SODIUM IODIDE I-131 solution, concentrate
Sodium Iodide I-131 by
Drug Labeling and Warnings
Sodium Iodide I-131 by is a Prescription medication manufactured, distributed, or labeled by International Isotopes Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Sodium Iodide I-131 safely and effectively. See full prescribing information for Sodium Iodide I-131.
Sodium Iodide I-131 (for the preparation of sodium iodide I 131 capsules and solution), therapeutic, for oral use.
Initial U.S. Approval: 1971INDICATIONS AND USAGE
Sodium Iodide I-131 is a radioactive therapeutic agent indicated for the treatment of hyperthyroidism and selected cases of carcinoma of the thyroid. (1)
DOSAGE AND ADMINISTRATION
- The concentrated sodium iodide I 131 solution provided must be diluted. ( 2.2)
- See Full Prescribing Information for important administration instructions and dilution and preparation instructions for sodium iodide l 131 capsules or oral solution. ( 2.2, 2.4)
- The recommended dose is based on the thyroid gland uptake as well as the size of the gland:
- Treatment of Hyperthyroidism: Recommended dosage is 148 to 370 megabecquerels (MBq) [4 to 10 millicuries (mCi). ( 2.3)
- Treatment of Thyroid Carcinoma: Recommended dosage is 1,110 to 33,700 MBq (30 to 100 mCi). ( 2.3)
DOSAGE FORMS AND STRENGTHS
Vials: Sodium Iodide I-131 solution (with a radioconcentration ranging between 18.5 - 129.5 GBq/mL (500 and 3500 mCi/mL) at the time of calibration) for the preparation of sodium iodide I-131 capsules, therapeutic or sodium iodide I-131 solution, therapeutic. ( 3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Radiation-induced thyroiditis may cause or worsen hyperthyroidism. Consider pre-treatment with anti-thyroid medications. ( 5.1)
- Multiple non-thyroid radiation toxicities, including hematopoietic suppression: Individualize dose and monitor for toxicity. ( 5.2)
- Fetal toxicity: May cause severe and irreversible hypothyroidism in the neonate. Verify absence of pregnancy before administering the product. ( 5.4, 8.1, 8.3)
- Radiation exposure to breast tissue with lactation: Sodium iodide I 131 concentrates in the breast of lactating women. Discontinue lactation 6 weeks prior to therapy. ( 5.5, 8.2)
ADVERSE REACTIONS
Common adverse reactions reported with therapeutic doses of sodium iodide I 131 include local swelling, radiation sickness, sialadenitis, salivary gland dysfunction, bone marrow depression, lacrimal gland dysfunction, hypothyroidism, hyperthyroidism, thyrotoxic crisis, acute leukemia, solid cancer. (6)
To report SUSPECTED ADVERSE REACTIONS, contact International Isotopes Inc. at 1-800-699-3108 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2017
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Radiation Safety
2.2 Important Administration Instructions
2.3 Recommended Dosage and Administration
2.4 Dilution and Preparation Instructions
2.5 Radiation Dosimetry
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Radiation-induced Thyroiditis
5.2 Radiation-induced Toxicities
5.3 Hypersensitivity Reactions
5.4 Fetal Toxicity
5.5 Increased Radiation Exposure to Breast Tissue with Lactation
5.6 Transient Infertility
5.7 Risk of Radiation Exposure
5.8 Risk of Decreased Effectiveness of Therapy
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
11.1 Physical Characteristics
11.2 External Radiation
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Radiation Safety
1. Sodium Iodide I-131 is a radioactive drug. Handle with appropriate safety measures to minimize radiation exposure to the patient and healthcare workers [see Warning and Precautions (5.7)]
2. Use only by, or under the direction of, physicians who are qualified by specific training and experience in the safe use and handling of radioactive materials, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radiopharmaceuticals.
3. Use waterproof gloves when handling and administering the product.
4. Maintain adequate shielding during the life of the product.
5. Measure patient dose with a suitable radioactivity calibration system immediately prior to administration.2.2 Important Administration Instructions
- Do not directly administer the concentrated sodium iodide I 131 solution provided to patients. The concentrated sodium iodide I 131 solution must be diluted and prepared prior to administration [see Dosage and Administration (2.4)] .
- Obtain a pregnancy test in females of reproductive potential prior to administration to verify the absence of pregnancy [see Contraindications (4) and Use in Specific Populations (8.1,8.3)] .
