OXYCODONE HYDROCHLORIDE solution
Oxycodone Hydrochloride by
Drug Labeling and Warnings
Oxycodone Hydrochloride by is a Prescription medication manufactured, distributed, or labeled by Safecor Health, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Oxycodone Hydrochloride Oral Solution safely and effectively. See full prescribing information for Oxycodone Hydrochloride Oral Solution.
Oxycodone Hydrochloride Oral Solution CII
Initial U.S. Approval: 1950WARNING: RISK OF MEDICATION ERRORS
See full prescribing information for complete boxed warning.Oxycodone Hydrochloride Oral Solution, 100 mg/5 mL (20 mg/mL) is available in one concentration, as 20 mg oxycodone hydrochloride per mL of solution. The 100 mg/5 mL (20 mg/mL) concentration is indicated for use in opioid-tolerant patients only. Take care to avoid dosing errors due to confusion between between mg and mL, and other oxycodone solutions with different concentrations, which could result in accidental overdose and death. (5.1)
Keep Oxycodone Hydrochloride Oral Solution out of the reach of children.INDICATIONS AND USAGE
Oxycodone Hydrochloride 20 mg per mL solution is an opioid agonist indicated for the management of moderate to severe acute and chronic pain in opioid-tolerant patients. (1)
DOSAGE AND ADMINISTRATION
- Oxycodone Hydrochloride Oral Solution: 5 to 15 mg every 4 to 6 hours as needed. (2.2)
DOSAGE FORMS AND STRENGTHS
Oral Solution 100 mg/5 mL (20 mg/mL): Each mL contains 20 mg of oxycodone hydrochloride. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Risk of Medication Errors: Use caution when prescribing, dispensing, and administering to avoid dosing errors due to confusion between different concentrations and between mg and mL, which could result in accidental overdose and death. (5.1)
- Respiratory depression: Increased risk in elderly, debilitated patients, those suffering from conditions accompanied by hypoxia, hypercapnia, or upper airway obstruction. (5.2)
- Misuse, Abuse and Diversion: Oxycodone hydrochloride is a Schedule II controlled substance with an abuse liability similar to other opioids. (5.3)
- CNS effects: Additive CNS depressive effects when used in conjunction with alcohol, other opioids, or illicit drugs. (5.4)
- Elevation of intracranial pressure: May be markedly exaggerated in the presence of head injury, other intracranial lesions. (5.5)
- Hypotensive effect: Increased risk with compromised ability to maintain blood pressure. (5.6)
- Prolonged gastric obstruction: In patients with gastrointestinal obstruction, especially paralytic ileus. (5.7)
- Sphincter of Oddi spasm and diminished biliary/pancreatic secretions. Increased risk with biliary tract disease. (5.8)
- Special Risk Groups: Use with caution and in reduced dosages in patients with severe renal or hepatic impairment, Addison's disease, hypothyroidism, prostatic hypertrophy, or urethral stricture, elderly, CNS depression, toxic psychosis, acute alcoholism and delirium tremens, may aggravate or induce seizures. (5.9)
- Impaired mental/physical abilities: Caution must be used with potentially hazardous activities. (5.10)
- Concomitant use of CYP3A4 inhibitors may increase opioid effects (5.11)
ADVERSE REACTIONS
Most common adverse reactions are nausea, constipation, vomiting, headache, pruritus, insomnia, dizziness, asthenia, and somnolence. (6)
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088, www.fda.gov/medwatch or call Pharm-Olam at 1-866-511-6754.
DRUG INTERACTIONS
- CNS depressants: Increased risk of respiratory depression, hypotension, profound sedation, or coma. Use with caution in reduced dosages. (7.1)
- Muscle relaxants: Enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. (7.2)
- Mixed agonist/antagonist opioid analgesics (i.e. pentazocine, nalbuphine, and butorphanol): May reduce the analgesic effect and/or may precipitate withdrawal symptoms. (7.3)
- The CYP3A4 enzyme plays a major role in the metabolism of oxycodone, drugs that inhibit CYP3A4 activity may cause decreased clearance of oxycodone which could lead to an increase in oxycodone plasma concentrations. (7.4)
- Monoamine oxidase inhibitors (MAOIs): No specific interaction has been observed but caution in the use of Oxycodone Hydrochloride in patients taking this class of drugs is appropriate. (7.5)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2013
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: RISK OF MEDICATION ERRORS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Individualization of Dosage
2.2 Conversion to Oral Oxycodone Hydrochloride
2.3 Maintenance of Therapy
2.4 Cessation of Therapy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Medication Errors
5.2 Respiratory Depression
5.3 Misuse, Abuse and Diversion of Opioids
5.4 Interactions with Alcohol and Drugs of Abuse
5.5 Use In Head Injury and Increased Intracranial Pressure
5.6 Hypotensive Effect
5.7 Gastrointestinal Effects
5.8 Use In Pancreatic/Biliary Tract Disease
5.9 Special Risk Groups
5.10 Driving and Operating Machinery
5.11 Cytochrome P450 3A4 Inhibitors and Inducers
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 CNS Depressants
7.2 Muscle Relaxants
7.3 Mixed Agonist/Antagonist Opioid Analgesics
7.4 Agents Affecting Cytochrome P450 Enzymes
7.5 Monoamine Oxidase Inhibitors (MAOIs)
7.6 Anticholinergics
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
10.1 Symptoms
10.2 Treatment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: RISK OF MEDICATION ERRORS
Oxycodone Hydrochloride Oral Solution is available as a 100 mg/5 mL (20 mg/mL) concentration and is indicated for use in opioid-tolerant patients only.
Take care when prescribing and administering Oxycodone Hydrochloride Oral Solution to avoid dosing errors due to confusion between mg and mL, and other oxycodone solutions with different concentrations, which could result in accidental overdose and death. Take care to ensure the proper dose is communicated and dispensed.
Keep Oxycodone Hydrochloride Oral Solution out of the reach of children. In case of accidental ingestion, seek emergency medical help immediately. -
1 INDICATIONS AND USAGE
Oxycodone hydrochloride oral solution 100 mg/5 mL (20 mg/mL) is an opioid analgesic indicated for the management of moderate to severe acute and chronic pain in opioid-tolerant patients.
Oxycodone hydrochloride oral solution 100 mg/5 mL (20 mg/mL) may cause fatal respiratory depression when administered to patients not previously exposed to opioids. Patients considered to be opioid tolerant are those who are taking at least 30 mg of oral oxycodone per day, or at least 60 mg oral morphine per day, or at least 12 mg hydromorphone per day, or an equianalgesic dose of another opioid, for a week or longer. -
2 DOSAGE AND ADMINISTRATION
Take care when prescribing and administering Oxycodone Hydrochloride Oral Solution to avoid dosing errors due to confusion between mg and mL, which could result in accidental overdose and death. Take care to ensure the proper dose is communicated and dispensed. When writing prescriptions, include both the total dose in mg and the total dose in volume. Always use the enclosed calibrated oral syringe when administering Oxycodone Hydrochloride Oral Solution, 100 mg/5 mL (20 mg/mL), to ensure the dose is measured and administered accurately.
