tendon rupture when being treated with a fluoroquinolone such as ciprofloxacin extended-release tablets. This risk is further increased in patients receiving concomitant corticosteroid therapy. Tendinitis or tendon rupture can involve the Achilles, hand, shoulder, or other tendon sites and can occur during or after completion of therapy; cases occurring up to several months after fluoroquinolone treatment have been reported. Caution should be used when prescribing ciprofloxacin extended-release tablets to elderly patients especially those on corticosteroids. Patients should be informed of this potential side effect and advised to discontinue ciprofloxacin extended-release tablets and contact their healthcare provider if any symptoms of tendinitis or tendon rupture occur [see Boxed Warning, Warnings and Precautions (5.2), and Adverse Reactions (6.2)].
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients [see Warnings and Precautions (5.8)].
In a large, prospective, randomized ciprofloxacin extended-release tablets clinical trial in cUTI, 49% (509/1035) of the patients were 65 and over, while 30% (308/1035) were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals on any drug therapy cannot be ruled out. Ciprofloxacin is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. No alteration of dosage is necessary for patients greater than 65 years of age with normal renal function. However, since some older individuals experience reduced renal function by virtue of their advanced age, care should be taken in dose selection for elderly patients, and renal function monitoring may be useful in these patients [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients [see Warnings and Precautions (5.8)].
In general, elderly patients may be more susceptible to drug-associated effects on the QT interval. Therefore, precaution should be taken when using ciprofloxacin extended-release tablets with concomitant drugs that can result in prolongation of the QT interval (for example, class IA or class III antiarrhythmics) or in patients with risk factors for torsade de pointes (for example, known QT prolongation, uncorrected hypokalemia) [see Warnings and Precautions (5.12)].
8.6 Renal Impairment
Ciprofloxacin is eliminated primarily by renal excretion; however, the drug is also metabolized and partially cleared through the biliary system of the liver and through the intestine. These alternate pathways of drug elimination appear to compensate for the reduced renal excretion in patients with renal impairment. No dosage adjustment is required for patients with uncomplicated UTIs receiving 500 mg ciprofloxacin extended-release tablets. Dosing in children (less than 18 years of age) with impaired renal function has not been studied [see Clinical Pharmacology (12.3)].
10 OVERDOSAGE
In the event of acute overdosage, reversible renal toxicity has been reported in some cases. Empty the stomach by inducing vomiting or by gastric lavage. Observe the patient carefully and give supportive treatment, including monitoring of renal function, urinary pH and acidify, if required, to prevent crystalluria and administration of magnesium, aluminum or calcium containing antacids, which can reduce the absorption of ciprofloxacin. Adequate hydration must be maintained. Only a small amount of ciprofloxacin (less than 10%) is removed from the body after hemodialysis or peritoneal dialysis.
11 DESCRIPTION
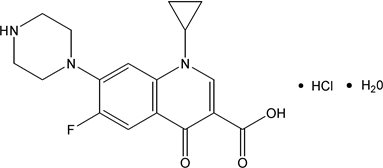
Ciprofloxacin* extended-release tablets, USP contain ciprofloxacin, a synthetic antimicrobial agent for oral administration. Ciprofloxacin extended-release tablets are coated, bilayer tablets consisting of an immediate-release layer and an erosion-matrix type controlled-release layer. The tablets contain a combination of two types of ciprofloxacin drug substance, ciprofloxacin hydrochloride, USP and ciprofloxacin, USP. Ciprofloxacin hydrochloride is 1-Cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinolinecarboxylic acid. It is provided as the monohydrate. The molecular formula is C17H18FN3O3 HCl H2O and its molecular weight is 385.8. The drug substance is faintly yellowish to light yellow crystals. The chemical structure is as follows:
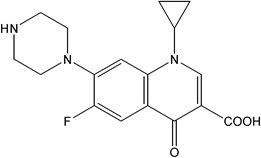
Ciprofloxacin is 1-Cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinolinecarboxylic acid, monohydrate. As the anhydrous form, its molecular formula is C17H18FN3O3 and its molecular weight is 331.3. It is a white to pale yellow crystalline powder and its chemical structure is as follows:
Ciprofloxacin extended-release tablets, USP are available in 500 mg and 1000 mg (ciprofloxacin equivalent) tablet strengths. Ciprofloxacin extended-release tablets are orange, film-coated, modified capsule shaped tablets. Each ciprofloxacin extended-release 500 mg tablet contains 500 mg of ciprofloxacin as ciprofloxacin hydrochloride, USP (287.5 mg, calculated as ciprofloxacin on the dried basis) and ciprofloxacin, USP (212.6 mg, calculated on the dried basis). Each ciprofloxacin extended-release 1000 mg tablet contains 1000 mg of ciprofloxacin as ciprofloxacin hydrochloride, USP (574.9 mg, calculated as ciprofloxacin on the dried basis) and ciprofloxacin, USP (425.2 mg, calculated on the dried basis). The inactive ingredients are carnauba wax, colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate (anhydrous), FD&C Yellow No. 6 Aluminum Lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, pregelatinized starch (corn), sodium lauryl sulfate, stearic acid, succinic acid, talc and titanium dioxide.
Meets USP Dissolution Test 2.
* as ciprofloxacin and ciprofloxacin hydrochloride
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ciprofloxacin is a member of the fluoroquinolone class of antibacterial agents [see Microbiology (12.4)].
12.3 Pharmacokinetics
Absorption
Ciprofloxacin extended-release tablets are formulated to release drug at a slower rate compared to immediate-release tablets. Approximately 35% of the dose is contained within an immediate-release component, while the remaining 65% is contained in a slow-release matrix.
Maximum plasma ciprofloxacin concentrations are attained between 1 and 4 hours after dosing with ciprofloxacin extended-release tablets. In comparison to the 250 mg and 500 mg ciprofloxacin immediate-release twice a day (BID) treatment, the Cmax of ciprofloxacin extended-release tablets 500 mg and 1000 mg once daily are higher than the corresponding BID doses, while the AUCs over 24 hours are equivalent.
The following table compares the pharmacokinetic parameters obtained at steady state for these four treatment regimens (500 mg once a day (QD) ciprofloxacin extended-release tablets versus 250 mg BID ciprofloxacin immediate-release tablets and 1000 mg QD ciprofloxacin extended-release tablets versus 500 mg BID ciprofloxacin immediate-release).
|
|
||||
|
Cmax
|
AUC0–24h (mgh/L) |
T1/2 (hr) |
Tmax (hr)* |
|
|
Ciprofloxacin extended-release tablets 500 mg QD Ciprofloxacin 250 mg BID |
1.59 ± 0.43 1.14 ± 0.23 |
7.97 ± 1.87 8.25 ± 2.15 |
6.6 ± 1.4 4.8 ± 0.6 |
1.5 (1 – 2.5) 1 (0.5 – 2.5) |
|
Ciprofloxacin extended-release tablets 1000 mg QD Ciprofloxacin 500 mg BID |
3.11 ± 1.08 2.06 ± 0.41 |
16.83 ± 5.65 17.04 ± 4.79 |
6.31 ± 0.72 5.66 ± 0.89 |
2 (1 – 4) 2 (0.5 – 3.5) |
Results of the pharmacokinetic studies demonstrate that ciprofloxacin extended-release tablets may be administered with or without food (for example, high-fat and low-fat meals or under fasted conditions).
