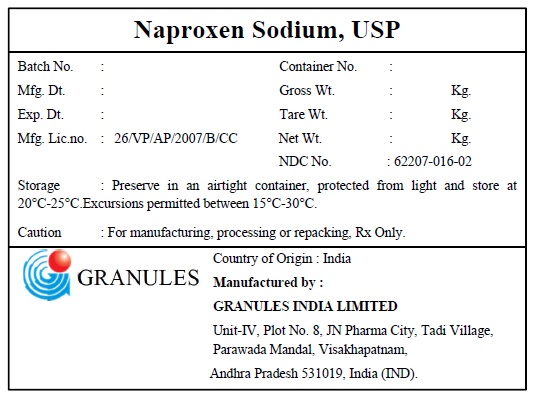
Naproxen Sodium by Granules India ltd / Granules India Ltd Naproxen sodium USP
Naproxen Sodium by
Drug Labeling and Warnings
Naproxen Sodium by is a Other medication manufactured, distributed, or labeled by Granules India ltd, Granules India Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
NAPROXEN SODIUM- naproxen sodium powder
Granules India ltd
----------
Naproxen sodium USP
| NAPROXEN SODIUM
naproxen sodium powder |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Granules India ltd (915000087) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Granules India Ltd | 650376275 | api manufacture(62207-016) , analysis(62207-016) , pack(62207-016) | |
Revised: 6/2023
Document Id: ff015d09-b076-30da-e053-6294a90afd26
Set id: bf5d44d0-773a-ffef-e053-2995a90afa88
Version: 3
Effective Time: 20230626
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.