RECOTHROM- thrombin topical recombinant kit
RECOTHROM by
Drug Labeling and Warnings
RECOTHROM by is a Other medication manufactured, distributed, or labeled by The Medicines Company, AbbVie BioResearch Center, Anderson Brecon Inc, Cook Pharmica LLC, Patheon Italia S.p.A, Sharp Corporation, Zymogenetics. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use RECOTHROM safely and effectively. See full prescribing information for RECOTHROM.
RECOTHROM, Thrombin topical (Recombinant)
Lyophilized Powder for Solution - For Topical Use Only
Initial U.S. Approval: 2008
INDICATIONS AND USAGE
RECOTHROM, Thrombin topical (Recombinant), is a topical thrombin indicated to aid hemostasis whenever oozing blood and minor bleeding from capillaries and small venules is accessible and control of bleeding by standard surgical techniques (such as suture, ligature, or cautery) is ineffective or impractical in adults and pediatric populations greater than or equal to one month of age. (1)
RECOTHROM may be used in conjunction with an absorbable gelatin sponge, USP. (1)
DOSAGE AND ADMINISTRATION
- For topical use only. DO NOT INJECT. (2)
- Reconstitute RECOTHROM powder with sterile 0.9% sodium chloride, USP, yielding a solution containing 1000 units (international units of potency) per mL. (2.1)
- Apply RECOTHROM solution directly to bleeding site surface or in conjunction with absorbable gelatin sponge. The amount required depends upon the area of tissue to be treated. (2.2)
DOSAGE FORMS AND STRENGTHS
RECOTHROM is available as 5000-unit and 20,000-unit vials of sterile recombinant topical thrombin lyophilized powder for solution. When reconstituted as directed, the final solution contains 1000 units/mL of RECOTHROM. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
ADVERSE REACTIONS
- The most common adverse reaction (incidence 6%) was thromboembolic events. (5.1,6)
- Antibody formation to RECOTHROM occurred in <1% of patients. None of the antibodies detected neutralized native human thrombin. (6.2)
To report SUSPECTED ADVERSE REACTIONS, contact The Medicines Company at 1-888-784-7662 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Pregnancy: No human or animal data. Use only if clearly needed. (8.1)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 9/2014
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Reconstitution of RECOTHROM
2.2 Application Techniques
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Thrombosis
5.2 Hypersensitivity Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Immunogenicity
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
RECOTHROM ®, Thrombin topical (Recombinant), is a topical thrombin indicated to aid hemostasis whenever oozing blood and minor bleeding from capillaries and small venules is accessible and control of bleeding by standard surgical techniques (such as suture, ligature, or cautery) is ineffective or impractical in adults and pediatric populations greater than or equal to one month of age.
RECOTHROM may be used in conjunction with an absorbable gelatin sponge, USP.
-
2 DOSAGE AND ADMINISTRATION
For topical use only. DO NOT INJECT.
2.1 Reconstitution of RECOTHROM
The volume of reconstituted RECOTHROM required will vary depending on the size and number of bleeding sites to be treated and the method of application.
Inspect the integrity of the RECOTHROM package and contents. Do not use if the packaging or contents have been damaged or opened.
Reconstitute the lyophilized powder using the supplied diluent.
Use aseptic technique when handling vials and syringes.
5000-unit RECOTHROM Reconstitution
Units used herein represent international units of potency determined using a reference standard that has been calibrated against the World Health Organization Second International Standard for Thrombin.
- Remove flip-off cap from the top of the RECOTHROM vial.
- Attach the needle-free transfer device and snap it into place on the vial by placing the vial on a flat surface and attaching the transfer device straight into the center of the vial stopper.
- Attach the prefilled diluent syringe to the needle-free transfer device.
- Inject the 5 mL of diluent from the syringe into the product vial. Keep the syringe plunger depressed.
- DO NOT reuse the diluent syringe for transfer of the reconstituted product. Remove and discard the diluent syringe.
- Gently swirl and invert the product vial until the powder is completely dissolved. Avoid excessive agitation. The powder should dissolve in less than one minute at room temperature.
- Apply the pre-printed "DO NOT INJECT" label to the sterile, empty transfer syringe provided, then draw up the RECOTHROM solution.
20,000-unit RECOTHROM Reconstitution
- Remove the flip-off cap from the top of the RECOTHROM vial and the diluent vial.