Distribution
The volume of distribution calculated for intravenous ciprofloxacin is approximately 2.1–2.7 L/kg. Studies with the oral and intravenous forms of ciprofloxacin have demonstrated penetration of ciprofloxacin into a variety of tissues. The binding of ciprofloxacin to serum proteins is 20% to 40%, which is not likely to be high enough to cause significant protein binding interactions with other drugs. Following administration of a single dose of ciprofloxacin extended-release tablets, ciprofloxacin concentrations in urine collected up to 4 hours after dosing averaged over 300 mg/L for both the 500 mg and 1000 mg tablets; in urine excreted from 12 to 24 hours after dosing, ciprofloxacin concentration averaged 27 mg/L for the 500 mg tablet, and 58 mg/L for the 1000 mg tablet.
Metabolism
Four metabolites of ciprofloxacin were identified in human urine. The metabolites have antimicrobial activity, but are less active than unchanged ciprofloxacin. The primary metabolites are oxociprofloxacin (M3) and sulfociprofloxacin (M2), each accounting for roughly 3% to 8% of the total dose. Other minor metabolites are desethylene ciprofloxacin (M1), and formylciprofloxacin (M4). The relative proportion of drug and metabolite in serum corresponds to the composition found in urine. Excretion of these metabolites was essentially complete by 24 hours after dosing. Ciprofloxacin is an inhibitor of CYP1A2 mediated metabolism. Co-administration of ciprofloxacin with other drugs primarily metabolized by CYP1A2 results in increased plasma concentrations of these drugs and could lead to clinically significant adverse events of the co-administered drug [see Contraindications (4.2), Warnings and Precautions (5.10, 5.16) and Drug Interactions (7)].
Elimination
The elimination kinetics of ciprofloxacin are similar for the immediate-release and the ciprofloxacin extended-release tablet. In studies comparing the ciprofloxacin extended-release tablets and immediate-release ciprofloxacin, approximately 35% of an orally administered dose was excreted in the urine as unchanged drug for both formulations. The urinary excretion of ciprofloxacin is virtually complete within 24 hours after dosing. The renal clearance of ciprofloxacin, which is approximately 300 mL/minute, exceeds the normal glomerular filtration rate of 120 mL/minute. Thus, active tubular secretion would seem to play a significant role in its elimination. Co-administration of probenecid with immediate-release ciprofloxacin results in about a 50% reduction in the ciprofloxacin renal clearance and a 50% increase in its concentration in the systemic circulation. Although bile concentrations of ciprofloxacin are several fold higher than serum concentrations after oral dosing with the immediate-release tablet, only a small amount of the dose administered is recovered from the bile as unchanged drug. An additional 1 to 2% of the dose is recovered from the bile in the form of metabolites. Approximately 20 to 35% of an oral dose of immediate-release ciprofloxacin is recovered from the feces within 5 days after dosing. This may arise from either biliary clearance or transintestinal elimination.
Specific Populations
Elderly
Pharmacokinetic studies of the immediate-release oral tablet (single dose) and intravenous (single and multiple dose) forms of ciprofloxacin indicate that plasma concentrations of ciprofloxacin are higher in elderly subjects (> 65 years) as compared to young adults. Cmax is increased 16 to 40%, and mean AUC is increased approximately 30%, which can be at least partially attributed to decreased renal clearance in the elderly. Elimination half-life is only slightly (~20%) prolonged in the elderly. These differences are not considered clinically significant [see Use in Specific Populations (8.5)].
Renal Impairment
In patients with reduced renal function, the half-life of ciprofloxacin is slightly prolonged. No dose adjustment is required for patients with uncomplicated UTIs receiving 500 mg ciprofloxacin extended-release tablets. For cUTI and AUP, where 1000 mg is the appropriate dose, the dosage of ciprofloxacin extended-release tablets should be reduced to ciprofloxacin extended-release tablets 500 mg q24h in patients with creatinine clearance equal to or below 30 mL/min [see Dosage and Administration (2.3)].
Hepatic Impairment
In preliminary studies in patients with stable chronic cirrhosis, no significant changes in ciprofloxacin pharmacokinetics have been observed. The kinetics of ciprofloxacin in patients with acute hepatic insufficiency, however, have not been fully elucidated [see Use in Specific Populations (8.7)].
Drug-Drug Interactions
Antacids
Concurrent administration of antacids containing magnesium hydroxide or aluminum hydroxide may reduce the bioavailability of ciprofloxacin by as much as 90% [see Dosage and Administration (2.2) and Drug Interactions (7)].
Histamine H2-receptor Antagonists
Histamine H2-receptor antagonists appear to have no significant effect on the bioavailability of ciprofloxacin.
Metronidazole
The serum concentrations of ciprofloxacin and metronidazole were not altered when these two drugs were given concomitantly.
Tizanidine
In a pharmacokinetic study, systemic exposure of tizanidine (4 mg single dose) was significantly increased (Cmax 7-fold, AUC 10-fold) when the drug was given concomitantly with ciprofloxacin (500 mg twice a day for 3 days). Concomitant administration of tizanidine and ciprofloxacin extended-release tablets is contraindicated due to the potentiation of hypotensive and sedative effects of tizanidine [see Contraindications (4.2)].
Ropinirole
In a study conducted in 12 patients with Parkinson’s disease who were administered 6 mg ropinirole once daily with 500 mg ciprofloxacin twice-daily, the mean Cmax and mean AUC of ropinirole were increased by 60% and 84%, respectively. Monitoring for ropinirole-related adverse reactions and appropriate dose adjustment of ropinirole is recommended during and shortly after co-administration with ciprofloxacin extended-release tablets [see Warnings and Precautions (5.16)].
Clozapine
Following concomitant administration of 250 mg ciprofloxacin with 304 mg clozapine for 7 days, serum concentrations of clozapine and N-desmethylclozapine were increased by 29% and 31%, respectively. Careful monitoring of clozapine associated adverse reactions and appropriate adjustment of clozapine dosage during and shortly after co-administration with ciprofloxacin extended-release tablets is advised.
Sildenafil
Following concomitant administration of a single oral dose of 50 mg sildenafil with 500 mg ciprofloxacin to healthy subjects, the mean Cmax and mean AUC of sildenafil were both increased approximately two-fold. Use sildenafil with caution when co-administered with ciprofloxacin extended-release tablets due to the expected two-fold increase in the exposure of sildenafil upon co-administration of ciprofloxacin [see Drug Interactions (7)].
Duloxetine
In clinical studies it was demonstrated that concomitant use of duloxetine with strong inhibitors of the CYP450 1A2 isozyme such as fluvoxamine, may result in a 5-fold increase in mean AUC and a 2.5-fold increase in mean Cmax of duloxetine.
