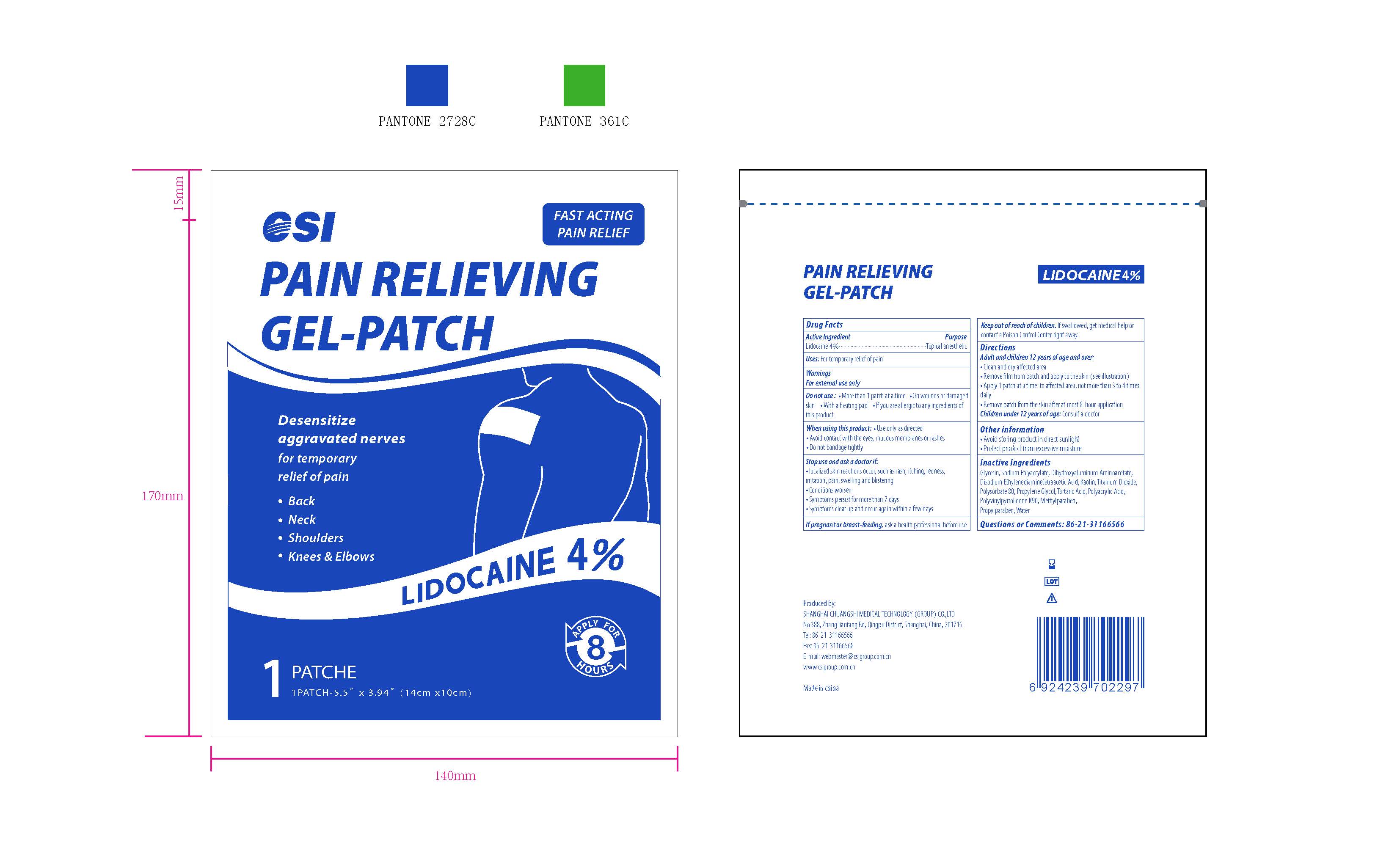
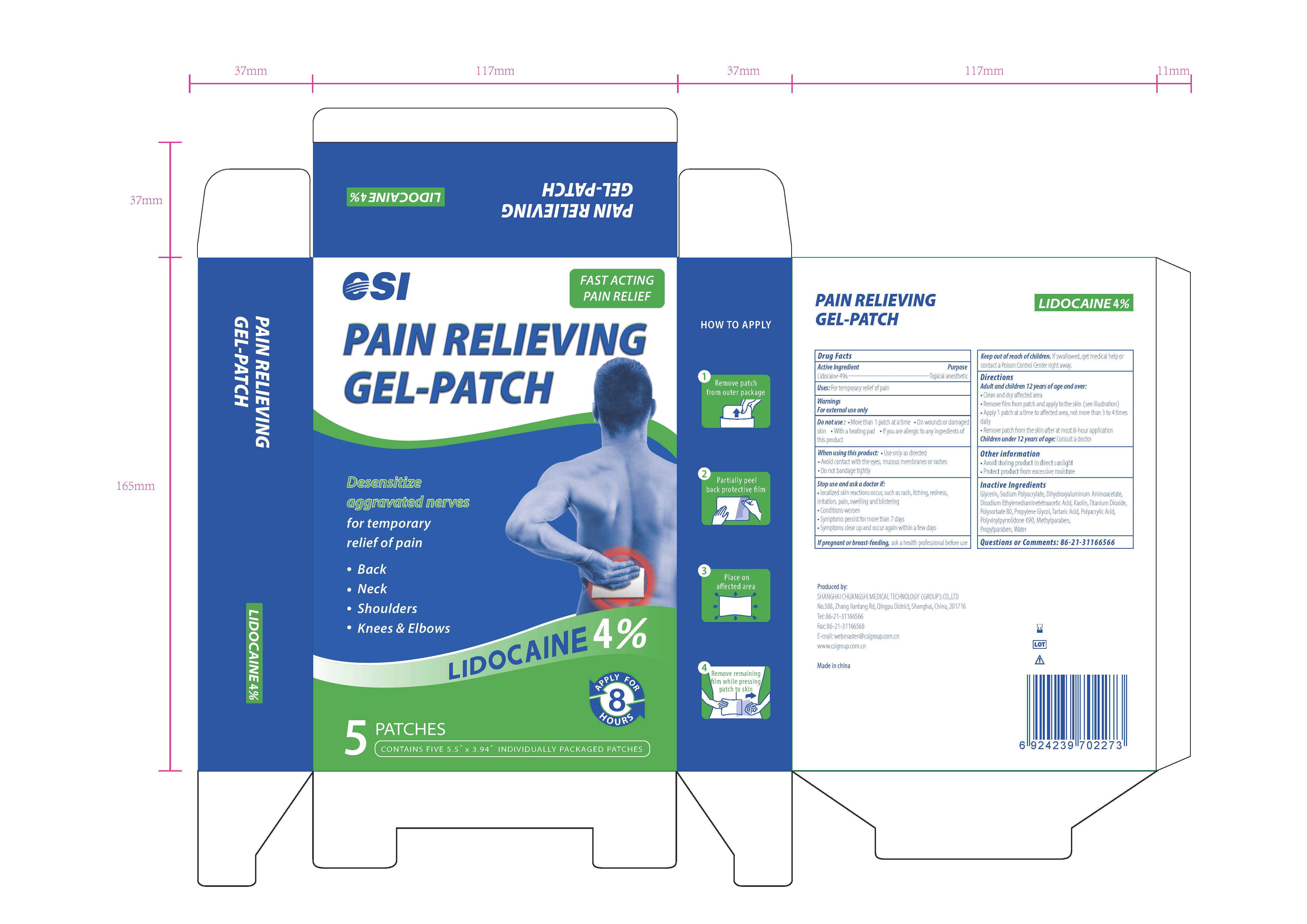
CSI, Pain Relieving Gel-Patch, Lidocaine 4%, 5 Patches
Pain Relieving Gel-Patch by
Drug Labeling and Warnings
Pain Relieving Gel-Patch by is a Otc medication manufactured, distributed, or labeled by Shanghai Chuangshi Medical Technology (Group) Co., Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
PAIN RELIEVING GEL-PATCH- lidocaine patch
Shanghai Chuangshi Medical Technology (Group) Co., Ltd.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
CSI, Pain Relieving Gel-Patch, Lidocaine 4%, 5 Patches
Ask a doctor if:
- localized skin reactions occur, such as rash, itching, redness, irritation, pain, swelling and blistering
- Conditions worsen
- Symptoms persists for more than 7 days
- Symptoms clear up and occur again within a few days
When using this product
- Use only as directed
- Avoid contact with the eyes, mucous membranes or rashed
- Do not bandage tightly
Do not use
- More than 1 patch at a time
- On wounds or damaged skin
- With a heating pad
- If you are allergic to any ingredients of this product
Stop use if:
- localized skin reactions occur, such as rash, itching, redness, irritation, pain, swelling and blistering
- Conditions worsen
- Symptoms persists for more than 7 days
- Symptoms clear up and occur again within a few days
Keep out of reach of children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adult and Children 12 years of age and over:
- Clean and dry affected area
- Remove film from patch and apply to the skin (see illustration)
- Apply 1 patch at a time to affected area, not more than 3 to 4 times daily
- Remove patch from the skin after at most 8-hour application
Children under 12 years of age: Consult a doctor
Dosage forms & strengths
This is a strip, patch dosage form.
The active ingredient strength is 4% w/w.
One patch one time, no more than 8 hours one time.
| PAIN RELIEVING GEL-PATCH
lidocaine patch |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) |
| Registrant - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Shanghai Chuangshi Medical Technology (Group) Co., Ltd. | 546872672 | manufacture(73557-156) , label(73557-156) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.