- Attach a needle-free transfer device (one each) to the RECOTHROM and diluent vials and snap them into place by placing the vial on a flat surface and attaching the transfer device straight into the center of the vial stopper.
- Open the sterile, empty 20-mL syringe package and apply the pre-printed "DO NOT INJECT" label to the syringe.
- Attach the labeled 20-mL syringe to the needle-free transfer device on the diluent vial (injection of air into the diluent vial may facilitate withdrawal of the diluent).
- Draw up 20 mL of diluent from the vial into the syringe.
- Remove the diluent-filled syringe from the diluent vial and attach it to the transfer device on the RECOTHROM vial.
- Transfer the 20 mL of diluent from the syringe into the RECOTHROM vial; the vacuum in the vial facilitates transfer.
- Leave the syringe attached and gently swirl and invert the RECOTHROM vial until the powder is completely dissolved. Avoid excessive agitation. The powder should dissolve in less than one minute at room temperature.
- With the same syringe, draw up the RECOTHROM solution.
2.2 Application Techniques
Topically apply RECOTHROM solution directly or in conjunction with absorbable gelatin sponge onto the bleeding site. DO NOT INJECT.
The amount required depends upon the area of tissue to be treated and the method of application.
Vials are for single use only. Discard unused contents.
Use with Absorbable Gelatin Sponge
Refer to the absorbable gelatin sponge labeling for safety information and instructions on appropriate use.
- Transfer solution from syringe to a sterile bowl or basin.
- Place the desired size pieces of the absorbable gelatin sponge into the bowl containing reconstituted RECOTHROM to completely saturate the sponge(s).
- Remove the saturated sponge(s) and squeeze gently to remove excess RECOTHROM.
- Apply the sponge to the bleeding site in a single layer.
Use with ZymoGenetics Spray Applicator Kit
- Hold the outer sealed tray, peel back the lid, and aseptically transfer the inner sealed sterile tray to the sterile field.
- Open the inner tray seal and use the sterile bowl as the receptacle for reconstituted RECOTHROM solution.
- Refer to Spray Applicator Kit instructions for spray pump and syringe spray assembly and use.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
- Do not inject directly into the circulatory system.
- Do not use for the treatment of massive or brisk arterial bleeding.
- Do not administer to patients with a history of hypersensitivity to RECOTHROM or any components of RECOTHROM.
- Do not use in patients with known hypersensitivity to hamster proteins.
-
5 WARNINGS AND PRECAUTIONS
5.1 Thrombosis
RECOTHROM may cause thrombosis if it enters the circulatory system. Apply topically. DO NOT INJECT.
5.2 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, may occur. RECOTHROM is produced in a genetically modified Chinese Hamster Ovary (CHO) cell line and may contain hamster or snake proteins [see Contraindications (4) and Description (11)].
-
6 ADVERSE REACTIONS
Thromboembolic adverse reactions were reported in 6% of surgical patients treated with RECOTHROM in all completed clinical trials (N=644) [see Warnings and Precautions (5.1)].
Antibody formation to RECOTHROM occurred in <1% of patients. None of the antibodies detected neutralized native human thrombin [see Adverse Reactions (6.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug product cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Clinical trials have been performed with RECOTHROM applied with absorbable gelatin sponge and applied with a spray applicator. A total of 644 patients were exposed to RECOTHROM in these studies.
RECOTHROM Used in Conjunction with Absorbable Gelatin Sponge
Four hundred eleven (411) patients were treated in a randomized, double-blind, controlled trial that compared RECOTHROM to bovine thrombin. Both thrombins were applied with a gelatin sponge in patients undergoing spinal surgery, hepatic resection, peripheral arterial bypass surgery, or arteriovenous graft formation for hemodialysis access.1 The incidence of thromboembolic adverse reactions was similar between the RECOTHROM and bovine thrombin treatment groups (see Table 1).
Table 1: Incidence of Adverse Reactions with RECOTHROM and Bovine Thrombin * THROMBIN-JMI® Thrombin, Topical (Bovine) Adverse Reaction Category RECOTHROM
(N=205)
n (%)Bovine Thrombin*
(N=206)
n (%)Thromboembolic events 11 (5%) 12 (6%) In an open-label, single-group trial (N=209), patients with documented or highly likely prior exposure to bovine thrombin within the previous three years were treated with RECOTHROM when undergoing surgeries (spinal, peripheral arterial bypass, or arteriovenous graft formation for hemodialysis access).2 The incidence of thromboembolic adverse reactions in this study was 9%.