Selection of patients for treatment with oxycodone hydrochloride should be governed by the same principles that apply to the use of similar opioid analgesics. Individualize treatment in every case, using non-opioid analgesics, opioids on an as needed basis and/or combination products, and chronic opioid therapy in a progressive plan of pain management such as outlined by the World Health Organization, the Agency for Healthcare Research and Quality, and the American Pain Society.2.1 Individualization of Dosage
As with any opioid drug product, adjust the dosing regimen for each patient individually, taking into account the patient's prior analgesic treatment experience. In the selection of the initial dose of oxycodone hydrochloride, give attention to the following:
- the total daily dose, potency and specific characteristics of the opioid the patient has been taking previously;
- the reliability of the relative potency estimate used to calculate the equivalent oxycodone hydrochloride dose needed;
- the patient's degree of opioid tolerance;
- the general condition and medical status of the patient;
- concurrent medications;
- the type and severity of the patient's pain;
- risk factors for abuse, addiction or diversion, including a prior history of abuse, addiction or diversion.
The following dosing recommendations, therefore, can only be considered suggested approaches to what is actually a series of clinical decisions over time in the management of the pain of each individual patient.
Continual re-evaluation of the patient receiving oxycodone hydrochloride is important, with special attention to the maintenance of pain control and the relative incidence of side effects associated with therapy. During chronic therapy, especially for non-cancer-related pain, periodically re-assess the continued need for the use of opioid analgesics.
During periods of changing analgesic requirements, including initial titration, frequent contact is recommended between physician, other members of the healthcare team, the patient, and the caregiver/family.2.2 Conversion to Oral Oxycodone Hydrochloride
The 100 mg/5 mL (20 mg/mL) oral solution formulation is for use in opioid-tolerant patients only who have already been receiving opioid therapy. Use this strength only for patients that have already been titrated to a stable analgesic regimen using lower strengths of oxycodone hydrochloride and who can benefit from use of a smaller volume of oral solution.
There is inter-patient variability in the potency of opioid drugs and opioid formulations.
Therefore, a conservative approach is advised when determining the total daily dose of Oxycodone Hydrochloride. It is better to underestimate a patient's 24-hour oral Oxycodone Hydrochloride dose and make available rescue medication than to overestimate the 24-hour oral Oxycodone Hydrochloride dose and manage an adverse experience of overdose.
Consider the following general points regarding opioid conversions.Conversion From Non-Oxycodone Opioids to Oral Oxycodone Hydrochloride.
In converting patients from other opioids to oxycodone hydrochloride, close observation and adjustment of dosage based upon the patient's response to oxycodone hydrochloride is imperative. Physicians and other healthcare professionals are advised to refer to published relative potency information, keeping in mind that conversion ratios are only approximate.
Conversion From Controlled-Release Oral Oxycodone to Oral Oxycodone Hydrochloride.
The relative bioavailability of Oxycodone Hydrochloride Oral Solution compound to controlled-release oxycodone is unknown. The extended duration of release of oxycodone hydrochloride from controlled-release tablets results in reduced maximum and increased minimum plasma oxycodone hydrochloride concentrations than with shorter acting oxycodone hydrochloride products. Conversion from controlled-release tablets could lead to excessive sedation at peak serum levels. Therefore, dosage adjustment with close observation is necessary.
Conversion From Oral Oxycodone Hydrochloride to Controlled-Release Oral Oxycodone
The relative bioavailability of Oxycodone Hydrochloride Oral Solution compared to controlled-release oxycodone is unknown, so conversion to controlled-release tablets must be accompanied by close observation for signs of excessive sedation.
2.3 Maintenance of Therapy
Continual re-evaluation of the patient receiving oxycodone hydrochloride is important, with special attention to the maintenance of pain management and the relative incidence of side effects associated with therapy. If the level of pain increases, effort should be made to identify the source of increased pain, while adjusting the dose as described above to decrease the level of pain. During chronic therapy, especially for non-cancer-related pain (or pain associated with other terminal illnesses), periodically reassess the continued need for the use of opioid analgesics.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Oxycodone hydrochloride is contraindicated in patients with respiratory depression in the absence of resuscitative equipment.
Oxycodone hydrochloride is contraindicated in any patient who has or is suspected of having paralytic ileus.
Oxycodone hydrochloride is contraindicated in patients with acute or severe bronchial asthma or hypercarbia.
Oxycodone hydrochloride is contraindicated in patients with known hypersensitivity to oxycodone, oxycodone salts, or any components of the product. -
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Medication Errors
Oxycodone Hydrochloride Oral Solution is available in 100 mg/5 mL (20 mg/mL) concentration. Use caution when prescribing, dispensing, and administering Oxycodone Hydrochloride Oral Solution to avoid dosing errors due to confusion between mg and mL, and other oxycodone solutions with different concentrations, which could result in accidental overdose and death. Use caution to ensure the dose is communicated clearly and dispensed accurately. Always use the enclosed calibrated oral syringe when administering Oxycodone Hydrochloride Oral Solution 100 mg/5 mL (20 mg/mL) to ensure the dose is measured and administered accurately.
5.2 Respiratory Depression
Respiratory depression is the primary risk of oxycodone hydrochloride. Respiratory depression occurs more frequently in elderly or debilitated patients and in those suffering from conditions accompanied by hypoxia, hypercapnia, or upper airway obstruction, in whom even moderate therapeutic doses may significantly decrease pulmonary ventilation.
Use oxycodone hydrochloride with extreme caution in patients with chronic obstructive pulmonary disease or cor pulmonale and in patients having a substantially decreased respiratory reserve (e.g., severe kyphoscoliosis), hypoxia, hypercapnia, or preexisting respiratory depression. In such patients, even usual therapeutic doses of oxycodone hydrochloride may increase airway resistance and decrease respiratory drive to the point of apnea. Consider alternative non-opioid analgesics, and use oxycodone hydrochloride only under careful medical supervision at the lowest effective dose in such patients.
Oxycodone hydrochloride 100 mg/5 mL (20 mg/mL) oral solution is for use in opioid-tolerant patients only. Administration of this formulation may cause fatal respiratory depression when administered to patients who are not tolerant to the respiratory depressant effects of opioids.5.3 Misuse, Abuse and Diversion of Opioids
Oxycodone hydrochloride is an opioid agonist and a Schedule II controlled substance. Such drugs are sought by drug abusers and people with addiction disorders. Diversion of Schedule II products is an act subject to criminal penalty.
