DIURIL- chlorothiazide suspension
DIURIL by
Drug Labeling and Warnings
DIURIL by is a Prescription medication manufactured, distributed, or labeled by Salix Pharmaceuticals, Inc, Paddock Laboratories, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
DESCRIPTION
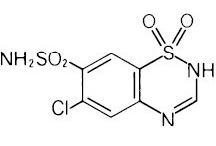
DIURIL® (chlorothiazide) is a diuretic and antihypertensive. It is 6-chloro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its empirical formula is C7H6CIN3O4S2 and its structural formula is:
It is a white, or practically white, crystalline powder with a molecular weight of 295.72, which is very slightly soluble in water, but readily soluble in dilute aqueous sodium hydroxide. It is soluble in urine to the extent of about 150 mg per 100 mL at pH 7.
DIURIL Oral Suspension contains 250 mg of chlorothiazide per 5 mL, alcohol 0.5 percent, with methylparaben 0.12 percent, propylparaben 0.02 percent, and benzoic acid 0.1 percent added as preservatives. The inactive ingredients are D&C Yellow 10, flavors, glycerin, purified water, sodium saccharin, sucrose and tragacanth.
-
CLINICAL PHARMACOLOGY
The mechanism of the antihypertensive effect of thiazides is unknown. DIURIL does not usually affect normal blood pressure.
DIURIL affects the distal renal tubular mechanism of electrolyte reabsorption. At maximal therapeutic dosage all thiazides are approximately equal in their diuretic efficacy.
DIURIL increases excretion of sodium and chloride in approximately equivalent amounts. Natriuresis may be accompanied by some loss of potassium and bicarbonate.
After oral use diuresis begins within 2 hours, peaks in about 4 hours and lasts about 6 to 12 hours.
Pharmacokinetics and Metabolism
DIURIL is not metabolized but is eliminated rapidly by the kidney. The plasma half-life of chlorothiazide is 45-120 minutes. After oral doses, 10-15 percent of the dose is excreted unchanged in the urine. Chlorothiazide crosses the placental but not the blood-brain barrier and is excreted in breast milk.
-
INDICATIONS AND USAGE
DIURIL is indicated as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
DIURIL has also been found useful in edema due to various forms of renal dysfunction such as nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
DIURIL is indicated in the management of hypertension either as the sole therapeutic agent or to enhance the effectiveness of other antihypertensive drugs in the more severe forms of hypertension.
Use in Pregnancy. Routine use of diuretics during normal pregnancy is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy and there is no satisfactory evidence that they are useful in the treatment of toxemia.
Edema during pregnancy may arise from pathologic causes or from the physiologic and mechanical consequences of pregnancy. Thiazides are indicated in pregnancy when edema is due to pathologic causes, just as they are in the absence of pregnancy (see PRECAUTIONS, Pregnancy). Dependent edema in pregnancy, resulting from restriction of venous return by the gravid uterus, is properly treated through elevation of the lower extremities and use of support stockings. Use of diuretics to lower intravascular volume in this instance is illogical and unnecessary. During normal pregnancy there is hypervolemia which is not harmful to the fetus or the mother in the absence of cardiovascular disease. However, it may be associated with edema, rarely generalized edema. If such edema causes discomfort, increased recumbency will often provide relief. Rarely this edema may cause extreme discomfort which is not relieved by rest. In these instances, a short course of diuretic therapy may provide relief and be appropriate.
- CONTRAINDICATIONS
-
WARNINGS
Use with caution in severe renal disease. In patients with renal disease, thiazides may precipitate azotemia. Cumulative effects of the drug may develop in patients with impaired renal function.
Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor alterations of fluid and electrolyte balance may precipitate hepatic coma.
Thiazides may add to or potentiate the action of other antihypertensive drugs.
Sensitivity reactions may occur in patients with or without a history of allergy or bronchial asthma.
The possibility of exacerbation or activation of systemic lupus erythematosus has been reported.