- Instruct patients to fast at least 2 hours before and 2 hours after administration to ensure absorption.
- Instruct patients to hydrate before and after administration of sodium iodide I 131 and to void frequently to enhance urinary elimination of the radioiodide that is not absorbed by the thyroid gland [see Warnings and Precautions (5.2)] .
- Instruct patients to maintain a low-iodide diet two weeks prior to radioiodide administration and continue for several days during the uptake or imaging process [see Warnings and Precautions (5.8) and Drug Interactions (7)] .
- Instruct patients to discontinue the anti-thyroid therapy three days before administration of sodium iodide I 131 [see Warnings and Precautions (5.8) and Drug Interactions (7)] .
- For patients with a history of renal impairment, evaluate renal function for therapeutic planning and consider dosimetry [see Use in Specific Populations (8.6)] .
- Obtain a complete blood count within one month of therapy. If patients show leukopenia or thrombocytopenia, dosimetry should be used to determine a safe sodium iodide I 131 activity, while delivering less than 2 Gy to the bone marrow [see Warnings and Precautions (5.2)] .
2.3 Recommended Dosage and Administration
Individualization of Therapy
The recommended dose for orally administered sodium iodide I 131 capsules or solution is based on the thyroid gland uptake as well as the size of the gland. Thyroidal uptake and size should be determined by the physician prior to treatment and may be useful in calculating the therapeutic dose to be administered to the individual patient.Treatment of Hyperthyroidism
The recommended dose is 148 to 370 MBq (4 to 10 mCi) administered orally. Toxic nodular goiter may require a larger dose.Treatment of Thyroid Carcinoma
The recommended dose is 1100 to 3700 MBq (30 to 100 mCi) administered orally. For subsequent ablation of metastases, the recommended dose is 3700 to 7400 MBq (100 to 200 mCi) administered orally.2.4 Dilution and Preparation Instructions
Preparation of Dilute Sodium Iodide I 131 Solution
1. Using the calibration date and radionuclidic concentration on the label of the product vial, calculate the required volume to produce the necessary dose in MBq or mCi.
2. Using a shielded syringe, remove the required volume.
3. Using the shielded syringe, transfer the required volume to a suitably shielded receiving vial.
4. Add diluent to the receiving vial to produce a final dose of the desired volume.
5. The recommended diluent is Purified Water with 300 mg of sodium phosphate dibasic anhydrous dissolved in the water. Acidic diluents should not be used as they may cause the pH to drop below 7.5 and stimulate the volatilization of Iodine I-131 hydriodic acid.
6. Present the dose in a shielded container for administration to the patient with a straw.
Preparation of Sodium Iodide I 131 Capsules
1. Use one large #1 empty gelatin capsule and one small #2 gelatin capsule for each dose to be prepared. Each large capsule is empty and each small capsule should contain approximately 300 mg of dibasic sodium phosphate anhydrous as the absorbing buffer.
2. Using the calibration date and radionuclidic concentration on the label of the sodium iodide I-131 lead shield, calculate the required volume to produce the necessary dose in MBq or mCi.
3. Open the larger #1 capsule by pulling apart the capsule into two pieces as illustrated below:
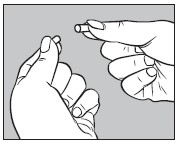
4. Insert an unopened small #2 capsule containing 300 mg of sodium phosphate dibasic anhydrous powder into the bottom half of the empty large capsule as illustrated below: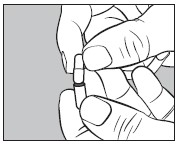
5. With an appropriate syringe, withdraw the required volume of sodium iodide I 131 Solution (maximum 150 microliters) from the vial as illustrated below:
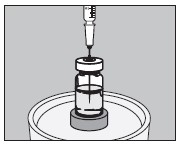
6. Inject into the center of the small capsule through the top as illustrated below:
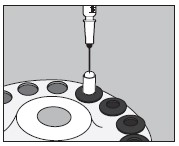
7. Slip the upper half of the large capsule over the bottom half to completely cover the small capsule and push down gently until locked as illustrated below:
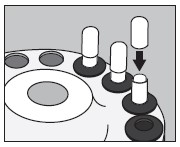
8. Measure the patient dose in a suitable radioactivity calibration system immediately prior to administration.
9. Prepared capsules may be stored in a suitable polypropylene container and place inside a lead pot until use, within seven days.