Oxycodone hydrochloride can be abused in a manner similar to other opioid agonists, legal or illicit. This should be considered when prescribing or dispensing oxycodone hydrochloride in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion.
Oxycodone hydrochloride may be abused by crushing, chewing, snorting or injecting the product. These practices pose a significant risk to the abuser that could result in overdose and death. [See DRUG ABUSE AND DEPENDENCE (9)]
Concerns about abuse, addiction, and diversion should not prevent the proper management of pain. Healthcare professionals should contact their State Professional Licensing Board or State Controlled Substances Authority for information on how to prevent and detect abuse or diversion of this product.5.4 Interactions with Alcohol and Drugs of Abuse
Oxycodone hydrochloride may be expected to have additive effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression because respiratory depression, hypotension, profound sedation, coma or death may result.
5.5 Use In Head Injury and Increased Intracranial Pressure
In the presence of head injury, intracranial lesions or a preexisting increase in intracranial pressure, the possible respiratory depressant effects of oxycodone hydrochloride and its potential to elevate cerebrospinal fluid pressure (resulting from vasodilation following CO2 retention) may be markedly exaggerated. Furthermore, oxycodone hydrochloride can produce effects on pupillary response and consciousness, which may obscure neurologic signs of further increases in intracranial pressure in patients with head injuries.
5.6 Hypotensive Effect
Oxycodone hydrochloride may cause severe hypotension in an individual whose ability to maintain blood pressure has already been compromised by a depleted blood volume or concurrent administration of drugs such as phenothiazines or general anesthetics. Oxycodone hydrochloride may produce orthostatic hypotension and syncope in ambulatory patients.
Administer oxycodone hydrochloride with caution to patients in circulatory shock, as vasodilation produced by the drug may further reduce cardiac output and blood pressure.5.7 Gastrointestinal Effects
Do not administer oxycodone hydrochloride to patients with gastrointestinal obstruction, especially paralytic ileus because oxycodone hydrochloride diminishes propulsive peristaltic waves in the gastrointestinal tract and may prolong the obstruction.
The administration of oxycodone hydrochloride may obscure the diagnosis or clinical course in patients with acute abdominal condition.5.8 Use In Pancreatic/Biliary Tract Disease
Use oxycodone hydrochloride with caution in patients with biliary tract disease, including acute pancreatitis, as oxycodone hydrochloride may cause spasm of the sphincter of Oddi and diminish biliary and pancreatic secretions.
5.9 Special Risk Groups
Use oxycodone hydrochloride with caution and in reduced dosages in patients with severe renal or hepatic impairment, Addison's disease, hypothyroidism, prostatic hypertrophy, or urethral stricture, and in elderly or debilitated patients. [See USE IN SPECIFIC POPULATIONS (8.5)] Exercise caution in the administration of oxycodone hydrochloride to patients with CNS depression, toxic psychosis, acute alcoholism and delirium tremens. All opioids may aggravate convulsions in patients with convulsive disorders, and all opioids may induce or aggravate seizures in some clinical settings.
Keep Oxycodone Hydrochloride Oral Solution out of the reach of children. In case of accidental ingestion, seek emergency medical help immediately.5.10 Driving and Operating Machinery
Caution patients that oxycodone hydrochloride could impair the mental and/or physical abilities needed to perform potentially hazardous activities such as driving a car or operating machinery.
Caution patients about the potential combined effects of oxycodone hydrochloride with other CNS depressants, including other opioids, phenothiazines, sedative/hypnotics and alcohol. [See DRUG INTERACTIONS (7)]5.11 Cytochrome P450 3A4 Inhibitors and Inducers
Since the CYP3A4 isoenzyme plays a major role in the metabolism of oxycodone, drugs that alter CYP3A4 activity may cause changes in clearance of oxycodone which could lead to changes in oxycodone plasma concentrations. The expected clinical results with CYP3A4 inhibitors would be an increase in oxycodone plasma concentrations and possibly increased or prolonged opioid effects. The expected clinical results with CYP3A4 inducers would be a decrease in oxycodone plasma concentrations, lack of efficacy or, possibly, development of an abstinence syndrome in a patient who had developed physical dependence to oxycodone.
If co-administration is necessary, caution is advised when initiating oxycodone treatment in patients currently taking, or discontinuing, CYP3A4 inhibitors or inducers. Evaluate these patients at frequent intervals and consider dose adjustments until stable drug effects are achieved. [Drug Interactions (7.4) and Clinical Pharmacology (12.3)] -
6 ADVERSE REACTIONS
Serious adverse reactions that may be associated with oxycodone therapy in clinical use are those observed with other opioid analgesics and include: respiratory depression, respiratory arrest, circulatory depression, cardiac arrest, hypotension, and/or shock.
The common adverse events seen on initiation of therapy with oxycodone are also typical opioid side effects. These events are dose dependent, and their frequency depends on the clinical setting, the patient's level of opioid tolerance, and host factors specific to the individual. They should be expected and managed as a part of opioid therapy. The most frequent adverse events include nausea, constipation, vomiting, headache, and pruritus.
The frequency of adverse events during initiation of opioid therapy may be minimized by careful individualization of starting dosage, slow titration and the avoidance of large rapid swings in plasma concentration of the opioid. Many of these common adverse events may abate as therapy is continued and some degree of tolerance is developed, but others may be expected to remain throughout therapy.
In all patients for whom dosing information was available (n=191) from the open-label and double-blind studies involving immediate-release oxycodone, the following adverse events were recorded in oxycodone treated patients with an incidence ≥ 3%. In descending order of frequency they were: nausea, constipation, vomiting, headache, pruritus, insomnia, dizziness, asthenia, and somnolence.
The following adverse experiences occurred in less than 3% of patients involved in clinical trials with oxycodone:
Body as a Whole: abdominal pain, accidental injury, allergic reaction, back pain, chills and fever, fever, flu syndrome, infection, neck pain, pain, photosensitivity reaction, and sepsis.
Cardiovascular: deep thrombophlebitis, heart failure, hemorrhage, hypotension, migraine, palpitation, and tachycardia.
Digestive: anorexia, diarrhea, dyspepsia, dysphagia, gingivitis, glossitis, and nausea and vomiting.
Hemic and Lymphatic: anemia and leukopenia.
Metabolic and Nutritional: edema, gout, hyperglycemia, iron deficiency anemia and peripheral edema.
Musculoskeletal: arthralgia, arthritis, bone pain, myalgia and pathological fracture.
Nervous: agitation, anxiety, confusion, dry mouth, hypertonia, hypesthesia, nervousness, neuralgia, personality disorder, tremor, and vasodilation.