Lithium generally should not be given with diuretics (see PRECAUTIONS, Drug Interactions).
-
PRECAUTIONS
General
All patients receiving diuretic therapy should be observed for evidence of fluid or electrolyte imbalance: namely, hyponatremia, hypochloremic alkalosis, and hypokalemia. Serum and urine electrolyte determinations are particularly important when the patient is vomiting excessively or receiving parenteral fluids. Warning signs or symptoms of fluid and electrolyte imbalance, irrespective of cause, include dryness of mouth, thirst, weakness, lethargy, drowsiness, restlessness, confusion, seizures, muscle pains or cramps, muscular fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances such as nausea and vomiting.
Hypokalemia may develop, especially with brisk diuresis, when severe cirrhosis is present or after prolonged therapy.
Interference with adequate oral electrolyte intake will also contribute to hypokalemia. Hypokalemia may cause cardiac arrhythmias and may also sensitize or exaggerate the response of the heart to the toxic effects of digitalis (e.g., increased ventricular irritability). Hypokalemia may be avoided or treated by use of potassium-sparing diuretics or potassium supplements such as foods with a high potassium content.
Although any chloride deficit is generally mild and usually does not require specific treatment except under extraordinary circumstances (as in liver disease or renal disease), chloride replacement may be required in the treatment of metabolic alkalosis.
Dilutional hyponatremia may occur in edematous patients in hot weather; appropriate therapy is water restriction, rather than administration of salt, except in rare instances when the hyponatremia is life-threatening. In actual salt depletion, appropriate replacement is the therapy of choice.
Hyperuricemia may occur or acute gout may be precipitated in certain patients receiving thiazides.
In diabetic patients dosage adjustments of insulin or oral hypoglycemic agents may be required. Hyperglycemia may occur with thiazide diuretics. Thus latent diabetes mellitus may become manifest during thiazide therapy.
The antihypertensive effects of the drug may be enhanced in the post-sympathectomy patient.
If progressive renal impairment becomes evident, consider withholding or discontinuing diuretic therapy.
Thiazides have been shown to increase the urinary excretion of magnesium; this may result in hypomagnesemia.
Thiazides may decrease urinary calcium excretion. Thiazides may cause intermittent and slight elevation of serum calcium in the absence of known disorders of calcium metabolism. Marked hypercalcemia may be evidence of hidden hyperparathyroidism. Thiazides should be discontinued before carrying out tests for parathyroid function.
Increases in cholesterol and triglyceride levels may be associated with thiazide diuretic therapy.
Laboratory Tests
Periodic determination of serum electrolytes to detect possible electrolyte imbalance should be done at appropriate intervals.
Drug Interactions
When given concurrently the following drugs may interact with thiazide diuretics.
Alcohol, Barbiturates, or Narcotics - potentiation of orthostatic hypotension may occur.
Antidiabetic Drugs (Oral Agents and Insulin) - dosage adjustment of the antidiabetic drug may be required.
Other Antihypertensive Drugs - additive effect or potentiation.
Cholestyramine and Colestipol Resins - Both cholestyramine and colestipol resins have the potential of binding thiazide diuretics and reducing diuretic absorption from the gastrointestinal tract.
Corticosteroids, ACTH - intensified electrolyte depletion, particularly hypokalemia.
Pressor Amines (e.g., Norepinephrine) - possible decreased response to pressor amines but not sufficient to preclude their use.
Skeletal Muscle Relaxants, Nondepolarizing (e.g., Tubocurarine) - possible increased responsiveness to the muscle relaxant.
Lithium - generally should not be given with diuretics. Diuretic agents reduce the renal clearance of lithium and add a high risk of lithium toxicity. Refer to the package insert for lithium preparations before use of such preparations with DIURIL.