2.5 Radiation Dosimetry
- The biokinetic modeling and radiation dose distributions associated with thyroid uptake of iodide I 131 depend on dietary intake of stable iodide and presume normal production of thyroid hormone. Table 1 shows a range of uptake percentages in an average adult (73.7 kg reference model). Table 1 is not intended to be used for treatment planning.
- For a thyroid blocked from iodide uptake in the production of hormones, the effective half-life of iodide I 131 is approximately 1.4 hours; for "low" to "high" uptake, the effective half-life of I 131 ranges from approximately 80 to 90 hours.
Table 1 * - * Table 1 is not intended for treatment planning.
- † These columns are not applicable to estimate organ or effective doses in patients following thyroidectomy. In patients with thyroid cancer following thyroidectomy, organ and effective doses can be estimated from the "blocked"-thyroid-uptake values.
- ‡ These values presume unimpeded production of thyroid hormone and may not be applicable to estimate thyroid dose and effective dose in patients who have had previous treatment with I 131 for hyperthyroidism
Absorbed dose per unit activity sodium iodide I 131 administered orally (mGy/MBq) in adult (73.7-kg reference model) Organ Thyroid uptake of I 131 (% administered activity A 0)
24 h after oral administrationBlocked thyroid
(0% A 0)Low uptake †
(16% A 0)Medium uptake †
(26% A 0)High uptake †
(36% A 0)Adrenals 0.044 0.051 0.055 0.059 Bone surfaces 0.030
0.089 0.12 0.16 Brain 0.021 0.093 0.13 0.17 Breast 0.020 0.038 0.048 0.058 Gallbladder wall 0.037 0.043 0.046 0.049 Gastrointestinal tract Esophagus 0.024 0.10 0.14 0.19 Stomach wall 0.87 0.77 0.71 0.66 Small intestine wall 0.035 0.033 0.032 0.032 Colon wall 0.14 0.14 0.14 0.14 (Upper large intestine wall) 0.12 0.12 0.12 0.12 (Lower large intestine wall) 0.17 0.17 0.17 0.16 Heart wall 0.062 0.089 0.10 0.12 Kidneys 0.27 0.27 0.27 0.27 Liver 0.050 0.093 0.12 0.14 Lungs 0.053 0.10 0.13 0.15 Muscles 0.026 0.084 0.12 0.15 Ovaries 0.038 0.037 0.036 0.035 Pancreas 0.060 0.064 0.066 0.068 Red marrow 0.031 0.072 0.095 0.12 Salivary glands 0.27 0.22 0.19 0.16 Skin 0.019 0.043 0.057 0.071 Spleen 0.064 0.069 0.072 0.075 Testes 0.025 0.024 0.023 0.22 Thymus 0.024 0.10 0.14 0.19 Thyroid 2.2 280 ‡ 430 ‡ 580 ‡ Urinary bladder wall 0.54 0.45 0.39 0.34 Uterus 0.045 0.042 0.040 0.038 Remaining organs 0.029 0.084 0.11 0.15 Effective dose per administered activity (mSv/MBq) 0.28 14 ‡ 22 ‡ 29 ‡ -
3 DOSAGE FORMS AND STRENGTHS
Sodium Iodide I-131 solution componentis available in customer ordered vials available in 1, 2, 3 and 5 mL V-Vials and 2 and 5 mL Serum Vials containing colorless, aqueous concentrated sodium iodide I-131 with strengths ranging between 18.5 - 129.5 GBq/mL (500 - 3500 mCi/mL) referenced to calibration time for the preparation of sodium iodide I-131 capsules, therapeutic or sodium iodide I-131 solution, therapeutic.
-
4 CONTRAINDICATIONS
Sodium Iodide I-131 is contraindicated in:
- Patients with vomiting and diarrhea [see Warning and Precautions (5.7)] .
- Pregnancy [see Warnings and Precautions (5.4), see Use in Specific Populations (8.1)] .
- Lactation [see Warnings and Precautions (5.5)] .
- Patients receiving concurrent anti-thyroid therapy [see Warning and Precautions (5.1) and Drug Interactions (7)] .