Respiratory: bronchitis, cough increased, dyspnea, epistaxis, laryngismus, lung disorder, pharyngitis, rhinitis, and sinusitis.
Skin and Appendages: herpes simplex, rash, sweating, and urticaria.
Special Senses: amblyopia.
Urogenital: urinary tract infection -
7 DRUG INTERACTIONS
7.1 CNS Depressants
Other central nervous system (CNS) depressants including sedatives, hypnotics, general anesthetics, antiemetics, phenothiazines, or other tranquilizers or alcohol increases the risk of respiratory depression, hypotension, profound sedation, or coma. Use oxycodone hydrochloride with caution and in reduced dosages in patients taking these agents.
7.2 Muscle Relaxants
Oxycodone hydrochloride may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression.
7.3 Mixed Agonist/Antagonist Opioid Analgesics
Do not administer mixed agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, butorphanol and buprenorphine) to patients who have received or are receiving a course of therapy with a pure opioid agonist analgesic such as oxycodone hydrochloride. In these patients, mixed agonist/antagonist analgesics may reduce the analgesic effect and/or may precipitate withdrawal symptoms.
7.4 Agents Affecting Cytochrome P450 Enzymes
CYP3A4 Inhibitors
A published study showed that the co-administration with voriconazole, a CYP3A4 inhibitor, significantly increased the plasma concentrations of oxycodone. Inhibition of CYP3A4 activity by its inhibitors, such as macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), and protease inhibitors (e.g., ritonavir), may prolong opioid effects. If co-administration is necessary, caution is advised when initiating therapy with, currently taking, or discontinuing CYP3A4 inhibitors. Evaluate these patients at frequent intervals and consider dose adjustments until stable drug effects are achieved. [see Clinical Pharmacology (12.3)]
CYP3A4 Inducers
A published study showed that the co-administration of rifampin, a drug metabolizing enzyme inducer, significantly decreased plasma oxycodone concentrations. Induction of CYP3A4 activity by its inducers, such as rifampin, carbamazepine, and phenytoin, may lead to a lack of efficacy or, possibly, development of an abstinence syndrome in a patient who had developed physical dependence to oxycodone. If co-administration is necessary, caution is advised when initiating therapy with, currently taking, or discontinuing CYP3A4 inducers. Evaluate these patients at frequent intervals and consider dose adjustments until stable drug effects are achieved. [see Clinical Pharmacology (12.3)]
CYP2D6 Inhibitors
Oxycodone is metabolized in part to oxymorphone via the cytochrome p450 isoenzyme CYP2D6. While this pathway may be blocked by a variety of drugs (e.g., certain cardiovascular drugs and antidepressants), such blockade has not yet been shown to be of clinical significance with this agent. However, clinicians should be aware of this possible interaction.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category B: There are no adequate and well-controlled studies of oxycodone use during pregnancy. Based on limited human data in the literature, oxycodone does not appear to increase the risk of congenital malformations. Because animal reproduction studies are not always predictive of human response, oxycodone should be used during pregnancy only if clearly needed.
Teratogenic effects
Reproduction studies in Sprague-Dawley rats and New Zealand rabbits revealed that when oxycodone was administered orally at doses up to 16 mg/kg (approximately 2 times the daily oral dose of 90 mg for adults on a mg/m2 basis) and 25 mg/kg (approximately 5 times the daily oral dose of 90 mg on a mg/m2 basis), respectively, was not teratogenic or embryo-fetal toxic.
8.2 Labor and Delivery
Opioids cross the placenta and may produce respiratory depression and psycho-physiologic effects in neonates. Oxycodone hydrochloride is not recommended for use in women during and immediately prior to labor. Occasionally, opioid analgesics may prolong labor through actions which temporarily reduce the strength, duration and frequency of uterine contractions. However this effect is not consistent and may be offset by an increased rate of cervical dilatation, which tends to shorten labor. Closely observe neonates whose mothers received opioid analgesics during labor for signs of respiratory depression. Have a specific opioid antagonist, such as naloxone or nalmefene, available for reversal of opioid-induced respiratory depression in the neonate.
8.3 Nursing Mothers
Low levels of oxycodone have been detected in maternal milk. The amount of oxycodone hydrochloride delivered to the infant depends on the plasma concentration of the mother, the amount of milk ingested by the infant, and the extent of first-pass metabolism. Because of the potential for serious adverse reactions in nursing infants from oxycodone hydrochloride including respiratory depression, sedation and possibly withdrawal symptoms, upon cessation of oxycodone hydrochloride administration to the mother, decide whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness and the pharmacokinetics of Oxycodone Hydrochloride Oral Solution in pediatric patients below the age of 18 have not been established.
8.5 Geriatric Use
Elderly patients (aged 65 years or older) may have increased sensitivity to oxycodone hydrochloride. In general, use caution when selecting a dose for an elderly patient, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
8.6 Hepatic Impairment
Since oxycodone is extensively metabolized, its clearance may be decreased in patients with hepatic impairment. Follow a conservative approach to dose initiation in patients with hepatic impairment, monitor patients closely and adjust the dose based on clinical response.
8.7 Renal Impairment
Information from oxycodone tablets indicate that patients with renal impairment (defined as a creatinine clearance <60 mL/min) had higher plasma concentrations of oxycodone than subjects with normal renal function. Use a conservative approach to dose initiation in patients with renal impairment, monitor patients closely and adjust the dose based on clinical response.
-
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Oxycodone hydrochloride is a mu-agonist opioid and is a Schedule II controlled substance. Oxycodone hydrochloride, like other opioids used in analgesia, can be abused and is subject to criminal diversion.
9.2 Abuse
Drug addiction is characterized by compulsive use, use for non-medical purposes, and continued use despite harm or risk of harm. Drug addiction is a treatable disease, utilizing a multi-disciplinary approach, but relapse is common.
"Drug-seeking" behavior is very common in addicts and drug abusers. Drug seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated "loss" of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). "Doctor shopping" to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may not be accompanied by concurrent tolerance and symptoms of physical dependence. The converse is also true. In addition, abuse of opioids can occur in the absence of true addiction and is characterized by misuse for nonmedical purposes, often in combination with other psychoactive substances. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests is strongly advised.
Oxycodone Hydrochloride is intended for oral use only. Abuse of oxycodone hydrochloride poses a risk of overdose and death. The risk is increased with concurrent abuse of alcohol and other substances. Parenteral drug abuse is commonly associated with transmission of infectious diseases such as hepatitis and HIV.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal symptoms. [See USE IN SPECIFIC POPULATIONS (8.2)]9.3 Dependence
Tolerance is the need for increasing doses of opioids to maintain a defined effect such as analgesia (in the absence of disease progression or other external factors). Physical dependence is manifested by withdrawal symptoms after abrupt discontinuation of a drug or upon administration of an antagonist. Physical dependence and tolerance are not unusual during chronic opioid therapy.
