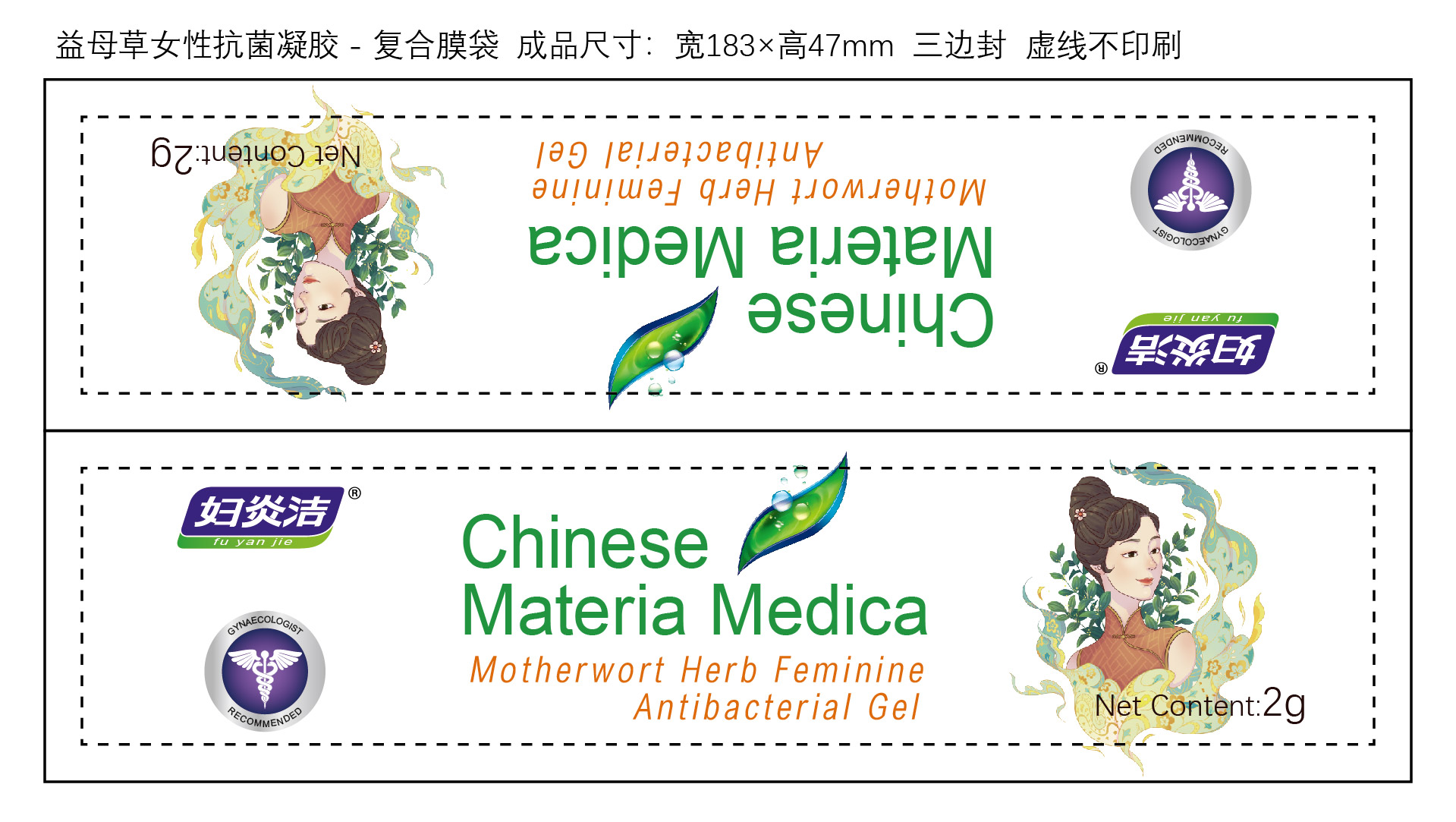
MOTHERWORT HERB FEMININE ANTIBACTERIAL GEL
MOTHERWORT HERB FEMININE ANTIBACTERIAL GELS by
Drug Labeling and Warnings
MOTHERWORT HERB FEMININE ANTIBACTERIAL GELS by is a Otc medication manufactured, distributed, or labeled by Jiangxi Renhetang pharmaceutical chain Co., Ltd., Jiangxi Kangmei medical and Health Products Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
MOTHERWORT HERB FEMININE ANTIBACTERIAL GELS- chlorhexidine diacetate gel
Jiangxi Renhetang pharmaceutical chain Co., Ltd.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
MOTHERWORT HERB FEMININE ANTIBACTERIAL GEL
Warnings
Disposable product. Please do not use if the package is broken.
This product is for external use only, not edible.
Avoid contact with eyes
This product cannot be used for STD prevention in sexual activity.
Women who are in menstrual, gestational or perinatal periods should be used with extreme caution.
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
1.Take a supine position, and support the hip with a cushion. Remove the socket at the top of the injector and use it as the plunger rod. Slowly insert the gel injector containing antibacterial gel into the deep area of vagina. Deliver the gel into the vagina by pushing the plunger.
2.If there is discomfort in the intimate area, squeeze a little of the gel and apply onto the area, then gently rub for a while.
3.While in use, it’s normal to see vaginal discharge, so it’s recommended to use panty liners meanwhile.
4.Better effect can be obtained if FUYANJIE WASH is used before applying this product.
Other information
This product can sterilize staphylococcus aureus, Escherichia coli, and Candida albicans.
Keep in sealed container, and place in a cool dry place.
| MOTHERWORT HERB FEMININE ANTIBACTERIAL GELS
chlorhexidine diacetate gel |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Jiangxi Renhetang pharmaceutical chain Co., Ltd. (410551226) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Kangmei medical and Health Products Co., Ltd | 528118770 | manufacture(81615-004) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.