In an open-label, single-group trial of re-exposure to RECOTHROM (N=31), patients with documented prior exposure to RECOTHROM were treated with RECOTHROM during surgery (spinal, peripheral arterial bypass, arteriovenous graft formation, or other procedures).3 The incidence of thromboembolic adverse reactions in this study was 3%.
In other randomized, double-blind trials across a range of surgical settings (N=130; spinal surgery, hepatic resection, peripheral arterial bypass surgery, or arteriovenous graft formation for hemodialysis access), the safety of RECOTHROM (n=88 patients) was compared to placebo (RECOTHROM excipients reconstituted with sterile 0.9% sodium chloride, USP) (n=42 patients). The incidence of thromboembolic adverse reactions in this study was 5% for RECOTHROM and 12% for placebo.
RECOTHROM Applied with Spray Applicator
RECOTHROM was applied with a spray applicator in two open-label clinical trials: a single-group trial in adult and pediatric burn patients (N=72; ≤16 years of age, (n=4) and ≥17 years of age, (n=68)) treated with RECOTHROM applied to the wound excision site prior to autologous skin grafting4; and in a single-group trial in pediatric patients (one month to 17 years of age) undergoing synchronous burn wound excision and autologous skin grafting (N=30; ≤16 years of age, (n=26); ≥17 years of age, (n=4)).5 In the first study, the incidence of thromboembolic adverse reactions was 1%. In the second study, there were no reported thromboembolic adverse reactions [see Use in Specific Populations (8.4)].
6.2 Immunogenicity
The detection of antibody formation is highly dependent upon the sensitivity and specificity of the assay. The absolute immunogenicity rates reported here are difficult to compare with the results from other products due to differences in assay methodology, patient populations, and other underlying factors.
The potential for development of antibodies to RECOTHROM was evaluated in multiple clinical trials and included patients with a single exposure to RECOTHROM as well as patients who were re-exposed to RECOTHROM during a subsequent surgical procedure. Only patients with both baseline and post-treatment antibody specimens available were evaluated for the development of specific anti-RECOTHROM product antibodies, which was defined as seroconversion or a ≥1.0 titer unit (≥10-fold) increase in antibody levels after study treatment. Five of 609 (0.8%; 95% CI, 0.4%-2.8%) evaluable patients developed specific anti-RECOTHROM product antibodies. None of these antibodies were found to neutralize native human thrombin. There was no difference in anti-RECOTHROM product antibody formation incidence among patients exposed to RECOTHROM applied with absorbable gelatin sponge, USP or with spray applicator.
In a clinical trial comparing RECOTHROM to bovine thrombin (N=411; n=398 antibody evaluable) for the development of specific anti-product antibodies, blood samples were collected at baseline and at Day 29 in both treatment groups and were analyzed by ELISA.1 At baseline, 1.5% of RECOTHROM patients (n=3/198) had positive anti-product antibody titers compared with 5% of bovine thrombin patients (n=10/200). Of these patients, none of the RECOTHROM group and eight in the bovine thrombin group exhibited ≥1.0 titer unit (≥10-fold) increases in anti-product antibody levels after study treatment.
At Day 29, three of 198 (1.5%; 95% CI, 0%-4%) patients in the RECOTHROM group developed specific anti-product antibodies (one patient also developed anti-CHO host cell protein antibodies); 43 of 200 patients in the bovine thrombin group (22%; 95% CI, 16%-28%) developed specific antibodies to bovine thrombin product. Treatment with RECOTHROM resulted in a statistically significant lower incidence of specific anti-product antibody development. Because the study was not powered to detect a difference in clinical outcomes attributable to antibody formation, no conclusions can be drawn regarding the clinical significance of the difference in antibody formation based on the results of this study. None of the antibodies in the RECOTHROM group neutralized native human thrombin. Antibodies against bovine thrombin product were not tested for neutralization of native human thrombin.
In a trial of patients with a high likelihood of prior exposure to bovine thrombin, 15.6% of patients (n=32/205) had anti-bovine thrombin product antibodies and 2% of patients (n=4/200) had anti-RECOTHROM product antibodies at baseline.2 Following treatment, none of the 200 evaluable patients (patients for whom post-treatment specimens were available) developed antibodies to RECOTHROM.