Lidocaine
In a study conducted in 9 healthy volunteers, concomitant use of 1.5 mg/kg IV lidocaine with 500 mg ciprofloxacin twice daily resulted in an increase of lidocaine Cmax and AUC by 12% and 26%, respectively. Although lidocaine treatment was well tolerated at this elevated exposure, a possible interaction with ciprofloxacin extended-release tablets and an increase in adverse reactions related to lidocaine may occur upon concomitant administration.
Metoclopramide
Metoclopramide significantly accelerates the absorption of oral ciprofloxacin resulting in a shorter time to reach maximum plasma concentrations. No significant effect was observed on the bioavailability of ciprofloxacin.
Omeprazole
When ciprofloxacin extended-release tablets were administered as a single 1000 mg dose concomitantly with omeprazole (40 mg once daily for three days) to 18 healthy volunteers, the mean AUC and Cmax of ciprofloxacin were reduced by 20% and 23%, respectively. The clinical significance of this interaction has not been determined.
12.4 Microbiology
Mechanism of Action
The bactericidal action of ciprofloxacin results from inhibition of the enzymes topoisomerase II (DNA gyrase) and topoisomerase IV (both Type II topoisomerases), which are required for bacterial DNA replication, transcription, repair, and recombination.
Mechanism of Resistance
The mechanism of action of fluoroquinolones, including ciprofloxacin, is different from that of other antimicrobial agents such as beta-lactams, macrolides, tetracyclines, or aminoglycosides; therefore, microorganisms resistant to these classes of drugs may be susceptible to ciprofloxacin. Resistance to fluoroquinolones occurs primarily by either mutations in the DNA gyrases, decreased outer membrane permeability, or drug efflux. In vitro resistance to ciprofloxacin develops slowly by multiple step mutations. Resistance to ciprofloxacin due to spontaneous mutations occurs at a general frequency of between < 10-9 to 1x10-6.
Cross Resistance
There is no known cross-resistance between ciprofloxacin and other classes of antimicrobials.
Ciprofloxacin has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections ciprofloxacin [see Indications and Usage (1)].
Gram-positive bacteria
Enterococcus faecalis
Staphylococcus saprophyticus
Gram-negative bacteria
Escherichia coli
Klebsiella pneumoniae
Proteus mirabilis
Pseudomonas aeruginosa
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for ciprofloxacin (≤ 1 mcg/mL). However, the efficacy of ciprofloxacin in treating clinical infections due to these bacteria has not been established in adequate and well-controlled clinical trials.
Gram-negative bacteria
Citrobacter koseri
Citrobacter freundii
Edwardsiella tarda
Enterobacter aerogenes
Enterobacter cloacae
Klebsiella oxytoca
Morganella morganii
Proteus vulgaris
Providencia rettgeri
Providencia stuartii
Serratia marcescens
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Eight in vitro mutagenicity tests have been conducted with ciprofloxacin, and the test results are listed below:
- Salmonella/Microsome Test (Negative)
- E. coli DNA Repair Assay (Negative)
- Mouse Lymphoma Cell Forward Mutation Assay (Positive)
- Chinese Hamster V79 Cell HGPRT Test (Negative)
- Syrian Hamster Embryo Cell Transformation Assay (Negative)
- Saccharomyces cerevisiae Point Mutation Assay (Negative)
- Saccharomyces cerevisiae Mitotic Crossover and Gene Conversion Assay (Negative)
- Rat Hepatocyte DNA Repair Assay (Positive)
Thus, 2 of the 8 tests were positive, but results of the following 3 in vivo test systems gave negative results:
- Rat Hepatocyte DNA Repair Assay
- Micronucleus Test (Mice)
- Dominant Lethal Test (Mice)
Ciprofloxacin was not carcinogenic or tumorigenic in 2-year carcinogenicity studies with rats and mice at daily oral dose levels of 250 mg/kg and 750 mg/kg, respectively (approximately 2 and 3-fold greater than the 1000 mg daily human dose based upon body surface area).
Results from photo co-carcinogenicity testing indicate that ciprofloxacin does not reduce the time to appearance of UV-induced skin tumors as compared to vehicle control. Hairless (Skh-1) mice were exposed to UVA light for 3.5 hours five times every two weeks for up to 78 weeks while concurrently being administered ciprofloxacin. The time to development of the first skin tumors was 50 weeks in mice treated concomitantly with UVA and ciprofloxacin (mouse dose approximately equal to the maximum recommended daily human dose of 1000 mg based upon body surface area), as opposed to 34 weeks when animals were treated with both UVA and vehicle. The times to development of skin tumors ranged from 16–32 weeks in mice treated concomitantly with UVA and other quinolones.
In this model, mice treated with ciprofloxacin alone did not develop skin or systemic tumors. There are no data from similar models using pigmented mice and/or fully haired mice. The clinical significance of these findings to humans is unknown.
Fertility studies performed in rats at oral doses of ciprofloxacin up to 100 mg/kg (1 times the highest recommended daily human dose of 1000 mg based upon body surface area) revealed no evidence of impairment.
13.2 Animal Toxicology and/or Pharmacology
Ciprofloxacin and other quinolones have been shown to cause arthropathy in immature animals of most species tested [see Warnings and Precautions (5.13)]. Damage of weight bearing joints was observed in juvenile dogs and rats. In young beagles, 100 mg/kg ciprofloxacin, given daily for 4 weeks, caused degenerative articular changes of the knee joint. At 30 mg/kg, the effect on the joint was minimal. In a subsequent study in young beagle dogs, oral ciprofloxacin doses of 30 mg/kg and 90 mg/kg given daily for 2 weeks caused articular changes which were still observed by histopathology after a treatment-free period of 5 months. At 10 mg/kg no effects on joints were observed. This dose was also not associated with arthrotoxicity after an additional treatment-free period of 5 months. In another study, removal of weight bearing from the joint reduced the lesions but did not totally prevent them.
Crystalluria, sometimes associated with secondary nephropathy, occurs in laboratory animals dosed with ciprofloxacin. This is primarily related to the reduced solubility of ciprofloxacin under alkaline conditions, which predominate in the urine of test animals; in man, crystalluria is rare since human urine is typically acidic. In rhesus monkeys, crystalluria without nephropathy has been noted after single oral doses as low as 5 mg/kg (approximately 0.1-times the highest recommended therapeutic dose based upon body surface area. After 6 months of intravenous dosing at 10 mg/kg/day, no nephropathological changes were noted; however, nephropathy was observed after dosing at 20 mg/kg/day for the same duration (approximately 0.4 times the highest recommended therapeutic dose based upon body surface area).
In dogs, ciprofloxacin administered at 3 and 10 mg/kg by rapid infusion injection (15 sec.) produces hypotensive effects. These effects are considered to be related to histamine release because they are partially antagonized by pyrilamine, an antihistamine. In rhesus monkeys, rapid infusion injection also produces hypotension, but the effect in this species is inconsistent and less pronounced.
