DYE-FREE CHILDRENS ALLERGY- diphenhydramine hydrochloride solution
Dye-Free Childrens ALLERGY by
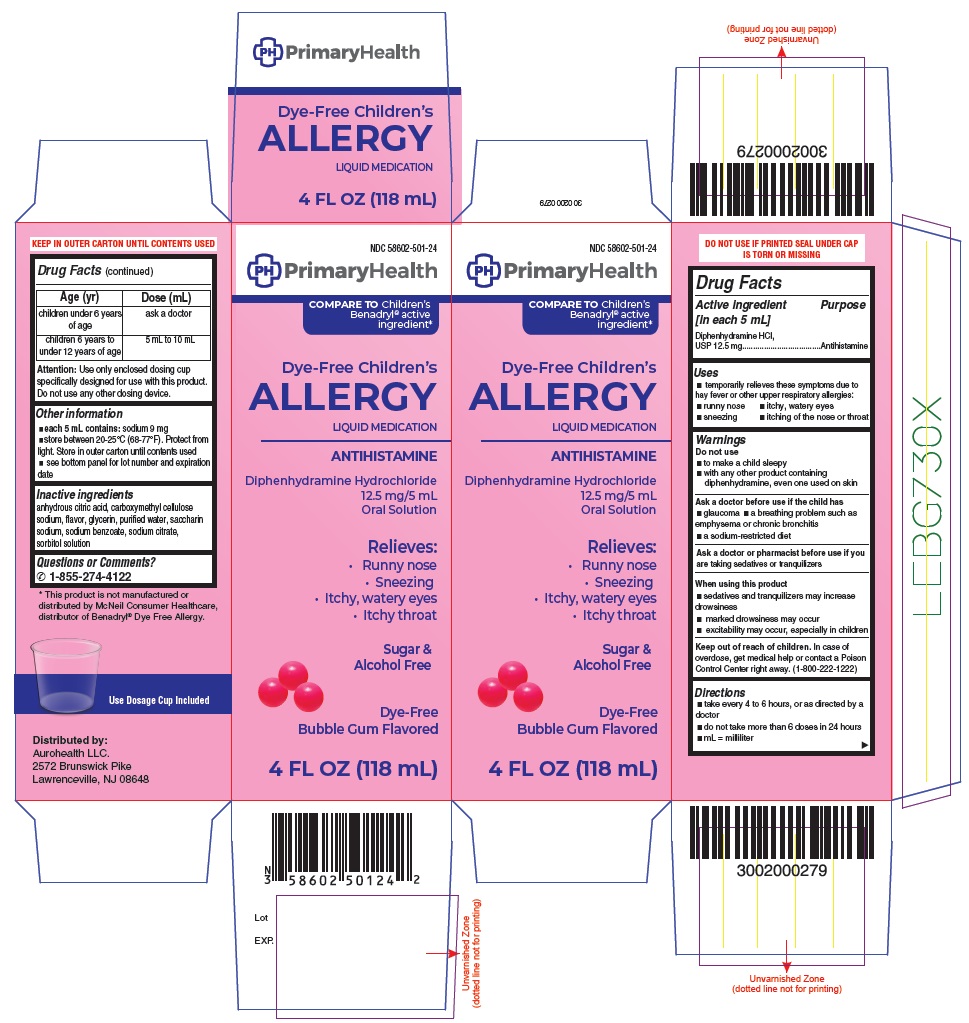
Drug Labeling and Warnings
Dye-Free Childrens ALLERGY by is a Otc medication manufactured, distributed, or labeled by Aurohealth LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- Purpose
- Uses
- Warnings
- Ask a doctor before use if the child has
- Ask a doctor or pharmacist before use if you
- When using this product
- Keep out of reach of children.
-
Directions
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 doses in 24 hours
- mL = milliliter
Age (yr)
Dose (mL)
children under 6 years of age
ask a doctor
children 6 years to under 12 years of age
5 mL to 10 mL
Attention: Use only enclosed dosing cup specifically designed for use with this product. Do not use any other dosing device.
- Other information
-
Inactive ingredients
anhydrous citric acid, carboxymethyl cellulose sodium, flavor, glycerin, purified water, saccharin sodium, sodium benzoate, sodium citrate, sorbitol solution
Questions or Comments?
1-855-274-4122
* This product is not manufactured or distributed by McNeil Consumer Healthcare, distributor of Benadryl® Dye Free Allergy.
Distributed by:
Aurohealth LLC.
2572 Brunswick Pike,
Lawrenceville, NJ 08648 -
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 FL OZ Container Label (118 mL Bottle)
NDC: 58602-501-24
PrimaryHealth
COMPARE TO children’s
Benadryl® active
ingredient*
Dye-Free Children's
ALLERGY
LIQUID MEDICATION
ANTIHISTAMINE
Diphenhydramine Hydrochloride
12.5 mg/5 mL
Oral Solution
Relieves:
- Runny Nose
- Sneezing
- Itchy, Watery Eyes
- Itchy Throat
Sugar &
Alcohol Free
Dye-Free
Bubble Gum Flavored
4 FL OZ (118 mL)

-
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 4 FL OZ Container Carton Label (118 mL Bottle)
NDC: 58602-501-24
PrimaryHealth
COMPARE TO children’s
Benadryl® active
ingredient*
Dye-Free Children's
ALLERGY
LIQUID MEDICATION
ANTIHISTAMINE
Diphenhydramine Hydrochloride
12.5 mg/5 mL
Oral Solution
Relieves:
- Runny Nose
- Sneezing
- Itchy, Watery Eyes
- Itchy Throat
Sugar &
Alcohol Free
Dye-Free
Bubble Gum Flavored
4 FL OZ (118 mL)
-
INGREDIENTS AND APPEARANCE
DYE-FREE CHILDRENS ALLERGY
diphenhydramine hydrochloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 58602-501 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SORBITOL (UNII: 506T60A25R) Product Characteristics Color WHITE (Colorless) Score Shape Size Flavor BUBBLE GUM Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 58602-501-24 1 in 1 CARTON 09/01/2018 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 09/01/2018 Labeler - Aurohealth LLC (078728447) Establishment Name Address ID/FEI Business Operations Aurohealth LLC 078728447 MANUFACTURE(58602-501)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.