The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
In general, taper opioids rather than abruptly discontinue. [See DOSAGE AND ADMINISTRATION (2.4)] -
10 OVERDOSAGE
10.1 Symptoms
Acute overdosage with oxycodone hydrochloride is manifested by respiratory depression (a decrease in respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis), extreme somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, in some cases, pulmonary edema, bradycardia, hypotension, cardiac arrest and death.
Oxycodone hydrochloride may cause miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origin may produce similar findings). Marked mydriasis rather than miosis may be seen with hypoxia in overdose situations. [See CLINICAL PHARMACOLOGY (12)]10.2 Treatment
Give primary attention to re-establishment of a patent airway and institution of assisted or controlled ventilation. Employ supportive measures (including oxygen and vasopressors) in the management of circulatory shock and pulmonary edema accompanying overdose as indicated. Cardiac arrest or arrhythmias may require cardiac massage or defibrillation.
The pure opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression resulting from opioid overdose. Since the duration of reversal is expected to be less than the duration of action of oxycodone hydrochloride, carefully monitor the patient until spontaneous respiration is reliably re-established. If the response to opioid antagonists is suboptimal or only brief in nature, administer additional antagonist as directed by the manufacturer of the product.
Do not administer opioid antagonists in the absence of clinically significant respiratory or circulatory depression secondary to oxycodone overdose. Administer such agents cautiously to persons who are known, or suspected to be physically dependent on oxycodone. In such cases, an abrupt or complete reversal of opioid effects may precipitate an acute abstinence syndrome. In an individual physically dependent on opioids, administration of the usual dose of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal symptoms experienced will depend on the degree of physical dependence and the dose of the antagonist administered. Reserve use of an opioid antagonist for cases where such treatment is clearly needed. If it is necessary to treat serious respiratory depression in the physically dependent patient, initiate administration of the antagonist with care and titrate with smaller than usual doses. -
11 DESCRIPTION
Oxycodone hydrochloride is a white, odorless crystalline powder derived from the opium alkaloid, thebaine. It is soluble in water and slightly soluble in alcohol.
Chemically, oxycodone hydrochloride is (5R,9R,13S,14S)-4, 5α-epoxy-14-hydroxy-3- methoxy-17-methylmorphinan-6-one hydrochloride with a molecular mass of 351.82.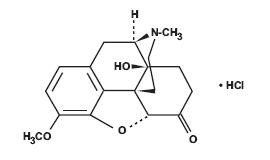
100 mg/5 mL (20 mg/mL): Each 1 mL of oral yellow solution contains 20 mg of oxycodone hydrochloride, USP and the following inactive ingredients: citric acid anhydrous, D&C Yellow #10, natural/artificial berry flavor, purified water, sodium citrate dihydrate, sodium benzoate, saccharin sodium, sorbitol.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Oxycodone hydrochloride, a pure opioid agonist, is relatively selective for the mu receptor, although it can interact with other opioid receptors at higher doses. In addition to analgesia, the widely diverse effects of oxycodone hydrochloride include drowsiness, changes in mood, respiratory depression, decreased gastrointestinal motility, nausea, vomiting, and alterations of the endocrine and autonomic nervous system.
Effects of the Central Nervous System (CNS)
The principal therapeutic action of oxycodone hydrochloride is analgesia. Although the precise mechanism of the analgesic action is unknown, specific CNS opioid receptors for endogenous compounds with oxycodone hydrochloride-like activity have been identified throughout the brain and spinal cord and are likely to play a role in the expression and perception of analgesic effects. In common with other opioids, oxycodone hydrochloride causes respiratory depression, in part by a direct effect on the brainstem respiratory centers. Oxycodone and related opioids depress the cough reflex by direct effect on the cough center in the medulla. Oxycodone causes miosis, even in total darkness.
Effects on the Gastrointestinal Tract And Other Smooth Muscle
Gastric, biliary and pancreatic secretions are decreased by oxycodone hydrochloride. Oxycodone hydrochloride, like other opioid analgesics, produces some degree of nausea and vomiting which is caused by direct stimulation of the chemoreceptor trigger zone (CTZ) located in the medulla. The frequency and severity of emesis gradually diminishes with time. Oxycodone hydrochloride may cause a decrease in the secretion of hydrochloric acid in the stomach, may reduce motility, while increasing the tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm. The end result may be constipation. Oxycodone hydrochloride may also cause spasm of the sphincter of Oddi and transient elevations in serum amylase.
Effects on the Cardiovascular System
In therapeutic doses, Oxycodone hydrochloride, produces peripheral vasodilatation (arteriolar and venous), decreased peripheral resistance, and inhibits baroreceptor reflexes. Manifestations of histamine release and/or peripheral vasodilatation may include pruritus, flushing, red eyes, sweating, and/or orthostatic hypotension. Caution should be used in hypovolemic patients, such as those suffering acute myocardial infarction, because oxycodone may cause or further aggravate their hypotension. Caution should also be used in patients with corpulmonale who have received therapeutic doses of opioids.
Endocrine System
Opioid agonists have been shown to have a variety of effects on the secretion of hormones. Opioids inhibit the secretion of ACTH, cortisol, and luteinizing hormone (LH) in humans. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon in humans and other species, rats and dogs. Thyroid stimulating hormone (TSH) has been shown to be both inhibited and stimulated by opioids.
12.3 Pharmacokinetics
The activity of oxycodone hydrochloride is primarily due to the parent drug oxycodone.
The oral bioavailability of oxycodone is 60% to 87%. Oxycodone is extensively metabolized and eliminated primarily in the urine as both conjugated and unconjugated parent and its metabolites. The apparent elimination half-life of oxycodone is approximately 4 hours.Absorption
About 60 to 87% of an oral dose reaches the systemic circulation in comparison to a parenteral dose. This high oral bioavailability (compared to other opioids) is due to lower pre-systemic and/or first-pass metabolism of oxycodone.
Food Effects
When oxycodone capsules are administered with a high-fat meal, mean AUC values are increased by 23% and peak concentrations are decreased by 14%. Food causes a delay in Tmax (1.00 to 3 hours). Similar effects of food are expected with the oral solution.
Distribution
Following intravenous administration, the volume of distribution (Vss) for oxycodone was 2.6 L/kg. Plasma protein binding of oxycodone at 37°C and a pH of 7.4 was about 45%. Oxycodone has been found in breast milk.