In mice, concomitant administration of nonsteroidal anti-inflammatory drugs such as phenylbutazone and indomethacin with quinolones has been reported to enhance the CNS stimulatory effect of quinolones.
Ocular toxicity seen with some related drugs has not been observed in ciprofloxacin-treated animals.
14 CLINICAL STUDIES
14.1 Uncomplicated Urinary Tract Infections (Acute Cystitis)
Ciprofloxacin extended-release tablets were evaluated for the treatment of uncomplicated UTIs (acute cystitis) in a randomized, double-blind, controlled clinical trial conducted in the U.S. This study compared ciprofloxacin extended-release tablets (500 mg once daily for three days) with ciprofloxacin immediate-release tablets (CIPRO® 250 mg two times a day (BID) for three days). Of the 905 patients enrolled, 452 were randomly assigned to the ciprofloxacin extended-release tablets treatment group and 453 were randomly assigned to the control group. The primary efficacy variable was bacteriologic eradication of the baseline organism(s) with no new infection or superinfection at test-of-cure (Day 4–11 Post-therapy).
The bacteriologic eradication and clinical success rates were similar between ciprofloxacin extended-release tablets and the control group. The eradication and clinical success rates and their corresponding 95% confidence intervals for the differences between rates (ciprofloxacin extended-release tablets minus control group) are given in Table 6:
|
|
||
|
Ciprofloxacin Extended-Release Tablets 500 mg QD x 3 Days |
Ciprofloxacin 250 mg BID x 3 Days |
|
|
Randomized Patients |
452 |
453 |
|
Per Protocol Patients* |
199 |
223 |
|
Bacteriologic Eradication at TOC (n/N)† |
188/199 (94.5%) |
209/223 (93.7%) |
|
CI [-3.5%, 5.1%] |
||
|
Bacteriologic Eradication (by organism) at TOC (n/N)‡ | ||
|
E. coli |
156/160 (97.5%) |
176/181 (97.2%) |
|
E. faecalis |
10/11 (90.9%) |
17/21 (81%) |
|
P. mirabilis |
11/12 (91.7%) |
7/7 (100%) |
|
S. saprophyticus |
6/7 (85.7%) |
9/9 (100%) |
|
Clinical Response at TOC (n/N)§ |
189/199 (95%) |
204/223 (91.5%) |
|
CI [-1.1%, 8.1%] |
||
14.2 Complicated Urinary Tract Infections and Acute Uncomplicated Pyelonephritis
Ciprofloxacin extended-release tablets were evaluated for the treatment of cUTI and acute uncomplicated pyelonephritis (AUP) in a randomized, double-blind, controlled clinical trial conducted in the U.S. and Canada. The study enrolled 1,042 patients (521 patients per treatment arm) and compared ciprofloxacin extended-release tablets (1000 mg once daily for 7 to 14 days) with immediate-release ciprofloxacin (500 mg BID for 7 to 14 days). The primary efficacy endpoint for this trial was bacteriologic eradication of the baseline organism(s) with no new infection or superinfection at 5 to 11 days post-therapy (test-of-cure or TOC) for the Per Protocol and Modified Intent-To-Treat (MITT) populations.
The Per Protocol population was defined as patients with a diagnosis of cUTI or AUP, a causative organism(s) at baseline present at ≥ 105 CFU/mL, no inclusion criteria violation, a valid test-of-cure urine culture within the TOC window, an organism susceptible to study drug, no premature discontinuation or loss to follow-up, and compliance with the dosage regimen (among other criteria). More patients in the ciprofloxacin extended-release tablets arm than in the control arm were excluded from the Per Protocol population and this should be considered in the interpretation of the study results. Reasons for exclusion with the greatest discrepancy between the two arms were no valid test-of-cure urine culture, an organism resistant to the study drug, and premature discontinuation due to adverse events.
An analysis of all patients with a causative organism(s) isolated at baseline and who received study medication, defined as the MITT population, included 342 patients in the ciprofloxacin extended-release tablets arm and 324 patients in the control arm. Patients with missing responses were counted as failures in this analysis. In the MITT analysis of cUTI patients, bacteriologic eradication was 160/271 (59%) versus 156/248 (62.9%) in ciprofloxacin extended-release tablets and control arm, respectively [97.5% CI* (-13.5%, 5.7%)]. Clinical cure was 184/271 (67.9%) for ciprofloxacin extended-release tablets and 182/248 (73.4%) for control arm, respectively [97.5% CI* (-14.4%, 3.5%)]. Bacterial eradication in the MITT analysis of patients with AUP at TOC was 47/71 (66.2%) and 58/76 (76.3%) for ciprofloxacin extended-release tablets and control arm, respectively [97.5% CI* (-26.8%, 6.5%)]. Clinical cure at TOC was 50/71 (70.4%) for ciprofloxacin extended-release tablets and 58/76 (76.3%) for the control arm [97.5% CI* (-22.0%, 10.4%)].
* confidence interval of the difference in rates (ciprofloxacin extended-release tablets minus control).
In the Per Protocol population, the differences between ciprofloxacin extended-release tablets and the control arm in bacteriologic eradication rates at the TOC visit were not consistent between AUP and cUTI patients. The bacteriologic eradication rate for cUTI patients was higher in the ciprofloxacin extended-release tablets arm than in the control arm. For AUP patients, the bacteriologic eradication rate was lower in the ciprofloxacin extended-release tablets arm than in the control arm. This inconsistency was not observed between the two treatment groups for clinical cure rates. Clinical cure rates were 96.1% (198/206) and 92.1% (211/229) for ciprofloxacin extended-release tablets and the control arm, respectively [difference: 4.0% with a two-sided 97.5% CI (-1.3%, 9.4%)].
The bacterial eradication and clinical cure rates by infection type for ciprofloxacin extended-release tablets and the control arm at the TOC visit and their corresponding 97.5% confidence intervals for the differences between rates (ciprofloxacin extended-release tablets minus control arm) are given in Table 7 for the Per Protocol population analysis.