-
5 WARNINGS AND PRECAUTIONS
5.1 Radiation-induced Thyroiditis
Sodium iodide I 131 may cause thyroiditis with release of thyroid hormone, which may aggravate hyperthyroidism and thyrotoxic cardiac disease [see Adverse Reactions (6)] . When treating hyperthyroidism, consider pre-treatment anti-thyroid medication to help deplete the thyroid hormone. Discontinue the anti-thyroid therapy three days before administration of sodium iodide I 131 [see Drug Interactions (7)] . Consider a beta-blocker pre or post-treatment to minimize the risk of hyperthyroidism and thyroid storm.
The thyroiditis may cause gland enlargement resulting in tenderness and swelling of the neck, pain on swallowing, sore throat, and cough; which may occur approximately the third day after sodium iodide I 131 administration. Consider management with pain-reliever or anti-inflammatory medications.
5.2 Radiation-induced Toxicities
Sodium Iodide I 131 may cause radiation induced toxicities [see Adverse Reactions (6)] :
- Dose-dependent fatalities (bone marrow suppression, malignancy).
- Dose-dependent hematopoietic suppression which manifests as a transient thrombocytopenia or neutropenia 3-5 weeks following sodium iodide I 131 administrations, may lead to increased susceptibility to infections or bleeding.
- Salivary gland toxicity: sialadenitis, xerostomia.
- Lacrimal gland toxicity: conjunctivitis, xerophthalmia, and epiphora.
Obtain a complete blood count within one month of therapy. If patients show leukopenia or thrombocytopenia, dosimetry should be used to determine a safe sodium iodide I 131 activity, while delivering less than 2 Gy to the bone marrow.
Advise good hydration for one week following sodium iodide I 131 administration and stimulate salivary flow via a sialagogue (e.g. sugar-free candy or gum, pilocarpine, and ascorbic acid) to reduce radiation exposure to the salivary glands.
Advise patients to void frequently after administration of radioiodide to enhance excretion.
5.3 Hypersensitivity Reactions
Hypersensitivity reactions including anaphylaxis may occur in patients who receive sodium iodide I 131. Although iodide is not considered an allergen, hypersensitivity reactions may occur in relation with excipients or chemical component of the capsule, such as sodium thiosulfate. Obtain and document an allergy history, particularly a sulfite allergy. Emergency resuscitation equipment and personnel should be immediately available [see Adverse Reactions (6)] .
5.4 Fetal Toxicity
Sodium Iodide I-131 is contraindicated in pregnancy because sodium iodide I 131 crosses the placenta and fetal exposure can lead to neonatal hypothyroidism. Multiple reports in the published literature describe hypothyroidism in the neonates following in utero exposure to sodium iodide I 131. Some cases of neonatal hypothyroidism were severe and irreversible. Verify pregnancy status of females of reproductive potential prior to initiating Sodium Iodide I-131 treatment. Advise females and males of reproductive potential to use effective contraception during treatment with Sodium Iodide I-131 and for at least six months after the last dose [see Use in Specific Populations (8.1, 8.3)].
5.5 Increased Radiation Exposure to Breast Tissue with Lactation
Sodium Iodide I-131 is contraindicated in lactating women because sodium iodide I 131 concentrates in the breast via the increased expression of the sodium iodide symporter in breast tissue with lactation. The literature describes moderate to marked radioiodine uptake in the breast tissue for 5-32 weeks post cessation of breast feeding. Advise lactating women to discontinue breast feeding at least 6 weeks prior to administration of sodium iodide I 131 to allow sufficient time for involution to occur and to avoid excess concentration of sodium iodide I 131 in breast tissue. Consider administration of drugs to suppress lactation. Consider diagnostic scintigraphy before administration of sodium iodide I 131 to assess the persistence of uptake by breast tissue. If sodium iodide I 131 is administered in the postpartum period, the lactating mother should not breastfeed the infant [see Use in Specific Populations (8.2)] .
5.6 Transient Infertility
Transient dose-related impairment of testicular function in men and transient ovarian insufficiency in women has been reported after sodium iodide I 131 therapy. Consider sperm banking for men who are anticipated to receive cumulative sodium iodide I 131 doses greater than 19,000 MBq (520 mCi) [see Use in Specific Populations (8.3)] .