Metabolism
Oxycodone hydrochloride is extensively metabolized by multiple metabolic pathways to noroxycodone, oxymorphone, and noroxymorphone, which are subsequently glucuronidated. CYP3A4 mediated N-demethylation to noroxycodone is the primary metabolic pathway of oxycodone with a less contribution from CYP2D6 mediated O-demethylation to oxymorphone. Therefore, the formation of these and related metabolites can, in theory, be affected by other drugs. The major circulating metabolite is noroxycodone with an AUC ratio of 0.6 relative to that of oxycodone. Noroxycodone is reported to be a considerably weaker analgesic than oxycodone. Oxymorphone, although possessing analgesic activity, is present in the plasma only in low concentrations. The correlation between oxymorphone concentrations and opioid effects was much less than that seen with oxycodone plasma concentrations. The analgesic activity profile of other metabolites is not known.
Excretion
Oxycodone and its metabolites are excreted primarily via the kidney. The amounts measured in the urine have been reported as follows: free oxycodone up to 19%; conjugated oxycodone up to 50%; free oxymorphone 0%; conjugated oxymorphone < 14%; both free and conjugated noroxycodone have been found in the urine but not quantified. The total plasma clearance was 0.8 L/min for adults.
Apparent elimination half-life of oxycodone following the administration of oxycodone is approximately 4 hours.
Special Populations
Elderly: Information obtained from oxycodone tablets indicate that the plasma concentrations of oxycodone did not appear to be increased in patients over of the age of 65.
Gender: Information obtained from oxycodone tablets support the lack of gender effect on the pharmacokinetics of oxycodone.
Drug-Drug Interactions
CYP3A4 Inhibitors
CYP3A4 is the major enzyme involved in noroxycodone formation. A published study showed that the co-administration of voriconazole, a CYP3A4 inhibitor, increased oxycodone AUC and Cmax by 3.6 and 1.7 fold, respectively.
CYP3A4 Inducers
A published study showed that the co-administration of rifampin, a drug metabolizing enzyme inducer, decreased oxycodone AUC and Cmax values by 86% and 63%, respectively.
CYP2D6 Inhibitors
Oxycodone is metabolized in part to oxymorphone via the cytochrome p450 isoenzyme CYP2D6. While this pathway may be blocked by a variety of drugs (e.g., certain cardiovascular drugs and antidepressants), such blockade has not yet been shown to be of clinical significance with this agent.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Studies of oxycodone hydrochloride to evaluate its carcinogenic potential have not been conducted.
Mutagenesis
Oxycodone hydrochloride was genotoxic in an in vitro mouse lymphoma assay in the presence of metabolic activation. There was no evidence of genotoxic potential in an in vitro bacterial reverse mutation assay (Salmonella typhimurium and Escherichia coli) and in an assay for chromosomal aberrations (in vivo mouse bone marrow micronucleus assay).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Oxycodone Hydrochloride Oral Solution
Oxycodone Hydrochloride Oral Solution 20 mg/mL is a yellow solution available in one strength as follows:
Supplied as an individual overwrapped and flagged calibrated oral syringe:
Syringes of 0.5 mL containing 0.25 mL (5 mg) NDC: 48433-347-47
Syringes of 1 mL containing 0.5 mL (10 mg) NDC: 48433-347-55 -
17 PATIENT COUNSELING INFORMATION
See Medication Guide
Provide the following information to patients receiving oxycodone hydrochloride or their caregivers:
- Advise patients that oxycodone hydrochloride is a narcotic pain reliever, and should be taken only as directed.
- Advise patients how to measure and take the correct dose of Oxycodone Hydrochloride Oral Solution, and to always use the enclosed oral syringe when administering Oxycodone Hydrochloride Oral Solution 100 mg/5 mL (20 mg/mL) to ensure the dose is measured and administered accurately.
- Advise patients whenever the prescribed concentration is changed to avoid dosing errors which could result in accidental overdose and death.
- Advise patients that the 100 mg/5 mL (20 mg/mL) formulation is only for patients who are already receiving opioid-therapy and have demonstrated opioid-tolerance and that sharing this medication can result in fatal overdose and death, particularly in persons who have not had previous exposure to opioids.
- Advise patients not to adjust the dose of oxycodone hydrochloride without consulting with a physician or other healthcare professional.
- Advise patients that oxycodone hydrochloride may cause drowsiness, dizziness, or lightheadedness and may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery). Advise patients started on oxycodone hydrochloride or patients whose dose has been adjusted to refrain from any potentially dangerous activity until it is established that they are not adversely affected.
- Advise patients that oxycodone hydrochloride will add to the effect of alcohol and other CNS depressants (such as antihistamines, sedatives, hypnotics, tranquilizers, general anesthetics, phenothiazines, other opioids, and monoamine oxidase [MAO] inhibitors).
- Instruct patients not to combine oxycodone hydrochloride with central nervous system depressants (sleep aids, tranquilizers) except by the orders of the prescribing physician, and not to combine with alcohol because dangerous additive effects may occur, resulting in serious injury or death.
- Instruct women of childbearing potential who become or are planning to become pregnant to consult a physician prior to initiating or continuing therapy with oxycodone hydrochloride.
- Advise patients that safe use in pregnancy has not been established and that prolonged use of opioid analgesics during pregnancy may cause fetal-neonatal physical dependence, and neonatal withdrawal may occur.
- If patients have been receiving treatment with oxycodone hydrochloride for more than a few weeks and cessation of therapy is indicated, counsel them on the importance of safely tapering the dose and that abruptly discontinuing the medication could precipitate withdrawal symptoms. Provide a dose schedule to accomplish a gradual discontinuation of the medication.
- Advise patients that oxycodone hydrochloride is a potential drug of abuse. They must protect it from theft. It should never be given to anyone other than the individual for whom it was prescribed.
- Instruct patients to keep oxycodone hydrochloride in a secure place out of the reach of children. When oxycodone hydrochloride is no longer needed, the unused solution should be destroyed by flushing down the toilet.
- Advise patients taking oxycodone hydrochloride of the potential for severe constipation; appropriate laxatives and/or stool softeners as well as other appropriate treatments should be initiated from the onset of opioid therapy.
- Advise patients of the most common adverse events that may occur while taking oxycodone hydrochloride: constipation, nausea, somnolence, lightheadedness, dizziness, sedation, vomiting, and sweating.
- Advise patients to call 911 or the local Poison Control center, and get emergency help immediately if they take more oxycodone than prescribed, or overdose.
- Advise patients, that if they miss a dose, to take the missed dose as soon as possible. If it is almost time for the next dose, skip the missed dose and go back to their regular dosing schedule. Do not take two doses at once unless instructed by their doctor.
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
Medication Guide
Oxycodone Hydrochloride (ox-ee-CO-dohn) CII
Oral SolutionIMPORTANT: Keep Oxycodone Hydrochloride Oral Solution in a safe place away from children. Accidental use by a child is a medical emergency and can cause death. If a child accidentally takes Oxycodone Hydrochloride Oral Solution, get emergency help right away.