|
|
||
|
Ciprofloxacin Extended-Release Tablets 1000 mg QD |
Ciprofloxacin 500 mg BID |
|
|
Randomized Patients |
521 |
521 |
|
Per Protocol Patients* |
206 |
229 |
|
cUTI Patients |
||
|
Bacteriologic Eradication at TOC (n/N)† |
148/166 (89.2%) |
144/177 (81.4%) |
|
CI [-0.7%, 16.3%] |
||
|
Bacteriologic Eradication (by organism) at TOC (n/N)‡ | ||
|
E. coli |
91/94 (96.8%) |
90/92 (97.8%) |
|
K. pneumoniae |
20/21 (95.2%) |
19/23 (82.6%) |
|
E. faecalis |
17/17 (100%) |
14/21 (66.7%) |
|
P. mirabilis |
11/12 (91.6%) |
10/10 (100%) |
|
P. aeruginosa |
3/3 (100%) |
3/3 (100%) |
|
Clinical Cure at TOC (n/N)§ |
159/166 (95.8%) |
161/177 (91.0%) |
|
CI [-1.1%, 10.8%] |
||
|
AUP Patients |
||
|
Bacteriologic Eradication at TOC (n/N)† |
35/40 (87.5%) |
51/52 (98.1%) |
|
CI [-34.8%, 6.2%] |
||
|
Bacteriologic Eradication of E. coli |
35/36 (97.2%) |
41/41 (100%) |
|
Clinical Cure at TOC (n/N)§ |
39/40 (97.5%) |
50/52 (96.2%) |
|
CI [-15.3%, 21.1%] |
||
Of the 166 cUTI patients treated with ciprofloxacin extended-release tablets, 148 (89%) had the causative organism(s) eradicated, 8 (5%) had persistence, 5 (3%) patients developed superinfections and 5 (3%) developed new infections. Of the 177 cUTI patients treated in the control arm, 144 (81%) had the causative organism(s) eradicated, 16 (9%) patients had persistence, 3 (2%) developed superinfections and 14 (8%) developed new infections. Of the 40 patients with AUP treated with ciprofloxacin extended-release tablets, 35 (87.5%) had the causative organism(s) eradicated, 2 (5%) patients had persistence and 3 (7.5%) developed new infections. Of the 5 ciprofloxacin extended-release tablets AUP patients without eradication at TOC, 4 were considered clinical cures and did not receive alternative antibiotic therapy. Of the 52 patients with AUP treated in the control arm, 51 (98%) had the causative organism(s) eradicated. One patient (2%) had persistence.
16 HOW SUPPLIED/STORAGE AND HANDLING
Ciprofloxacin Extended-Release Tablets, USP are available containing ciprofloxacin, USP and ciprofloxacin hydrochloride, USP equivalent to 500 mg or 1000 mg of ciprofloxacin.
The 500 mg tablets are orange, film-coated, modified capsule shaped, unscored tablets debossed with M 1743 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 0378-1743-89
bottles of 50 tablets
The 1000 mg tablets are orange, film-coated, modified capsule shaped, unscored tablets debossed with M 1745 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 0378-1745-89
bottles of 50 tablets
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Serious Adverse Reactions: Advise patients to stop taking ciprofloxacin extended-release tablets if they experience an adverse reaction and to call their healthcare provider for advice on completing the full course of treatment with another antibacterial drug.
Inform patients of the following serious adverse reactions that have been associated with ciprofloxacin extended-release tablets or other fluoroquinolone use:
- Disabling and potentially irreversible serious adverse reactions that may occur together: Inform patients that disabling and potentially irreversible serious adverse reactions, including tendinitis and tendon rupture, peripheral neuropathies, and central nervous system effects, have been associated with use of ciprofloxacin extended-release tablets and may occur together in the same patient. Inform patients to stop taking ciprofloxacin extended-release tablets immediately if they experience an adverse reaction and to call their healthcare provider.
- Tendinitis and Tendon Rupture: Instruct patients to contact their healthcare provider if they experience pain, swelling, or inflammation of a tendon, or weakness or inability to use one of their joints; rest and refrain from exercise; and discontinue ciprofloxacin extended-release tablets treatment. Symptoms may be irreversible. The risk of severe tendon disorder with fluoroquinolones is higher in older patients usually over 60 years of age, in patients taking corticosteroid drugs, and in patients with kidney, heart or lung transplants.
- Peripheral Neuropathies: Inform patients that peripheral neuropathies have been associated with ciprofloxacin use, symptoms may occur soon after initiation of therapy and may be irreversible. If symptoms of peripheral neuropathy including pain, burning, tingling, numbness and/or weakness develop, immediately discontinue ciprofloxacin extended-release tablets and tell them to contact their physician.
- Central Nervous System Effects (for example, convulsions, dizziness, lightheadedness, increased intracranial pressure): Inform patients that convulsions have been reported in patients receiving fluoroquinolones, including ciprofloxacin. Instruct patients to notify their physician before taking this drug if they have a history of convulsions. Inform patients that they should know how they react to ciprofloxacin extended-release tablets before they operate an automobile or machinery or engage in other activities requiring mental alertness and coordination. Instruct patients to notify their physician if persistent headache with or without blurred vision occurs.
- Exacerbation of Myasthenia Gravis: Instruct patients to inform their physician of any history of myasthenia gravis. Instruct patients to notify their physician if they experience any symptoms of muscle weakness, including respiratory difficulties.
- Hypersensitivity Reactions: Inform patients that ciprofloxacin can cause hypersensitivity reactions, even following a single dose, and to discontinue the drug at the first sign of a skin rash, hives or other skin reactions, a rapid heartbeat, difficulty in swallowing or breathing, any swelling suggesting angioedema (for example, swelling of the lips, tongue, face, tightness of the throat, hoarseness), or other symptoms of an allergic reaction.
- Hepatotoxicity: Inform patients that severe hepatotoxicity (including acute hepatitis and fatal events) has been reported in patients taking ciprofloxacin extended-release tablets. Instruct patients to inform their physician if they experience any signs or symptoms of liver injury including: loss of appetite, nausea, vomiting, fever, weakness, tiredness, right upper quadrant tenderness, itching, yellowing of the skin and eyes, light colored bowel movements or dark colored urine.
- Aortic Aneurysm and Dissection: Inform patients to seek emergency medical care if they experience sudden chest, stomach, or back pain.
- Diarrhea: Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, instruct patients to contact their physician as soon as possible.
- Prolongation of the QT Interval: Instruct patients to inform their physician of any personal or family history of QT prolongation or proarrhythmic conditions such as hypokalemia, bradycardia, or recent myocardial ischemia; if they are taking any Class IA (quinidine, procainamide), or Class III (amiodarone, sotalol) antiarrhythmic agents. Instruct patients to notify their physician if they have any symptoms of prolongation of the QT interval, including prolonged heart palpitations or a loss of consciousness.
- Musculoskeletal Disorders in Pediatric Patients: Instruct parents to inform their child’s physician if the child has a history of joint-related problems before taking this drug. Inform parents of pediatric patients to notify their child’s physician of any joint-related problems that occur during or following ciprofloxacin therapy [see Warnings and Precautions (5.13) and Use in Specific Populations (8.4)].
- Tizanidine: Instruct patients not to use ciprofloxacin if they are already taking tizanidine. Ciprofloxacin extended-release tablets increase the effects of tizanidine (Zanaflex®).
- Theophylline: Inform patients that ciprofloxacin extended-release tablets may increase the effects of theophylline. Life-threatening CNS effects and arrhythmias can occur. Advise the patients to immediately seek medical help if they experience seizures, palpitations, or difficulty breathing.
- Caffeine: Inform patients that ciprofloxacin extended-release tablets may increase the effects of caffeine. There is a possibility of caffeine accumulation when products containing caffeine are consumed while taking quinolones.