Non-steroidal Anti-inflammatory Drugs Including Selective Cyclooxygenase-2 (COX-2) Inhibitors - In some patients, the administration of a non-steroidal anti-inflammatory agent including a selective COX-2 inhibitor can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing and thiazide diuretics. Therefore, when DIURIL and non-steroidal anti-inflammatory agents or selective COX-2 inhibitors are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
In some patients with compromised renal function (e.g., elderly patients or patients who are volume-depleted, including those on diuretic therapy) who are being treated with non-steroidal anti-inflammatory drugs, including selective COX-2 inhibitors, the co-administration of angiotensin II receptor antagonists or ACE inhibitors may result in a further deterioration of renal function, including possible acute renal failure. These effects are usually reversible.
These interactions should be considered in patients taking NSAIDs including selective COX-2 inhibitors concomitantly with diuretics and angiotensin II antagonists or ACE inhibitors. Therefore, the combination should be administered with caution, especially in the elderly.
Drug/Laboratory Test Interactions
Thiazides should be discontinued before carrying out tests for parathyroid function (see PRECAUTIONS, General).
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with chlorothiazide.
Chlorothiazide was not mutagenic in vitro in the Ames microbial mutagen test (using a maximum concentration of 5 mg/plate and Salmonella typhimurium strains TA98 and TA100) and was not mutagenic and did not induce mitotic nondisjunction in diploid-strains of Aspergillus nidulans.
Chlorothiazide had no adverse effects on fertility in female rats at doses up to 60 mg/kg/day and no adverse effects on fertility in male rats at doses up to 40 mg/kg/day. These doses are 1.5 and 1 times 1 the recommended maximum human dose, respectively, when compared on a body weight basis.
- 1 Calculations based on a human body weight of 50 kg
Pregnancy
Teratogenic Effects: Although reproduction studies performed with chlorothiazide doses of 50 mg/kg/day in rabbits, 60 mg/kg/day in rats and 500 mg/kg/day in mice revealed no external abnormalities of the fetus or impairment of growth and survival of the fetus due to chlorothiazide, such studies did not include complete examinations for visceral and skeletal abnormalities. It is not known whether chlorothiazide can cause fetal harm when administered to a pregnant woman; however, thiazides cross the placental barrier and appear in cord blood. DIURIL should be used during pregnancy only if clearly needed (see INDICATIONS AND USAGE).
Nursing Mothers
Because of the potential for serious adverse reactions in nursing infants from DIURIL, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
There are no well-controlled clinical trials in pediatric patients. Information on dosing in this age group is supported by evidence from empiric use in pediatric patients and published literature regarding the treatment of hypertension in such patients. (See DOSAGE AND ADMINISTRATION, Infants and Children.)
Geriatric Use
Clinical studies of DIURIL did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS).
-
ADVERSE REACTIONS
The following adverse reactions have been reported and, within each category, are listed in order of decreasing severity.
Body as a Whole: Weakness.
Cardiovascular: Hypotension, including orthostatic hypotension (may be aggravated by alcohol, barbiturates, narcotics or antihypertensive drugs).
Digestive: Pancreatitis, jaundice (intrahepatic cholestatic jaundice), diarrhea, vomiting, sialadenitis, cramping, constipation, gastric irritation, nausea, anorexia.
Hematologic: Aplastic anemia, agranulocytosis, leukopenia, hemolytic anemia, thrombocytopenia.
Hypersensitivity: Anaphylactic reactions, necrotizing angiitis (vasculitis and cutaneous vasculitis), respiratory distress including pneumonitis and pulmonary edema, photosensitivity, fever, urticaria, rash, purpura.
Metabolic: Electrolyte imbalance (see PRECAUTIONS), hyperglycemia, glycosuria, hyperuricemia.
Musculoskeletal: Muscle spasm.
Nervous System/Psychiatric: Vertigo, paresthesias, dizziness, headache, restlessness.
Renal: Renal failure, renal dysfunction, interstitial nephritis (see WARNINGS).
Skin: Erythema multiforme including Stevens-Johnson syndrome, exfoliative dermatitis including toxic epidermal necrolysis, alopecia.