In a trial of patients previously exposed to RECOTHROM, 31 patients were re-exposed to RECOTHROM during a subsequent surgery.3 None of the evaluable patients (n=30) had anti-RECOTHROM product antibodies at baseline and none developed antibodies at Day 29.
In a trial of RECOTHROM, including 26 pediatric patients (aged one month to 16 years) and four patients 17 years of age, one patient without prior thrombin exposure had pre-existing anti-RECOTHROM product antibodies at baseline.5 None of the 27 evaluable patients developed anti-RECOTHROM product antibodies at Day 29.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Animal reproduction studies have not been conducted with RECOTHROM. It is also not known whether RECOTHROM can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. RECOTHROM should be given to a pregnant woman only if clearly needed.
8.4 Pediatric Use
A total of 30 pediatric patients, ages 0 to 16 years (one month to 2 years, n=10; 2 to 12 years, n=12; 12 to 16 years, n=8), were treated in clinical trials with RECOTHROM using a spray applicator to burn wound excision sites prior to autologous skin grafting. No patient experienced a thromboembolic adverse reaction. The safety of RECOTHROM in pediatric patients greater than or equal to one month of age is supported by these data and by extrapolation of efficacy from adequate and well-controlled studies of RECOTHROM in adults. Safety and efficacy have not been established in neonates [see Adverse Reactions (6)].
8.5 Geriatric Use
Of 644 patients in clinical studies of RECOTHROM, 36% (n=232/644) were ≥65 years old and 15% (n=95/644) were ≥75 years old.
No differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
-
11 DESCRIPTION
RECOTHROM, Thrombin topical (Recombinant), is a human coagulation protein produced via recombinant DNA technology from a genetically modified CHO cell line. RECOTHROM is identical in amino acid sequence and structurally similar to naturally occurring human thrombin. RECOTHROM precursor is secreted to culture medium as single chain form that is proteolytically converted to a two-chain active form (using a protein derived from snakes) and is purified by a chromatographic process that yields a product having hemostatic activities similar to native human thrombin. The cell line used to manufacture RECOTHROM has been tested and shown to be free of known infectious agents. The cell culture process used in the manufacture of RECOTHROM employs no additives of human or animal origin. The purification process includes solvent-detergent treatment and nano-filtration steps dedicated to viral clearance.
RECOTHROM is provided as a sterile, white to off-white, preservative-free, lyophilized powder in vials for reconstitution with diluent (sterile 0.9% sodium chloride, USP). Reconstitution with the provided diluent, as described [see Dosage and Administration (2.1)], yields a solution with a pH of 6.0 containing 1000 units/mL of recombinant thrombin for topical use. The formulated product is a clear, colorless solution upon reconstitution and contains the following excipients: histidine, mannitol, sucrose, polyethylene glycol 3350, sodium chloride, and calcium chloride dihydrate, USP.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
RECOTHROM, Thrombin topical (Recombinant), is a specific human serine protease that promotes hemostasis and acts locally when applied topically to a site of bleeding.
RECOTHROM activates platelets and catalyzes the conversion of fibrinogen to fibrin, which are steps that are essential for blood clot formation.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In vitro cytotoxicity studies have been performed in mouse L929 fibroblast cell cultures and demonstrate a concentration-dependent effect on cell morphology. The thrombin-induced morphological changes were similar to those seen with bovine thrombin.
13.2 Animal Toxicology and/or Pharmacology
In a study in nonhuman primates, RECOTHROM was applied directly to a liver wound with an absorbable gelatin sponge, USP. In a second study, RECOTHROM was administered subcutaneously once weekly for four weeks to nonhuman primates following repeat doses of 5405 units/m2. In both studies, RECOTHROM had no effect on clinical signs, serum chemistry, coagulation parameters, or histopathology; only normal postsurgical findings were observed. No animals developed anti-RECOTHROM product antibodies in either study.
RECOTHROM was found to be non-irritating when instilled in the eyes (200 units) or applied to normal or abraded skin of rabbits (up to 1000 units/site).
To evaluate RECOTHROM inhibition and clearance from the bloodstream, radiolabeled RECOTHROM was administered intravenously or subcutaneously to nonhuman primates and applied with an absorbable gelatin sponge, USP, in a rabbit hepatic wound model. RECOTHROM did not circulate in the blood as free, active molecule, but was rapidly inactivated (<5 minutes) after formation of complexes with endogenous inhibitors (e.g., antithrombin III); these complexes were cleared by the liver.