- Photosensitivity/Phototoxicity: Inform patients that photosensitivity/phototoxicity has been reported in patients receiving fluoroquinolones. Inform patients to minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while taking quinolones. If patients need to be outdoors while using quinolones, instruct them to wear loose-fitting clothes that protect skin from sun exposure and discuss other sun protection measures with their physician. If a sunburn-like reaction or skin eruption occurs, instruct patients to contact their physician.
- Blood Glucose Disturbances: Inform the patients that if they are diabetic and are being treated with insulin or an oral hypoglycemic agent and a hypoglycemic reaction occurs, they should discontinue ciprofloxacin extended-release tablets and consult a physician.
Antibacterial Resistance: Inform patients that antibacterial drugs including ciprofloxacin extended-release tablets should only be used to treat bacterial infections. They do not treat viral infections (for example, the common cold). When ciprofloxacin extended-release tablets are prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by ciprofloxacin extended-release tablets or other antibacterial drugs in the future.
Administration with Food, Fluids, and Concomitant Medications: Inform patients that ciprofloxacin extended-release tablets may be taken with or without food.
Inform patients to drink fluids liberally while taking ciprofloxacin extended-release tablets to avoid formation of a highly concentrated urine and crystal formation in the urine.
Inform patients that antacids containing magnesium, or aluminum, as well as sucralfate, metal cations such as iron, and multivitamin preparations with zinc or didanosine should be taken at least two hours before or six hours after ciprofloxacin extended-release tablets administration. Ciprofloxacin extended-release tablets should not be taken with dairy products (like milk or yogurt) or calcium-fortified juices alone since absorption of ciprofloxacin may be significantly reduced; however, ciprofloxacin extended-release tablets may be taken with a meal that contains these products.
Drug Interactions Oral Antidiabetic Agents: Inform patients that hypoglycemia has been reported when ciprofloxacin and oral antidiabetic agents were co-administered; if low blood sugar occurs with ciprofloxacin extended-release tablets, instruct them to consult their physician and that their antibacterial medicine may need to be changed.
Medication Guide
Ciprofloxacin Extended-Release Tablets, USP
(sip" roe flox' a sin)
Read this Medication Guide before you start taking ciprofloxacin extended-release tablets and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is the most important information I should know about ciprofloxacin extended-release tablets?
Ciprofloxacin extended-release tablets, a fluoroquinolone antibacterial medicine, can cause serious side effects. Some of these serious side effects can happen at the same time and could result in death.
If you get any of the following serious side effects while you take ciprofloxacin extended-release tablets, you should stop taking ciprofloxacin extended-release tablets immediately and get medical help right away.
- 1. Tendon rupture or swelling of the tendon (tendinitis).
-
Tendon problems can happen in people of all ages who take ciprofloxacin extended-release tablets. Tendons are tough cords of tissue that connect muscles to bones. Symptoms of tendon problems may include:
- o Pain, swelling, tears and swelling of the tendons including the back of the ankle (Achilles), shoulder, hand, or other tendon sites.
-
The risk of getting tendon problems while you take ciprofloxacin extended-release tablets is higher if you:
- o are over 60 years of age
- o are taking steroids (corticosteroids)
- o have had a kidney, heart or lung transplant
- Tendon problems can happen in people who do not have the above risk factors when they take ciprofloxacin extended-release tablets.
-
Other reasons that can increase your risk of tendon problems can include:
- o physical activity or exercise
- o kidney failure
- o tendon problems in the past, such as in people with rheumatoid arthritis (RA)
- Stop taking ciprofloxacin extended-release tablets immediately and get medical help right away at the first sign of tendon pain, swelling or inflammation. The most common area of pain and swelling is the Achilles tendon at the back of your ankle. This can also happen with other tendons.
- Tendon rupture can happen while you are taking or after you have finished taking ciprofloxacin extended-release tablets. Tendon ruptures can happen within hours or days of taking ciprofloxacin extended-release tablets and have happened up to several months after people have finished taking their fluoroquinolone.
-
Stop taking ciprofloxacin extended-release tablets immediately and get medical help right away if you get any of the following signs or symptoms of a tendon rupture:
- o hear or feel a snap or pop in a tendon area
- o bruising right after an injury in a tendon area
- o unable to move the affected area or bear weight
- 2.
Changes in sensation and possible nerve damage (Peripheral Neuropathy). Damage to the nerves in arms, hands, legs, or feet can happen in people who take fluoroquinolones, including ciprofloxacin extended-release tablets. Stop taking ciprofloxacin extended-release tablets immediately and talk to your healthcare provider right away if you get any of the following symptoms of peripheral neuropathy in your arms, hands, legs, or feet:
- pain
- burning
- tingling
- numbness
- weakness
- Ciprofloxacin extended-release tablets may need to be stopped to prevent permanent nerve damage.
- 3.
Central Nervous System (CNS) effects. Seizures have been reported in people who take fluoroquinolone antibacterial medicines, including ciprofloxacin extended-release tablets. Tell your healthcare provider if you have a history of seizures before you start taking ciprofloxacin extended-release tablets. CNS side effects may happen as soon as after taking the first dose of ciprofloxacin extended-release tablets. Stop taking ciprofloxacin extended-release tablets immediately and talk to your healthcare provider right away if you get any of these side effects, or other changes in mood or behavior:
- seizures
- hear voices, see things, or sense things that are not there (hallucinations)
- feel restless
- tremors
- feel anxious or nervous
- confusion
- depression
- trouble sleeping
- nightmares
- feel lightheaded or dizzy
- feel more suspicious (paranoia)
- suicidal thoughts or acts
- headaches that will not go away, with or without blurred vision
- 4. Worsening of myasthenia gravis (a problem that causes muscle weakness). Fluoroquinolones like ciprofloxacin extended-release tablets may cause worsening of myasthenia gravis symptoms, including muscle weakness and breathing problems. Tell your healthcare provider if you have a history of myasthenia gravis before you start taking ciprofloxacin extended-release tablets. Call your healthcare provider right away if you have any worsening muscle weakness or breathing problems.
What are ciprofloxacin extended-release tablets?
Ciprofloxacin extended-release tablets are a fluoroquinolone antibacterial medicine used in adults age 18 years and older to treat certain infections caused by certain germs called bacteria. These bacterial infections include:
- urinary tract infection
- chronic prostate infection
- lower respiratory tract infection
- sinus infection
- skin infection
- bone and joint infection
- nosocomial pneumonia
- intra-abdominal infection, complicated
- infectious diarrhea
- typhoid (enteric) fever
- cervical and urethral gonorrhea, uncomplicated
- people with a low white blood cell count and a fever
- inhalational anthrax
- plague
- Studies of ciprofloxacin extended-release tablets for use in the treatment of plague and anthrax were done in animals only, because plague and anthrax could not be studied in people.
- Ciprofloxacin extended-release tablets should not be used in patients with acute exacerbation of chronic bronchitis, acute uncomplicated cystitis, and sinus infections, if there are other treatment options available.
- Ciprofloxacin extended-release tablets should not be used as the first choice of antibacterial medicine to treat lower respiratory tract infections caused by a certain type of bacterial called Streptococcus pneumoniae.