Special Senses: Transient blurred vision, xanthopsia.
Urogenital: Impotence.
Whenever adverse reactions are moderate or severe, thiazide dosage should be reduced or therapy withdrawn.
To report SUSPECTED ADVERSE REACTIONS, contact Valeant Pharmaceuticals North America LLC at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
The most common signs and symptoms observed are those caused by electrolyte depletion (hypokalemia, hypochloremia, hyponatremia) and dehydration resulting from excessive diuresis. If digitalis has also been administered, hypokalemia may accentuate cardiac arrhythmias.
In the event of overdosage, symptomatic and supportive measures should be employed. Emesis should be induced or gastric lavage performed. Correct dehydration, electrolyte imbalance, hepatic coma and hypotension by established procedures. If required, give oxygen or artificial respiration for respiratory impairment.
The degree to which chlorothiazide sodium is removed by hemodialysis has not been established.
The oral LD50 of chlorothiazide is 8.5 g/kg, greater than 10 g/kg, and greater than 1 g/kg, in the mouse, rat and dog, respectively.
-
DOSAGE AND ADMINISTRATION
Therapy should be individualized according to patient response. Use the smallest dosage necessary to achieve the required response.
Adults
For Edema
The usual adult dosage is 500 mg to 1,000 mg (10 mL to 20 mL) once or twice a day. Many patients with edema respond to intermittent therapy, i.e., administration on alternate days or on three to five days each week. With an intermittent schedule, excessive response and the resulting undesirable electrolyte imbalance are less likely to occur.
Infants and Children
For Diuresis and For Control of Hypertension
The usual pediatric dosage is 5 mg to 10 mg per pound (10 mg/kg to 20 mg/kg) per day in single or two divided doses, not to exceed 375 mg per day (2.5 mL to 7.5 mL or ½ to 1½ teaspoonfuls of the oral suspension daily) in infants up to 2 years of age or 1,000 mg per day in children 2 to 12 years of age. In infants less than 6 months of age, doses up to 15 mg per pound (30 mg/kg) per day in two divided doses may be required (see PRECAUTIONS, Pediatric Use).
-
HOW SUPPLIED
DIURIL (chlorothiazide) Oral Suspension, 250 mg of chlorothiazide per 5 mL, is a yellow, creamy suspension, and is supplied as follows:
- NDC: 65649-311-12 250 mg/5mL 237 mL
Storage
DIURIL (chlorothiazide) Oral Suspension: Keep container tightly closed. Protect from freezing, –20°C (–4°F) and store at room temperature, 15° to 30°C (59° to 86°F).
Manufactured for:
Salix Pharmaceuticals, a division of Valeant Pharmaceuticals North America LLC,
Bridgewater, NJ 08807 USADIURIL is a registered trademark of Merck Sharp & Dohme Corp. used under license.
© Valeant Pharmaceuticals North America LLC
Rev. 07/2017
- 9516901 2201986 Rev 04-17 B 8F000 9L J1
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIURIL
chlorothiazide suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 65649-311 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLOROTHIAZIDE (UNII: 77W477J15H) (CHLOROTHIAZIDE - UNII:77W477J15H) CHLOROTHIAZIDE 250 mg in 5 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) BENZOIC ACID (UNII: 8SKN0B0MIM) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SUCROSE (UNII: C151H8M554) TRAGACANTH (UNII: 2944357O2O) Product Characteristics Color YELLOW (YELLOW) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 65649-311-12 237 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/15/1962 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA011870 02/15/1962 Labeler - Salix Pharmaceuticals, Inc (793108036) Establishment Name Address ID/FEI Business Operations Paddock Laboratories, LLC 967694121 MANUFACTURE(65649-311)
Trademark Results [DIURIL]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DIURIL 72051925 0676884 Live/Registered |
MERCK & CO., INC. 1958-05-19 |
 DIURIL 72029293 0660963 Dead/Expired |
MERCK & CO., INC. 1957-05-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.