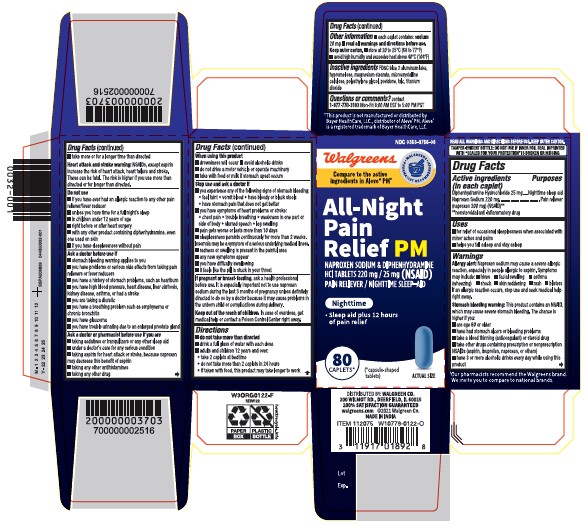
NAPROXEN SODIUM PM Naproxen Sodium & Diphenhydramine HCl Tablets 220mg/25mg (NSAID) Pain Reliever/Nighttime sleep aid Sleep aid plus 12 hours pain relieving
Naproxen Sodium Diphenhydramine HCl by
Drug Labeling and Warnings
Naproxen Sodium Diphenhydramine HCl by is a Otc medication manufactured, distributed, or labeled by WALGREEN CO.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
NAPROXEN SODIUM DIPHENHYDRAMINE HCL- naproxen sodium diphenhydramine hcl tablet, film coated
WALGREEN CO.
----------
NAPROXEN SODIUM PM
Naproxen Sodium & Diphenhydramine HCl Tablets 220mg/25mg (NSAID)
Pain Reliever/Nighttime sleep aid
Sleep aid plus 12 hours pain relieving
Active ingredients
(in each caplet)
Diphenhydramine Hydrochloride 25 mg
Naproxen Sodium 220 mg (naproxen 200 mg) (NSAID)*
Uses
for relief of occasional sleeplessness when associated with minor aches and pains
helps you fall asleep and stay asleep
Allergy alert
Naproxen sodium may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
hives facial swelling asthma (wheezing) shock skin reddening rash blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
are age 60 or older
have had stomach ulcers or bleeding problems
take a blood thinning (anticoagulant) or steroid drug
take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen or others)
have 3 or more alcoholic drinks every day while using this product
take more or for a longer time than directed
Heart attack and stroke warning
NSAIDs, except aspirin increase the risk of heart attack, heart failure and stroke. These can be fatal. The risk is higher if use more than directed or for longer than directed.
Do not use
if you have ever had an allergic reaction to any other pain reliever/fever reducer
unless you have time for a full night’s sleep
in children under 12 years of age
right before or after heart surgery
with any other product containing diphenhydramine, even one used on skin
if you have sleeplessness without pain
Ask a doctor before use if
stomach bleeding warning applies to you
you have problems or serious side effects from taking pain relievers or fever reducers
you have a history of stomach problems, such as heartburn
you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma or had a stroke
you are taking a diuretic
you have a breathing problem such as emphysema or chronic bronchitis
you have glaucoma
you have trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are
taking sedatives or tranquilizers, or any other sleep-aid
under a doctor's care for any serious condition
taking aspirin for heart attack or stroke, because naproxen may decrease this benefit of aspirin
taking any other antihistamines
taking any other drug
When using this product
drowsiness will occur
avoid alcoholic drinks
do not drive a motor vehicle or operate machinery
take with food or milk if stomach upset occurs
Stop use and ask a doctor if
you experience any of the following signs of stomach bleeding: feel faint vomit blood have bloody or black stools
have stomach pain that does not get better
you have symptoms heart problems or stroke:
chest pain trouble breathing weakness in one part or side of body slurred speech leg swelling
pain gets worse or lasts more than 10 days
sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
redness or swelling is present in the painful area
any new symptoms appear
you have difficulty swallowing
it feels like the pill is stuck in your throat
If pregnant or breast-feeding,
ask a health professional before use. It is especially important not to use naproxen sodium during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause problems in the unborn child or complications during delivery
Keep out of reach of children
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
do not take more than directed
drink a full glass of water with each dose
adults and children 12 years and over: take 2 caplets at bedtime
do not take more than 2 caplets in 24 hours
if taken with food, this product may take longer to work
Other information
each caplet contains:
sodium 20 mg
read all warnings and directions before use. keep outer carton.
store at 20 to 25°C (68 to 77°F)
avoid high humidity and excessive heat above 40°C (104°F)
| NAPROXEN SODIUM DIPHENHYDRAMINE HCL
naproxen sodium diphenhydramine hcl tablet, film coated |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - WALGREEN CO. (008965063) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.