- Ciprofloxacin is also used in children younger than 18 years of age to treat complicated urinary tract and kidney infections or who may have breathed in anthrax germs, have plague or have been exposed to plague germs.
- Children younger than 18 years of age have a higher chance of getting bone, joint, or tendon (musculoskeletal) problems such as pain or swelling while taking ciprofloxacin. Ciprofloxacin should not be used as the first choice of antibacterial medicine in children under 18 years of age.
- Ciprofloxacin extended-release tablets are only used in adults 18 years of age and older to treat urinary tract infections (complicated and uncomplicated), including kidney infections (pyelonephritis).
- It is not known if ciprofloxacin extended-release tablets are safe and effective in children under 18 years of age.
Who should not take ciprofloxacin extended-release tablets?
Do not take ciprofloxacin extended-release tablets if you:
- Have ever had a severe allergic reaction to an antibacterial medicine known as a fluoroquinolone, or are allergic to ciprofloxacin hydrochloride or any of the ingredients in ciprofloxacin extended-release tablets. See the end of this Medication Guide for a complete list of ingredients in ciprofloxacin extended-release tablets.
- Also take a medicine called tizanidine (Zanaflex®).
Ask your healthcare provider if you are not sure.
What should I tell my healthcare provider before taking ciprofloxacin extended-release tablets?
Before you take ciprofloxacin extended-release tablets, tell your healthcare provider if you:
- have tendon problems; ciprofloxacin extended-release tablets should not be used in patients who have a history of tendon problems
- have a disease that causes muscle weakness (myasthenia gravis); ciprofloxacin extended-release tablets should not be used in patients who have a known history of myasthenia gravis
- have liver problems
- have central nervous system problems (such as epilepsy)
- have nerve problems; ciprofloxacin extended-release tablets should not be used in patients who have a history of a nerve problem called peripheral neuropathy
- have or anyone in your family has an irregular heartbeat, especially a condition called “QT prolongation”
- have or have had seizures
- have kidney problems. You may need a lower dose of ciprofloxacin extended-release tablets if your kidneys do not work well.
- have joint problems including rheumatoid arthritis (RA)
- have trouble swallowing pills
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if ciprofloxacin will harm your unborn baby.
- are breastfeeding or plan to breastfeed. Ciprofloxacin passes into breast milk. You and your healthcare provider should decide whether you will take ciprofloxacin extended-release tablets or breastfeed. You should not do both.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
- Ciprofloxacin extended-release tablets and other medicines can affect each other causing side effects.
-
Especially tell your healthcare provider if you take:
- o a steroid medicine
- o an anti-psychotic medicine
- o a tricyclic antidepressant
- o a water pill (diuretic)
- o theophylline (such as Theo-24®, Elixophyllin®, Theochron®, Uniphyl®, Theolair®)
- o a medicine to control your heart rate or rhythm (antiarrhythmics)
- o an oral anti-diabetes medicine
- o phenytoin (Fosphenytoin Sodium®, Cerebyx®, Dilantin-125®, Dilantin® , Extended Phenytoin Sodium®, Prompt Phenytoin Sodium®, Phenytek®)
- o cyclosporine (Gengraf®, Neoral®, Sandimmune®, Sangcya®)
- o a blood thinner (such as warfarin, Coumadin®, Jantoven®)
- o methotrexate (Trexall®)
- o ropinirole (Requip®)
- o clozapine (Clozaril®, Fazaclo® ODT®)
- o a Non-Steroidal Anti-Inflammatory Drug (NSAID). Many common medicines for pain relief are NSAIDs. Taking an NSAID while you take ciprofloxacin extended-release tablets or other fluoroquinolones may increase your risk of central nervous system effects and seizures.
- o sildenafil (Viagra®, Revatio®)
- o duloxetine
- o products that contain caffeine
- o probenecid (Probalan®, Col-probenecid®)
-
Certain medicines may keep ciprofloxacin tablets, ciprofloxacin oral suspension from working correctly. Take ciprofloxacin tablets and oral suspension either 2 hours before or 6 hours after taking these medicines, vitamins, or supplements:
- o an antacid, multivitamin, or other medicine or supplements that have magnesium, calcium, aluminum, iron, or zinc
- o sucralfate (Carafate®)
- o didanosine (Videx®, Videx EC®)
Ask your healthcare provider for a list of these medicines if you are not sure.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take ciprofloxacin extended-release tablets?
- Take ciprofloxacin extended-release tablets exactly as your healthcare provider tells you to take them.
- Your healthcare provider will tell you how many ciprofloxacin extended-release tablets to take and when to take them.
- Take ciprofloxacin extended-release tablets one time each day at about the same time each day. Swallow the tablet whole. Do not split, crush or chew the tablet. Tell your healthcare provider if you cannot swallow the tablet whole.
- Ciprofloxacin extended-release tablets can be taken with or without food.
- Ciprofloxacin extended-release tablets should not be taken with dairy products (like milk or yogurt) or calcium-fortified juices alone, but may be taken with a meal that contains these products.
- Drink plenty of fluids while taking ciprofloxacin extended-release tablets.
-
Do not skip any doses of ciprofloxacin extended-release tablets, or stop taking them, even if you begin to feel better, until you finish your prescribed treatment unless:
- o you have tendon problems. See “What is the most important information I should know about ciprofloxacin extended-release tablets?”.
- o you have nerve problems. See “What is the most important information I should know about ciprofloxacin extended-release tablets?”.
- o you have central nervous system problems. See “What is the most important information I should know about ciprofloxacin extended-release tablets?”.
- o you have a serious allergic reaction. See “What are the possible side effects of ciprofloxacin extended-release tablets?”.
- o your healthcare provider tells you to stop taking ciprofloxacin extended-release tablets.
- Taking all of your ciprofloxacin extended-release tablets doses will help make sure that all of the bacteria are killed. Taking all of your ciprofloxacin extended-release tablets doses will help lower the chance that the bacteria will become resistant to ciprofloxacin extended-release tablets. If you become resistant to ciprofloxacin extended-release tablets, ciprofloxacin extended-release tablets and other antibacterial medicines may not work for you in the future.
- If you take too many ciprofloxacin extended-release tablets, call your healthcare provider or get medical help right away.
What should I avoid while taking ciprofloxacin extended-release tablets?
- Ciprofloxacin extended-release tablets can make you feel dizzy and lightheaded. Do not drive, operate machinery, or do other activities that require mental alertness or coordination until you know how ciprofloxacin extended-release tablets affect you.
- Avoid sunlamps, tanning beds, and try to limit your time in the sun. Ciprofloxacin extended-release tablets can make your skin sensitive to the sun (photosensitivity) and the light from sunlamps and tanning beds. You could get a severe sunburn, blisters or swelling of your skin. If you get any of these symptoms while you take ciprofloxacin extended-release tablets, call your healthcare provider right away. You should use a sunscreen and wear a hat and clothes that cover your skin if you have to be in sunlight.
What are the possible side effects of ciprofloxacin extended-release tablets?