Read the Medication Guide that comes with Oxycodone Hydrochloride Oral Solution before you start taking it and each time you get a new prescription. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is the most important information I should know about Oxycodone Hydrochloride Oral Solution? Oxycodone Hydrochloride Oral solution can cause serious side effects, including, death.
- Take Oxycodone Hydrochloride Oral Solution exactly as prescribed by your healthcare provider. If you take the wrong dose or strength of Oxycodone Hydrochloride Oral Solution, you could overdose and die.
- It is especially important when you take Oxycodone Hydrochloride Oral Solution that you know exactly what dose and strength to take, and the right way to measure your medicine. Your healthcare provider or pharmacist should show you the right way to measure your medicine.
- Oral Syringe. Always use the oral syringe provided with Oxycodone Hydrochloride Oral Solution, 100 mg per 5 mL (20 mg per mL) to help make sure you measure the right amount.
- Do not drink alcohol. Using alcohol with Oxycodone Hydrochloride Oral Solution may increase your risk of dangerous side effects, including death.
- Opioid Tolerance. Do not take oxycodone Hydrochloride Oral Solution, 100 mg per 5 mL (20 mg per mL), unless you are "opioid tolerant." Opioid tolerant means that you take another opioid medicine for your constant (around the clock) pain and your body is used to it.
What is Oxycodone Hydrochloride Oral Solution?
Oxycodone Hydrochloride Oral Solution is in a group of drugs called narcotic pain relievers.
Oxycodone Hydrochloride Oral Solution is only for adults who have moderate to severe pain.
- A prescription medicine that is used to manage moderate to severe pain that is expected to last a short period of time (acute), and pain that continues around-the-clock and is expected to last for a long period of time (chronic).
- Oxycodone Hydrochloride Oral Solution is a federally controlled substance (CII) because it is a strong opioid pain medicine that can be abused by people who abuse prescription medicines or street drugs.
- Prevent theft, misuse or abuse. Keep Oxycodone Hydrochloride Oral Solution in a safe place to keep it from being stolen. Oxycodone Hydrochloride Oral Solution can be a target for people who misuse or abuse prescription medicines or street drugs.
- Never give Oxycodone Hydrochloride Oral Solution to anyone else, even if they have the same symptoms you have. It may harm them or even cause death.
- Selling or giving away this medicine is against the law.
- It is not known if Oxycodone Hydrochloride Oral Solution is safe and effective in children under age 18 years of age.
- Oxycodone Hydrochloride 100 mg per 5 mL (20 mg per mL) Oral Solution is only for adults with moderate to severe pain who are already using an opioid narcotic pain medicine and have been using this medicine continuously for several weeks or longer.
Who should not take Oxycodone Hydrochloride Oral Solution?
Do not take Oxycodone Hydrochloride Oral Solution if you:- are allergic to oxycodone, oxycodone salts, or any of the ingredients in Oxycodone Hydrochloride Oral Solution. See the end of this Medication Guide for a complete list of ingredients in Oxycodone Hydrochloride Oral Solution.
- are having breathing problems and there is no emergency medical equipment nearby
- are having an asthma attack or have severe asthma, trouble breathing, or lung problems
- have a bowel blockage called paralytic ileus
What should I tell my healthcare provider before taking Oxycodone Hydrochloride Oral Solution?
Before taking Oxycodone Hydrochloride Oral Solution, tell your healthcare provider if you:- have trouble breathing or lung problems
- have had a head injury
- have liver or kidney problems
- have adrenal gland problems, such as Addison's disease
- have severe scoliosis that affects your breathing
- have thyroid problems
- have problems urinating or enlargement of your prostate
- have or had convulsions or seizures
- have a past or present drinking problems or alcoholism
- have hallucinations (seeing or hearing things that are not really there) or other severe mental problems
- have constipation of other bowel problems
- have problems with your pancreas or gallbladder
- have past or present substance abuse or drug addiction
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if Oxycodone Hydrochloride Oral Solution will harm your unborn baby.
Talk to your healthcare provider if you are pregnant or plan to become pregnant.
If you take Oxycodone Hydrochloride Oral Solution regularly before your baby is born, your newborn baby may have signs of withdrawal because their body has become used to the medicine. Signs of withdrawal in a newborn baby can include:
- irritability
- being very active
- problems sleeping
- high pitched cry
- shaking (tremors)
- vomiting
- diarrhea or more stools than normal
- weight loss
If you are taking Oxycodone Hydrochloride Oral Solution right before your baby is born, your baby could have breathing problems.
- If you are breast-feeding or plan to breastfeed, some Oxycodone Hydrochloride Oral Solution passes into your breast milk. A nursing baby could become very sleepy or have difficulty breathing or feeding well. If you stop breastfeeding, your baby may have withdrawal symptoms. See the list of withdrawal symptoms above. You and your healthcare provider should decide if you will take Oxycodone Hydrochloride Oral Solution or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Sometimes the doses of medicines that you take with Oxycodone Hydrochloride Oral Solution may need to be changed if used together. Be especially careful about taking other medicines that make you sleepy such as:
- # sleeping pills
- # other pain medicines
- # anti-nausea medicines
- # tranquilizers
- # muscle relaxants
- # antihistamines
- # anti-anxiety medicines
- # anti-depressants
- # monoamine oxidase inhibitors (MAOIs)
- # anticholinergic medicines
Ask your healthcare provider if you are not sure if your medicine is one listed above.
- Do not take other medicines while using Oxycodone Hydrochloride Oral Solution until you have talked with your healthcare provider or pharmacist. They will tell you if it is safe to take other medicines with Oxycodone Hydrochloride Oral Solution.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take Oxycodone Hydrochloride Oral Solution?- See "What is the most important information I should know about Oxycodone Hydrochloride Oral Solution?"
- Take Oxycodone Hydrochloride Oral Solution exactly as prescribed. Do not change your dose unless your healthcare provider tells you to. Your healthcare provider may change your dose after seeing how the medicine affects you. Call your healthcare provider if your pain is not well controlled with your prescribed dose of Oxycodone Hydrochloride Oral Solution.
- Make sure you understand exactly how to measure your dose. Always use the oral syringe provided with your Oxycodone Hydrochloride Oral Solution, 100 mg per 5 mL (20 mg per mL), to help make sure you measure the right amount. See the Patient Instructions for Use at the end of this Medication Guide for information about how to measure your dose the right way. Ask your healthcare provider or pharmacist if you are not sure what dose of Oxycodone Hydrochloride Oral Solution you should take or if you are not sure how to use the oral syringe.