5.7 Risk of Radiation Exposure
Household Contacts
Instruct patients to follow radiation safety precautions after receiving Sodium Iodide I-131 to minimize the radiation contamination of other persons or the environment. Patients should avoid close contact with others, especially pregnant women and children, and take care to avoid contamination of other persons or the environment with body fluids.Patients and Healthcare Providers
Sodium Iodide I-131 contributes to a patient’s overall long-term cumulative radiation exposure, which is associated with an increased risk of cancer. Follow safe handling and administration to minimize radiation exposure to the patient and healthcare providers.5.8 Risk of Decreased Effectiveness of Therapy
Certain food or drugs may alter the thyroid uptake of sodium iodide I 131 and diminish its effectiveness. Recent intake of stable iodide in any form, or the use of thyroid or anti-thyroid drugs may diminish thyroid uptake of sodium iodide I 131 [see Drug Interactions (7)] .
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in labeling:
- Radiation-induced Thyroiditis [see Warnings and Precautions (5.1)] .
- Radiation-induced Toxicities [see Warnings and Precautions (5.2)].
- Hypersensitivity Reactions [see Warnings and Precautions (5.3)].
- Fetal Toxicity [see Warnings and Precautions (5.4), Use in Specific Population (8.1)].
- Increased Radiation Exposure to Breast Tissue with Lactation [see Warnings and Precautions (5.5), Use in Specific Populations (8.2)].
- Transient Infertility [see Warnings and Precautions (5.6), Use in Specific Population (8.3)].
- Radiation Exposure [see Warnings and Precautions (5.7)].
The following adverse reactions have been identified during post-approval use of sodium iodide I 131. Because these reactions are voluntarily reported from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Gastrointestinal disorders: sialadenitis, salivary gland dysfunction, nausea, vomiting, gastritis.
- Cardiac disorders: chest pain, tachycardia.
- Skin and subcutaneous tissue disorders: itching, rash, hives.
- Endocrine disorders: hypothyroidism, hyperthyroidism, thyrotoxic crisis, hypoparathyroidism
- General disorders and administration site conditions: local swelling of thyroid or sites of iodide avid tumor.
- Hematologic and lymphatic disorders including fatalities: bone marrow depression, anemia, leukopenia, thrombocytopenia, and blood dyscrasia.
- Neoplasms benign, malignant and unspecified (including cysts and polyps): acute leukemia, solid cancer.
- Eye disorders: lacrimal gland dysfunction.
- Congenital, familial and genetic disorders: congenital hypothyroidism, chromosomal abnormalities.
- Immune system disorders: bronchospasm.
- Nervous system disorders: headache, *cerebral edema.
- Respiratory, thoracic and mediastinal disorders: **radiation pneumonitis, **pulmonary fibrosis.
* In patients with iodide-avid brain metastases
** In patients with iodide-avid lung metastases -
7 DRUG INTERACTIONS
- Concomitant use of bone marrow depressants may enhance the depression of the hematopoietic system caused by the use of large doses of sodium iodide I 131 [see Warnings and Precautions (5.2)] .
- Many drugs and iodide-containing foods interfere with the accumulation of radioiodide by the thyroid. Review the patients history, current medications, and recent diagnostic tests prior to the administration of sodium iodide I 131 [see Warnings and Precautions (5.8)] .
- Advise patients to maintain a low-iodide diet two weeks prior to radioiodide administration and continue for several days during the uptake or imaging process and to discontinue taking the following products before they undergo the procedure as shown in Table 2.
Table 2 Pharmaceuticals/OTCs/Agents Blocking Radioiodine Uptake Type of Medication Recommended time of withdrawal Thionamide medications
(e.g., propylthiouracil, methimazole, carbimazole)3 days Multivitamins containing iodide 10 days Natural or synthetic thyroid hormones
triiodothyronine
thyroxine2 weeks
4 weeks
Iodide-containing foods: iodized salt, dairy products, egg yolks, seafood, turkey and liver 2 weeks Kelp, agar, carrageenan, Lugol solution 3 weeks Saturated solution of potassium iodide 3 weeks Topical iodide
(e.g., surgical skin preparation)3 weeks Intravenous radiographic contrast agents
Water soluble
Lipophilic2 months
6 months
Amiodarone 6 months -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Sodium Iodide I-131 is contraindicated in pregnancy because fetal exposure can lead to neonatal hypothyroidism, which in some cases is severe and irreversible. Data from the published literature describe thyroid abnormalities after fetal exposure; including agenesis of the thyroid and hypothyroidism (see Data). No animal reproductive studies have been conducted.Clinical Considerations.