Ciprofloxacin extended-release tablets may cause serious side effects, including:
- See “What is the most important information I should know about ciprofloxacin extended-release tablets?”.
-
Serious allergic reactions. Serious allergic reactions, including death, can happen in people taking fluoroquinolones, including ciprofloxacin extended-release tablets, even after only 1 dose. Stop taking ciprofloxacin extended-release tablets and get emergency medical help right away if you get any of the following symptoms of a severe allergic reaction:
- o hives
- o trouble breathing or swallowing
- o swelling of the lips, tongue, face
- o throat tightness, hoarseness
- o rapid heartbeat
- o faint
- o skin rash
- Skin rash may happen in people taking ciprofloxacin extended-release tablets even after only 1 dose. Stop taking ciprofloxacin extended-release tablets at the first sign of a skin rash and call your healthcare provider. Skin rash may be a sign of a more serious reaction to ciprofloxacin extended-release tablets.
-
Liver damage (hepatotoxicity). Hepatotoxicity can happen in people who take ciprofloxacin extended-release tablets. Call your healthcare provider right away if you have unexplained symptoms such as:
- o nausea or vomiting
- o stomach pain
- o fever
- o weakness
- o abdominal pain or tenderness
- o itching
- o unusual tiredness
- o loss of appetite
- o light colored bowel movements
- o dark colored urine
- o
yellowing of your skin or the whites of your eyes
Stop taking ciprofloxacin extended-release tablets and tell your healthcare provider right away if you have yellowing of your skin or white part of your eyes, or if you have dark urine. These can be signs of a serious reaction to ciprofloxacin extended-release tablets (a liver problem).
- Aortic Aneurysm and Dissection: Tell your healthcare provider if you have ever been told that you have an aortic aneurysm, a swelling of the large artery that carries blood from the heart to the body. Get emergency medical help right away if you have sudden chest, stomach, or back pain.
- Intestine infection (Pseudomembranous colitis). Pseudomembranous colitis can happen with many antibacterial medicines, including ciprofloxacin extended-release tablets. Call your healthcare provider right away if you get watery diarrhea, diarrhea that does not go away, or bloody stools. You may have stomach cramps and a fever. Pseudomembranous colitis can happen 2 or more months after you have finished your antibacterial medicine.
-
Serious heart rhythm changes (QT prolongation and torsade de pointes). Tell your healthcare provider right away if you have a change in your heart beat (a fast or irregular heartbeat), or if you faint. Ciprofloxacin extended-release tablets may cause a rare heart problem known as prolongation of the QT interval. This condition can cause an abnormal heartbeat and can be very dangerous. The chances of this event are higher in people:
- o who are elderly
- o with a family history of prolonged QT interval
- o with low blood potassium (hypokalemia)
- o who take certain medicines to control heart rhythm (antiarrhythmics)
- Joint Problems. Increased chance of problems with joints and tissues around joints in children under 18 years old can happen. Tell your child’s healthcare provider if your child has any joint problems during or after treatment with ciprofloxacin extended-release tablets.
- Sensitivity to sunlight (photosensitivity). See “What should I avoid while taking ciprofloxacin extended-release tablets?”.
- Changes in blood sugar. People who take ciprofloxacin extended-release tablets and other fluoroquinolone medicines with oral anti-diabetes medicines or with insulin can get low blood sugar (hypoglycemia) and high blood sugar (hyperglycemia). Follow your healthcare provider’s instructions for how often to check your blood sugar. If you have diabetes and you get low blood sugar while taking ciprofloxacin extended-release tablets, stop taking ciprofloxacin extended-release tablets and call your healthcare provider right away. Your antibiotic medicine may need to be changed.
The most common side effects of ciprofloxacin extended-release tablets include:
- nausea
- diarrhea
- changes in liver function tests
- vomiting
- rash
Tell your healthcare provider about any side effect that bothers you, or that does not go away.
These are not all the possible side effects of ciprofloxacin extended-release tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ciprofloxacin extended-release tablets?
- Store ciprofloxacin extended-release tablets between 20° to 25°C (68° to 77°F).
Keep ciprofloxacin extended-release tablets and all medicines out of the reach of children.
General Information about the safe and effective use of ciprofloxacin extended-release tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use ciprofloxacin extended-release tablets for a condition for which they are not prescribed. Do not give ciprofloxacin extended-release tablets to other people, even if they have the same symptoms that you have. They may harm them.
This Medication Guide summarizes the most important information about ciprofloxacin extended-release tablets. If you would like more information about ciprofloxacin extended-release tablets, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about ciprofloxacin extended-release tablets that is written for healthcare professionals.
For more information, call Mylan at 1-877-446-3679 (1-877-4-INFO-RX).
What are the ingredients in ciprofloxacin extended-release tablets?
- Active ingredients: ciprofloxacin and ciprofloxacin hydrochloride
- Inactive ingredients: carnauba wax, colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate (anhydrous), FD&C Yellow No. 6 Aluminum Lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, pregelatinized starch (corn), sodium lauryl sulfate, stearic acid, succinic acid, talc and titanium dioxide
This Medication Guide has been approved by the U.S. Food and Drug Administration.
The brands listed are trademarks of their respective owners.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Revised: 5/2019
CIPER:R17mh/MG:CIPER:R15mh
PRINCIPAL DISPLAY PANEL – 500 mg
NDC: 0378-1743-89
Once Daily
Ciprofloxacin
Extended-Release
Tablets, USP
500 mg*
PHARMACIST: Dispense the Medication
Guide provided separately to each patient.
Rx only 50 Tablets
*Each film-coated tablet contains
ciprofloxacin, USP and ciprofloxacin
hydrochloride, USP equivalent to
500 mg of ciprofloxacin.
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Keep this and all medication
out of the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Usual Dosage: See accompanying
prescribing information.
Medication Guides may be obtained
at Mylan.com/MedCenter or call
1-888-839-8993.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Mylan.com
RM1743K7
PRINCIPAL DISPLAY PANEL – 1000 mg
NDC: 0378-1745-89
Once Daily
Ciprofloxacin
Extended-Release
Tablets, USP
1000 mg*
PHARMACIST: Dispense the Medication
Guide provided separately to each patient.
Rx only 50 Tablets
*Each film-coated tablet contains
ciprofloxacin, USP and ciprofloxacin
hydrochloride, USP equivalent to
1000 mg of ciprofloxacin.
Dispense in a tight, light-resistant
container as defined in the USP
using a child-resistant closure.
Keep container tightly closed.
Keep this and all medication
out of the reach of children.
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room
Temperature.]
Usual Dosage: See accompanying
prescribing information.
Medication Guides may be obtained
at Mylan.com/MedCenter or call
1-888-839-8993.
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.
Mylan.com
RM1745K7
| CIPROFLOXACIN
ciprofloxacin tablet, film coated, extended release |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| CIPROFLOXACIN
ciprofloxacin tablet, film coated, extended release |
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||
| Labeler - Mylan Pharmaceuticals Inc. (059295980) |