- Do not stop taking Oxycodone Hydrochloride Oral Solution suddenly. If you have been taking Oxycodone Hydrochloride Oral Solution for more than a few weeks, stopping it suddenly can make you sick with withdrawal symptoms (for example, nausea, vomiting, diarrhea, anxiety, and shivering). If your healthcare provider decides you no longer need Oxycodone Hydrochloride Oral Solution, ask how to slowly reduce this medicine. Do not stop taking Oxycodone Hydrochloride Oral Solution without talking to your healthcare provider.
- If you take more Oxycodone than prescribed, or overdose, call your local emergency number (such as 911) or your local Poison Control Center right away or get emergency help.
- Talk with your healthcare provider regularly about your pain to see if you still need to take Oxycodone.
What should I avoid while taking Oxycodone Hydrochloride Oral Solution?
- You should not drink alcohol while using Oxycodone Hydrochloride Oral Solution. Drinking alcohol with Oxycodone Hydrochloride Oral Solution may increase your risk of having dangerous side effects or death.
-
Do not drive, operate heavy machinery, or do other dangerous activities, especially when you start taking Oxycodone Hydrochloride Oral Solution and when your dose is changed, until you know how Oxycodone Hydrochloride Oral Solution affects you.
Oxycodone can make you sleepy. Ask your healthcare provider to tell you when it is okay to do these activites.
What are the possible side effects of Oxycodone Hydrochloride Oral Solution?
Oxycodone Hydrochloride Oral Solution can cause serious side effects, including:- See "What is the most important information I should know about Oxycodone Hydrochloride Oral Solution?"
-
Oxycodone can cause serious breathing problems that can become life-threatening, especially if Oxycodone Hydrochloride Oral Solution is used the wrong way. Call your healthcare provider or get help right away if:
- your breathing slows down
- you have shallow breathing (little chest movement with breathing)
- you feel faint, dizzy, confused, or
- you have any other unusual symptoms
These can be symptoms that you have taken too much Oxycodone Hydrochloride Oral Solution (overdose) or the dose is too high for you. These symptoms may lead to serious problems or death if not treated right away.
- Oxycodone Hydrochloride Oral Solution can cause your blood pressure to drop. This can make you feel dizzy if you get up too fast from sitting or lying down. Low blood pressure is also more likely to happen of you take other medicines that can also lower your blood pressure. Severe low blood pressure can happen if you lose blood or take certain other medicines.
-
Oxycodone can cause physical dependence. Do not stop taking Oxycodone or any other opioid without talking to your healthcare provider about how to slowly stop your medicine. You could become sick with uncomfortable withdrawal symptoms because your body has become used to these medicines. Physical dependence is not the same as drug addiction. Tell your healthcare provider if you have any of these symptoms of withdrawal while slowly stopping Oxycodone:
- # feel restless
- # tearing eyes
- # sweating
- # chills or hair on your arms "stand up"
- # muscle aches, backache
- # dilated pupils of your eyes
- # feel irritable or anxious
- # trouble sleeping
- # runny nose
- # yawning
- # nausea, loss of appetite, vomiting, diarrhea, stomach-area (abdominal) cramps
- # increase in your blood pressure, breathing faster, or your heart beats faster
- There is a chance of abuse or addiction with Oxycodone Hydrochloride Oral Solution. The chance is higher if you are or have been addicted to or abused other medicines, street drugs, or alcohol, or if you have a history of mental problems.
- Seizures. Oxycodone Hydrochloride Oral Solution may cause seizures or make seizures that you already have worse.
Call your healthcare provider if you have any of the symptoms listed above.
Common side effects of Oxycodone Hydrochloride Oral Solution include:- nausea
- constipation
- vomiting
- headache
- itchiness
- trouble sleeping
- dizziness
- weakness
- drowsiness
- sweating
- lightheadedness
Constipation (not often enough or hard bowel movements) is a very common side effect of pain medicines (opioids) including Oxycodone Hydrochloride Oral Solution. Talk to your healthcare provider about dietary changes, and the use of laxatives (medicines to treat constipation) and stool softeners to prevent or treat constipation while taking Oxycodone Hydrochloride Oral Solution.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Oxycodone Hydrochloride Oral Solution. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store Oxycodone Hydrochloride Oral Solution?
- Store Oxycodone Hydrochloride Oral Solution in a dry area at room temperature 59°F to 86°F (15°C to 30°C)
- Protect Oxycodone Hydrochlorde Oral Solution from moisture and light.
- After you stop taking Oxycodone, flush the unused oral solution down the toilet.
Keep Oxycodone Hydrochloride Oral Solution out of the reach of children. Accidental overdose by a child is a medical emergency and can lead to death.
General information about Oxycodone Hydrochloride Oral Solution
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Oxycodone Hydrochloride Oral Solution for a condition for which it was not prescribed.
Do not give your Oxycodone Hydrochloride Oral Solution to other people, even if they have the same symptoms you have. Selling or giving away Oxycodone Hydrochloride Oral Solution may harm others, may cause death, and is against the law.
This Medication Guide summarizes the most important information about Oxycodone Hydrochloride Oral Solution. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Oxycodone Hydrochloride Oral Solution that is written for healthcare professionals.
For more information about Oxycodone Hydrochloride Oral Solution, go to www.lvtechinc.com or call Pharm-Olam at 1-866-511-6754.
What are the ingredients in Oxycodone Hydrochloride Oral Solution?
Active ingredient: oxycodone hydrochloride, USP
Inactive ingredients: citric acid anhydrous, D&C Yellow #10, natural/artificial berry flavor, purified water, sodium citrate dihydrate, sodium benzoate, saccharin sodium, and sorbitol.Manufactured by:
Lehigh Valley Technologies, Inc.
Allentown, PA 18102
Distributed by:
Glenmark Generics Inc., USA
Mahwah, NJ 07430
Repackaged by:
Safecor Health, LLC
Columbus, OH 43204
Rev. 201-01 12/2013
This Medication Guide has been approved by the U.S. Food and Drug Administration. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
OXYCODONE HYDROCHLORIDE
oxycodone hydrochloride solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 48433-347(NDC:68462-347) Route of Administration ORAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYCODONE HYDROCHLORIDE (UNII: C1ENJ2TE6C) (OXYCODONE - UNII:CD35PMG570) OXYCODONE HYDROCHLORIDE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CITRATE (UNII: 1Q73Q2JULR) SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) Product Characteristics Color YELLOW Score Shape Size Flavor BERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 48433-347-47 .25 mL in 1 SYRINGE; Type 0: Not a Combination Product 09/23/2013 2 NDC: 48433-347-55 .5 mL in 1 SYRINGE; Type 0: Not a Combination Product 09/23/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA200535 09/23/2013 Labeler - Safecor Health, LLC (828269675) Establishment Name Address ID/FEI Business Operations Safecor Health, LLC 828269675 repack(48433-347)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
