ENTERIC COATED STIMULANT LAXATIVE by RUGBY LABORATORIES INC / TIME CAP LABS, INC / TIME CAP LABORATORIES, INC. RUGBY 14R
ENTERIC COATED STIMULANT LAXATIVE by
Drug Labeling and Warnings
ENTERIC COATED STIMULANT LAXATIVE by is a Otc medication manufactured, distributed, or labeled by RUGBY LABORATORIES INC, TIME CAP LABS, INC, TIME CAP LABORATORIES, INC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
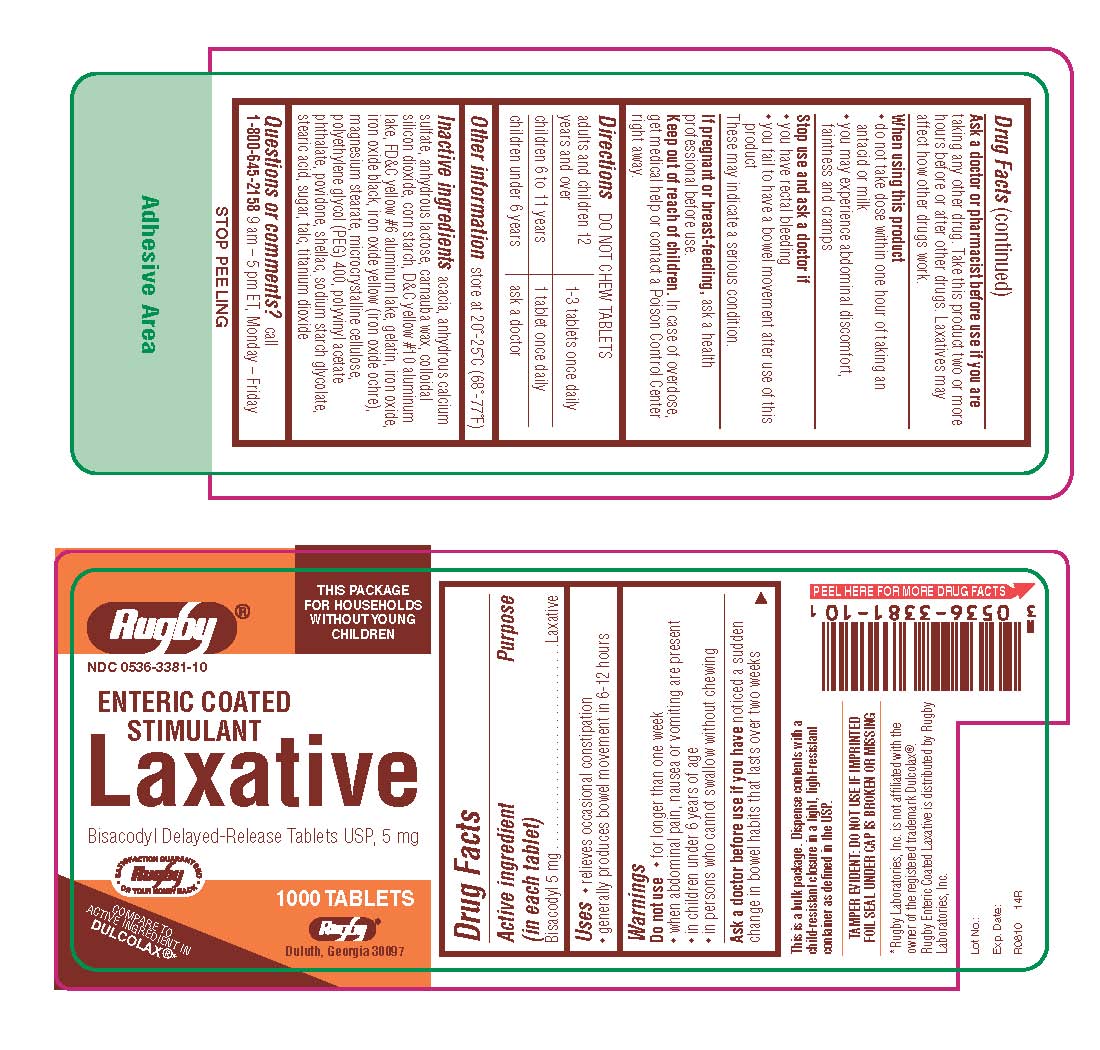
ENTERIC COATED STIMULANT LAXATIVE- bisacodyl tablet, delayed release
Rugby Laboratories, Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
RUGBY 14R
|
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. |
|
Directions
|
|
Inactive ingredients acacia, anhydrous calcium sulfate, anhydrous lactose, carnauba wax, colloidal silicon dioxide, corn starch, D-C yellow #10 aluminum lake, FD-C yellow #6 aluminum lake, gelatin, iron oxide, iron oxide black, iron oxide yellow (iron oxide ochre), magnesium stearate, microcrystalline cellulose, polyethylene glycol (PEG) 400, polyvinyl acetate phthalate, povidone, shellac, sodium starch glycolate, stearic acid, sugar, talc, titanium dioxide |
| ENTERIC COATED STIMULANT LAXATIVE
bisacodyl tablet, delayed release |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Rugby Laboratories, Inc (079246066) |
| Registrant - Time Cap Labs, Inc (037052099) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Time Cap Labs, Inc | 037052099 | manufacture(0536-3381) | |