Hy-Vee, Inc. Milk Of Magnesia Drug Facts
milk of magnesia by
Drug Labeling and Warnings
milk of magnesia by is a Otc medication manufactured, distributed, or labeled by HyVee Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
MILK OF MAGNESIA- magnesium hydroxide suspension
HyVee Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Hy-Vee, Inc. Milk Of Magnesia Drug Facts
Uses
- relieves occasional constipation (irregularity)
- generally produces bowel movement in ½ to 6 hours
Warnings
Ask a doctor before use if you have
- kidney disease
- a magnesium-restricted diet
- stomach pain, nausea, or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
Ask a doctor or pharmacist before use if you are
taking any other drug. Take this product two or more hours before or after other drugs. Laxatives may affect how other drugs work.
Directions
- do not exceed the maximum recommended daily dose in a 24 hour period
- shake well before use
- dose may be taken once a day preferably at bedtime, in divided doses, or as directed by a doctor
- drink a full glass (8 oz) of liquid with each dose
- for accurate dosing, use dose cup provided
- mL = milliliter
|
adults and children 12 years and older |
30 mL to 60 mL |
|
children 6 to 11 years |
15 mL to 30 mL |
|
children under 6 years |
ask a doctor |
Other information
- each 15 mL contains: calcium 20 mg, magnesium 595 mg, and sodium 2 mg
- does not meet USP requirements for preservative effectiveness
- store at 20-25°C (68-77°F)
- close cap tightly after use
- avoid freezing
Principal Display Panel
COMPARE TO the Active Ingredient of Phillips’® Milk of Magnesia
Milk of Magnesia
Saline Laxative
Magnesium Hydroxide, 1200 mg per 15 mL
Fresh Mint Flavor
#1 Doctor Recommended Saline Laxative Active Ingredient
Comfortable, Cramp Free Relief
Sugar Free – Stimulant Free
12 FL OZ (355 mL)
| MILK OF MAGNESIA
magnesium hydroxide suspension |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - HyVee Inc (006925671) |
Trademark Results [milk of magnesia]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
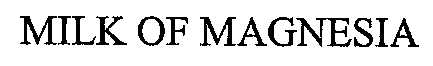 MILK OF MAGNESIA 70024049 0024049 Dead/Cancelled |
Charles H. Phillips Chemical Company ThE 1893-12-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