RECOTHROM applied with an absorbable gelatin sponge, USP, was shown to decrease time to hemostasis (TTH) when compared to saline in a rabbit hepatic wound model and rat heminephrectomy model. RECOTHROM also reduced TTH when directly applied in a porcine partial-thickness excisional skin-wound model as compared to saline control (or no treatment).
RECOTHROM applied with a gauze sponge decreased TTH in a concentration-dependent manner in both the rabbit and rat models. Concentrations of RECOTHROM >1000 units/mL were no different than 1000 units/mL while the effect of RECOTHROM diluted to a concentration of 100 units/mL on TTH was indistinguishable from placebo.
-
14 CLINICAL STUDIES
RECOTHROM was evaluated in a randomized, double-blind comparative clinical trial to bovine thrombin. Each thrombin was topically applied to bleeding sites with an absorbable gelatin sponge at a nominal concentration of 1000 units/mL.1 Patients (N=411) were a heterogeneous surgical population undergoing surgery in one of four surgical settings: spinal surgery (n=122, 30%), hepatic resection (n=125, 30%), peripheral arterial bypass surgery (n=88, 21%), and arteriovenous graft formation for hemodialysis access (n=76, 18%). Patients with prior heparin-induced thrombocytopenia were excluded. Patient ages ranged from 21 to 89 years, gender was 53% male and 47% female, and the distribution by race was 68% white, 18% black or African American, and 14% other. The distribution of these characteristics was similar in both the RECOTHROM and bovine thrombin treatment groups.
The objectives of the study were to evaluate the comparative efficacy, safety, and immunogenicity of RECOTHROM and bovine thrombin in combination with an absorbable gelatin sponge as adjuncts to hemostasis in surgery. Efficacy was evaluated by the incidence of hemostasis within 10 minutes. Bleeding appropriate for evaluation was defined as mild to moderate bleeding, either on its own or remaining after brisk bleeding was controlled by standard surgical modalities. Although multiple bleeding sites could be treated, only one bleeding site per patient was selected to determine effectiveness.
Table 2 summarizes the incidence of hemostasis within 10 minutes for each treatment for the 401 efficacy evaluable patients. The incidence of hemostasis within 10 minutes was comparable for the RECOTHROM and bovine thrombin groups.
Table 2: Hemostasis Within 10 Minutes* * Evaluation of hemostasis at ≤10 minutes for patients treated at 1 of 4 primary TTH bleeding site types: epidural venous plexus, hepatic resection site, peripheral arterial bypass proximal anastomosis, and arteriovenous graft arterial anastomosis.
† THROMBIN-JMI® Thrombin, Topical (Bovine)RECOTHROM
(N=198)
(%)Bovine Thrombin*
(N=203)
(%)Overall 95% 95% Spinal surgery 98% 98% Hepatic resection 98% 97% Peripheral arterial bypass 85% 86% Arteriovenous graft formation 97% 97% The percentage of patients achieving hemostasis at 1.5, 3, 6, and 10 minutes is listed in Table 3.
Table 3: Cumulative Incidence of Hemostasis Over Time * THROMBIN-JMI® Thrombin, Topical (Bovine) Time (Minutes) RECOTHROM
(N=198)
(%)Bovine Thrombin*
(N=203)
(%)1.5 48% 46% 3 81% 72% 6 92% 88% 10 95% 95%
-
15 REFERENCES
- Chapman WC, Singla N, Genyk Y, et al. A phase 3, randomized, double-blind comparative study of the efficacy and safety of topical recombinant human thrombin and bovine thrombin in surgical hemostasis. J Am Coll Surg. 2007;205(2):256-265.
- Singla NK, Ballard JL, Moneta G, et al. A phase 3b open-label, single-group immunogenicity and safety study of topical recombinant thrombin in surgical hemostasis. J Am Coll Surg. 2009;209(1):68-74.
- Singla NK, Gasparis AP, Ballard JL, et al. Immunogenicity and safety of re-exposure to recombinant human thrombin in surgical hemostasis. J Am Coll Surg. 2011;213(6):722-727.
- Greenhalgh DG, Gamelli RL, Collins J, et al. Recombinant thrombin: safety and immunogenicity in burn wound excision and grafting. J Burn Care Res. 2009;30(3):371-379.