Fetal/ Neonatal Adverse Reactions
A fetus exposed to sodium iodide I 131 can develop neonatal hypothyroidism. Delay in diagnosis of neonatal hypothyroidism after exposure to sodium iodide I 131 in utero can result in severe sequelae such as decreased mental capacity and delayed bone age. Monitor thyroid function in any infant born after in utero exposure to sodium iodide I 131.Data
Human Data
Sodium iodide I 131 crosses the placenta and the fetal thyroid begins to concentrate iodide during the 10-12 th week of gestation. In literature reports of maternal exposures to sodium iodide I 131 at doses of 333 – 8325 MBq (9 – 225 mCi) during 4-26 weeks gestational age, the most common adverse outcomes were hypothyroid infants and children.8.2 Lactation
Risk Summary
Sodium Iodide I-131 is contraindicated during lactation because I 131 concentrates in the breast during lactation via the increased expression of the sodium iodide symporter in breast tissue [see Warnings and Precautions (5.5)] . If sodium iodide I 131 is administered in the postpartum period, the lactating mother should not breastfeed. In addition, to minimize the absorbed radiation dose to the breast tissue, breastfeeding and breast-pumping should be discontinued for at least 6 weeks before administration of sodium iodide I 131 (see Data). Women may breast feed with the birth of another child.Infants exposed to sodium iodide I 131 through breast milk are at risk for development of hypothyroidism because sodium iodide I 131 is distributed into breast milk and may reach concentrations equal to or greater than concentrations in maternal plasma (see Data).
Data
Human Data
Limited published literature describes sodium iodide I 131 transfer into breast milk and thyroidal uptake by the breastfed infant. The amount of sodium Iodide I 131 detected in the breast milk at 36-48 hours after administration is 1-27% of the injected dose (with injected doses between 1.1 – 5143 MBq).8.3 Females and Males of Reproductive Potential
Sodium Iodide I-131 is contraindicated in pregnancy because of the risk of fetal hypothyroidism [see Use in Specific Populations (8.1)] .
Pregnancy Testing
Obtain a pregnancy test in females of reproductive potential and verify the absence of pregnancy before initiating treatment [see Dosage and Administration (2.2), Warnings and Precautions (5.4)].Contraception
Advise females and males of reproductive potential to use effective contraception during treatment with Sodium Iodide I-131 and for at least six months after the last dose of Sodium Iodide I-131.Infertility
Females
Fertility may be impaired with sodium iodide I 131 treatment. Transient amenorrhea and ovarian insufficiency have been observed after sodium iodide I 131 therapy in females. The literature describes reports of transient menstrual cycle irregularities, including amenorrhea, and ovarian failure in females treated with cumulative doses of 1000 – 59,000 MBq (27 – 1594 mCi) sodium iodide I 131.Males
Fertility may be impaired with sodium iodide I 131 treatment. Discuss sperm banking for males who are expected to receive a high cumulative dose of sodium iodide I 131. Transient dose-related impairment of testicular function after sodium iodide I 131 therapy has been reported in the published literature. The literature describes reports of males treated with sodium iodide I 131 at doses of 370 MBq – 22,000 MBq (10 – 595 mCi) resulting in transiently impaired testicular function (including spermatogenesis). Permanent impairment is described with high cumulative doses ranging from 19,000 – 29,000 MBq (520 – 800 mCi).8.4 Pediatric Use
Safety and efficacy in pediatric patients have not been established. Pediatric patients are at an increased lifetime risk for malignancy from radiation exposure
8.5 Geriatric Use
Clinical experience has not identified differences in safety or effectiveness in geriatric patients compared to younger patients. However, elderly patients are more likely to have decreased renal function and radiation exposure is greater in patients with impaired renal function [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
8.6 Renal Impairment
Sodium Iodide I 131 is primarily excreted by the kidneys. Renal function impairment decreases excretion of sodium iodide I 131 and increases the radiation exposure and risk of radiation toxicity. For patients with a history of renal impairment, evaluate renal function for therapeutic planning and consider dosimetry. Sodium Iodide I 131 is dialyzable. Hemodialysis can be used to reduce total body radiation exposure [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
In case of exposure to a radioactive dose of sodium iodide I 131 exceeding the intended therapeutic dose, provide general supportive care, promote frequent voiding, monitor for bone marrow and thyroid suppression. Consider administering a thyroid blocking agent (e.g. potassium iodide (KI) or perchlorate) promptly within 4 to 6 hours after the exposure. Assess the benefit of administering a thyroid blocking agent against the risk of failure of sodium iodide I 131 therapy. Appropriate replacement therapy is recommended if hypothyroidism occurs.
