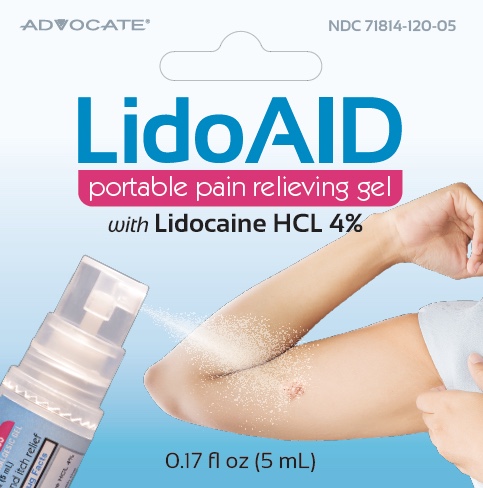
Lido AID by Diabetic Supply of Suncoast, Inc. Lido AID 0.17 oz (5mL)
Lido AID by
Drug Labeling and Warnings
Lido AID by is a Otc medication manufactured, distributed, or labeled by Diabetic Supply of Suncoast, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LIDO AID- lidocaine hcl gel
Diabetic Supply of Suncoast, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Lido AID 0.17 oz (5mL)
Warnings
For external use only. Avoid contact with the eyes.
Directions
▪adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
▪children under 2 years of age: consult a doctor
Other information
▪ store in a cool dry place between (59-86°F)15-30°C
▪ don’t use if seal over cap is broken, torn, or missing
Inactive ingredients
imidurea, methylparaben, menthol, propylene glycol, propylparaben, trolamine, water.
| LIDO AID
lidocaine hcl gel |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Diabetic Supply of Suncoast, Inc. (043081723) |