- Foster KN, Mullins RF, Greenhalgh DG, et al. Recombinant human thrombin: safety and immunogenicity in pediatric burn wound excision. J Ped Surg. 2011;46(10):1992-1999.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
RECOTHROM, Thrombin topical (Recombinant), is supplied in single-use, preservative-free vials in the following packages:
NDC: 65293-006-41
A 5000-unit vial of RECOTHROM with a 5-mL prefilled diluent syringe (containing sterile 0.9% sodium chloride, USP), a sterile needle-free transfer device, a 5-mL sterile empty syringe, and a pre-printed label.NDC: 65293-007-41
A 20,000-unit vial of RECOTHROM with a 20-mL vial of diluent (containing sterile 0.9% sodium chloride, USP), 2 sterile needle-free transfer devices, a 20-mL sterile empty syringe, and a pre-printed label.NDC: 65293-007-50
The 20,000-unit RECOTHROM kit co-packaged with The Medicines Company Spray Applicator Kit containing a spray pump, a spray bottle, a syringe spray tip, a syringe, a bowl, and 2 blank labels.No RECOTHROM kit components contain latex.
Store RECOTHROM sterile powder vials at 2°C to 25°C (36°F to 77°F). Do not freeze.
Reconstituted solutions of RECOTHROM prepared with sterile 0.9% sodium chloride, USP, may be stored for up to 24 hours at 2°C to 25°C (36°F to 77°F). Discard reconstituted solution after 24 hours.
-
17 PATIENT COUNSELING INFORMATION
Because topical thrombin may cause the formation of clots in blood vessels if absorbed systemically, advise patients to consult their physician if they experience leg tenderness or swelling, chest pain, shortness of breath, or difficulty speaking or swallowing [see Warnings and Precautions (5.1)].
Manufactured for The Medicines Company
The Medicines Company, 8 Sylvan Way, Parsippany NJ 07054
U.S. License No. 1902RECOTHROM is a registered trademark of ZymoGenetics, Inc. All other trademarks are the property of their respective owners.
[PN 6001- 1 PI; Rev March 2014]
-
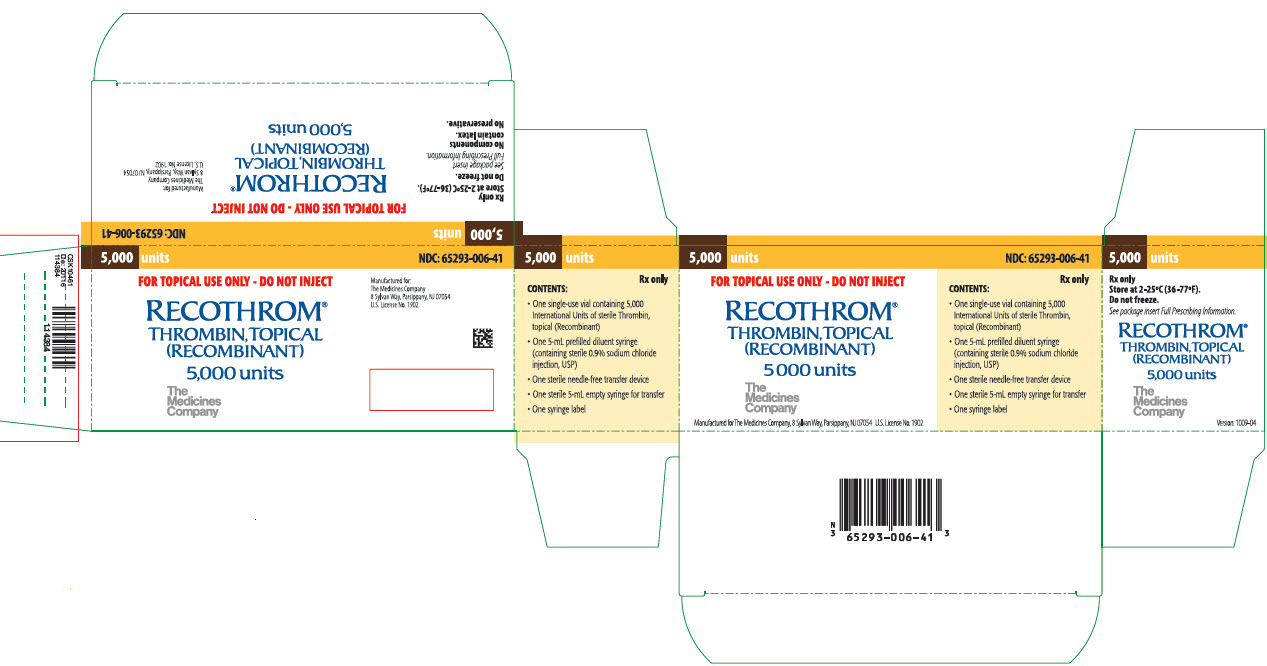
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 5000-UNIT VIAL
5,000 units
NDC: 65293-006-41
FOR TOPICAL USE ONLY - DO NOT INJECT
RECOTHROM®
THROMBIN, TOPICAL
(RECOMBINANT)5,000 units
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 20000-UNIT VIAL
20,000 units
NDC: 65293-007-41
FOR TOPICAL USE ONLY - DO NOT INJECT
RECOTHROM®
THROMBIN, TOPICAL
(RECOMBINANT)20,000 units

-
INGREDIENTS AND APPEARANCE
RECOTHROM
thrombin topical recombinant kitProduct Information Product Type PLASMA DERIVATIVE Item Code (Source) NDC: 65293-006 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 65293-006-41 1 in 1 KIT; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 5 mL Part 2 1 SYRINGE 5 mL Part 1 of 2 RECOTHROM