-
11 DESCRIPTION
The concentrated sodium iodide I 131 solution is available in strengths ranging from 18.5 - 129.5 GBq/mL (500 - 3500 mCi/mL) and is used for the preparation of sodium iodide I 131 capsules or sodium iodide I 131 solution of varying strengths for oral administration for therapy. Each mL of the concentrated solution contains the labeled amount of no carrier added sodium iodide I 131 formulated in 0.05M sodium hydroxide with 0.02M sodium thiosulfate added as a stabilizer. The sodium hydroxide maintains the pH above 7.5 to avoid the formation of volatile HI, and the sodium thiosulfate serves as a reducing agent. The pH of the sodium iodide I 131 solution is greater than 7.5.
Sodium iodide I 131 solution is designated chemically as Na 131I and has a molecular weight of 153.99.
11.1 Physical Characteristics
Iodine I-131 decays by beta emission and associated gamma emission with a physical half-life of 8.025 days. The principal radiation emissions are listed in Table 3.
Table 3 Principal Radiation Emission Data from Decay of Sodium Iodide I 131 Radiation Mean % per
DisintegrationMean Energy
(keV)Beta-1 2.1% 69.4 Beta-3 7.2% 96.6 Beta-4 89.4% 191.6 Gamma-7 6.1% 284.3 Gamma-14 81.2% 364.5 Gamma-18 7.1% 637.0 11.2 External Radiation
The specific gamma-ray constant for iodide I 131 is 4.26 × 10 -13 C·m 2·kg -1·MBq -1·s -1 (2.2 R·cm 2/mCi·hr). The first half-value thickness of lead (Pb) for iodide I 131 is 0.27 cm. A range of values for the relative attenuation of the radiation emitted by iodide I 131 that results from interposition of various thicknesses of Pb is shown in Table 4. For example, the use of 2.59 cm of Pb will decrease the external radiation exposure by a factor of about 1,000.
Table 4 Radiation Attenuation of Iodine I 131 by Lead Shielding Shield Thickness
(Pb) cmCoefficient of Attenuation 0.27 0.5 0.56 0.25 0.99 10 -1 2.59 10 -2 4.53 10 -3 To correct for physical decay of iodine I 131, the fractions that remain at selected intervals after the time of calibration are shown in Table 5.
Table 5 Physical Decay Chart: Iodine I-131 Half-Life 8.025 days *Calibration time Days Fraction Remaining Days Fraction Remaining Days Fraction Remaining 0* 1.000 11 .388 22 .151 1 .918 12 .356 23 .138 2 .842 13 .327 24 .127 3 .773 14 .300 25 .116 4 .709 15 .275 26 .107 5 .651 16 .253 27 .098 6 .597 17 .232 28 .090 7 .548 18 .213 29 .083 8 .503 19 .195 30 .076 9 .461 20 .179 10 .423 21 .164 -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Iodide is actively transported by the sodium-iodide symporter (NIS) protein, in thyroid follicular cells. Iodide is concentrated in follicular cells to levels up to 50 times higher than in the plasma. Iodide is metabolically oxidized by thyroid peroxidase to iodinium (I +) which in turn iodinates tyrosine residues of thyroglobulin (tri or tetra-iodinated tyrosine). The beta emission of I 131 is responsible for the therapeutic effect.
12.2 Pharmacodynamics
The relationship between the extent of iodide I 131 exposure and pharmacologic effects has not been explored in clinical trials.
