SunMark Children Allergy Relief Dye Free Cherry
children allergy relief dye free cherry by
Drug Labeling and Warnings
children allergy relief dye free cherry by is a Otc medication manufactured, distributed, or labeled by Strategic Sourcing Services LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
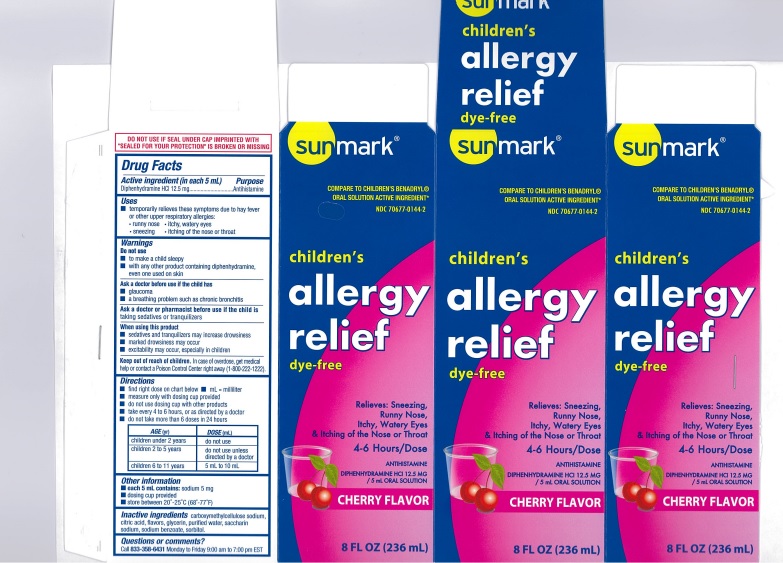
CHILDREN ALLERGY RELIEF DYE FREE CHERRY- diphenhydramine hcl solution
Strategic Sourcing Services LLC
----------
SunMark Children Allergy Relief Dye Free Cherry
USES
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
DO NOT USE
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
WHEN USING THIS PRODUCT
- sedatives and tranquilizers may increase drowsiness
- marked drowsiness may occur
- excitability may occur, especially in children
KEEP OUT OF REACH OF CHILDREN
In case of overdose, get medical help or contact a poison control center right away
(1-800-222-1222).
DIRECTIONS
- find right dose on chart below
- mL=milliliter
- measure only with dosing cup provided
- do not use dosing cup with other products
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 doses in 24 hours
| AGE(yr) | DOSE(mL) |
| children under 2 years | do not use |
| children 2 to 5 years | do not use unless directed by a doctor |
| children 6 to 11 years | 5 mL to 10 mL |
OTHER INFORMATION
- each 5 mL contains: sodium 5 mg
- dosing cup provided
- store between 20 - 25oC (68o- 77oF)
INACTIVE INGREDIENTS
carboxymethylcellulose sodium, citric acid, flavors, glycerin, purified water, saccharin sodium, sodium benzoate, sorbitol.
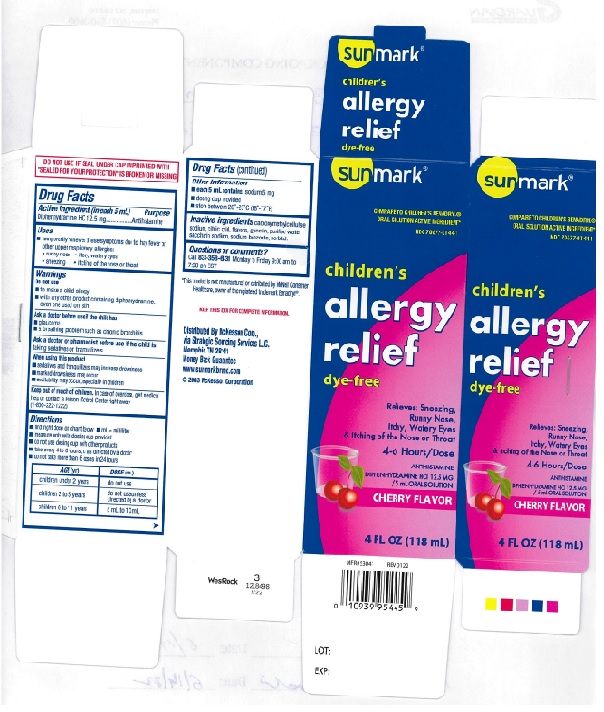
PRINCIPAL DISPLAY PANEL
SUNMARK®
COMPARE TO CHILDREN'S BENADRYL®
ORAL SOLUTION ACTIVE INGREDIENT*
NDC: 70677-0144-1
children's allergy relief dye-free
Relieves: Sneezing,
Runny Nose,
Itchy, Watery Eyes
& Itching of the Nose or Throat
4-6 Hours/Dose
ANTIHISTAMINE
DIPHENHYDRAMINE HCl 12.5 MG/5 mL ORAL SOLUTION
CHERRY FLAVOR
4 FL OZ(118 mL)
| CHILDREN ALLERGY RELIEF DYE FREE CHERRY
diphenhydramine hcl solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Strategic Sourcing Services LLC (116956644) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Guardian Drug Company | 119210276 | manufacture(70677-0144) | |