thrombin topical recombinant powder, for solutionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength THROMBIN ALFA (UNII: SCK81AMR7R) (THROMBIN ALFA - UNII:SCK81AMR7R) THROMBIN ALFA 1000 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) SUCROSE (UNII: C151H8M554) SODIUM CHLORIDE (UNII: 451W47IQ8X) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) HISTIDINE (UNII: 4QD397987E) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 5 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 01/29/2008 Part 2 of 2 SODIUM CHLORIDE
normal saline solutionProduct Information Route of Administration TOPICAL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 5 mL in 1 SYRINGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 01/29/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 01/29/2008 RECOTHROM
thrombin topical recombinant kitProduct Information Product Type PLASMA DERIVATIVE Item Code (Source) NDC: 65293-007 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 65293-007-41 1 in 1 KIT; Type 0: Not a Combination Product 2 NDC: 65293-007-50 1 in 1 KIT; Type 0: Not a Combination Product Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 20 mL Part 2 1 VIAL 20 mL Part 1 of 2 RECOTHROM
thrombin topical recombinant powder, for solutionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength THROMBIN ALFA (UNII: SCK81AMR7R) (THROMBIN ALFA - UNII:SCK81AMR7R) THROMBIN ALFA 1000 [iU] in 1 mL Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) SUCROSE (UNII: C151H8M554) SODIUM CHLORIDE (UNII: 451W47IQ8X) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) HISTIDINE (UNII: 4QD397987E) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 20 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 06/09/2008 Part 2 of 2 SODIUM CHLORIDE
sodium chloride solutionProduct Information Route of Administration TOPICAL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 20 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 06/09/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125248 06/09/2008 Labeler - The Medicines Company (040861601) Establishment Name Address ID/FEI Business Operations AbbVie BioResearch Center 078521730 ANALYSIS(65293-006, 65293-007) , MANUFACTURE(65293-006, 65293-007) Establishment Name Address ID/FEI Business Operations Anderson Brecon Inc 053217022 LABEL(65293-006, 65293-007) , PACK(65293-006, 65293-007) Establishment Name Address ID/FEI Business Operations Cook Pharmica LLC 172209277 ANALYSIS(65293-006, 65293-007) , MANUFACTURE(65293-006, 65293-007) Establishment Name Address ID/FEI Business Operations Patheon Italia S.p.A 434078638 ANALYSIS(65293-006, 65293-007) , MANUFACTURE(65293-006, 65293-007) Establishment Name Address ID/FEI Business Operations Sharp Corporation 143696495 LABEL(65293-006, 65293-007) , PACK(65293-006, 65293-007) Establishment Name Address ID/FEI Business Operations Zymogenetics 017854688 ANALYSIS(65293-006, 65293-007)
Trademark Results [RECOTHROM]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 RECOTHROM 77146747 3432311 Live/Registered |
BAXTER INTERNATIONAL INC. 2007-04-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.