12.3 Pharmacokinetics
Absorption
Following oral administration of sodium iodide I 131, 90% of the administered radioactivity of Iodide I 131 is systemically absorbed in the first 60 minutes.Distribution
Following absorption, I 131 is distributed within the extra-cellular space. It is actively transported by the sodium-iodide symporter (NIS) protein, and binds to thyroglobulin resulting in accumulation in the thyroid. The thyroid uptake of iodide is usually increased in hyperthyroidism and in goiter, and is decreased in hypothyroidism. Sodium Iodide I 131 also accumulates in the stomach, choroid plexus, salivary glands, breast, liver, gall bladder, and kidneys.Elimination
Metabolism
In thyroidal follicular cells iodide is oxidized through the action of thyroid peroxidase to iodinium (I +) which in turn iodinates tyrosine residues of thyroglobulin.Excretion
Sodium iodide I 131 is excreted in urine and feces. The normal range of urinary excretion is 37% to 75% of the administered dose, varying with the thyroid and renal function of the patient. Fecal excretion is about 10%. -
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
The Sodium Iodide I-131 Solution can be supplied in any of the following vials: 1, 2, 3 and 5 mL clear glass V-Vials and in 2 and 5 mL serum vials.The strength is specified by the customer from 18.5 to 129.5 GBq/mL (500 to 3500 mCi/mL) at time of calibration.
Kit for Preparation of Sodium Iodide I-131 Capsules and Solution NDC Code Size/Type Container 69208-005-15 1 mL V-vial 69208-005-25 2 mL V-vial 69208-005-35 3 mL V-vial 69208-005-55 5 mL V-vial 69208-005-60 2 mL Serum vial 69208-005-70 5 mL Serum vial 16.2 Storage
The Sodium Iodide I-131 should be stored between 2°C and 25°C (36°F and 77°F). Store and dispose of sodium iodide I 131 in compliance with the appropriate regulations of the government agency authorized to license the use of this radionuclide.
Discard unused capsules after all sodium iodide I 131solution has been dispensed or expired. New blister packages of hard gelatin capsules are provided with each new shipment of Sodium Iodide I-131 Solution.
This radiopharmaceutical is approved for use by persons under license by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
-
17 PATIENT COUNSELING INFORMATION
Radiation Safety Precautions [see Warnings and Precautions (5.7)] .
Review the most recent professional society guidelines and publications that describe important components of the patient counseling process and discuss measures to minimize inadvertent radiation exposure to the patient.Fetal Toxicity [see Contraindications (4), Warnings and Precautions (5.4) and Use in Specific Populations (8.1,8.3)].
- Advise female patients of the risk to a fetus.
- Advise females and males of reproductive potential to use effective contraception during treatment with sodium iodide I 131 and for at least 6 months after the last dose.
- Advise female patients to contact their healthcare provider with a known or suspected pregnancy.
Lactation
Instruct women to stop breastfeeding and breast-pumping at least 6 weeks prior to sodium iodide I 131 administration [see Contraindications (4), Warnings and Precautions (5.5) and Use in Specific Populations (8.2)].Effects on Fertility
Advise females and males of reproductive potential of the potential for impaired fertility with Sodium Iodide I-131 treatment [see Warnings and Precautions (5.6) and Use in Specific Populations (8.3)].Manufactured by:
International Isotopes Inc., Idaho Falls, Idaho 83401, USA - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SODIUM IODIDE I-131
sodium iodide i-131 solution, concentrateProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69208-005 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM IODIDE I-131 (UNII: 29VCO8ACHH) (IODIDE ION I-131 - UNII:4GC1FOQ22U) IODIDE ION I-131 3500 mCi in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) 0.05 mmol in 1 mL SODIUM THIOSULFATE (UNII: HX1032V43M) 0.02 mmol in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69208-005-15 1 in 1 BOX 09/01/2010 1 1 in 1 CANISTER 1 1 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product 2 NDC: 69208-005-25 1 in 1 BOX 09/01/2010 2 1 in 1 CANISTER 2 2 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product 3 NDC: 69208-005-35 1 in 1 BOX 09/01/2010 3 1 in 1 CANISTER 3 3 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product 4 NDC: 69208-005-55 1 in 1 BOX 09/01/2010 4 1 in 1 CANISTER 4 5 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product 5 NDC: 69208-005-60 1 in 1 BOX 09/01/2010 5 1 in 1 CANISTER 5 2 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product 6 NDC: 69208-005-70 1 in 1 BOX 09/01/2010 6 1 in 1 CANISTER 6 5 mL in 1 VIAL, DISPENSING; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/01/2010 Labeler - International Isotopes Inc (933155509) Registrant - International Isotopes Inc (933155509) Establishment Name Address ID/FEI Business Operations International Isotopes Inc 933155509 manufacture(69208-005) , analysis(69208-005)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

