KROGER- polyethylene glycol 3350 powder, for solution
Kroger by
Drug Labeling and Warnings
Kroger by is a Otc medication manufactured, distributed, or labeled by KROGER COMPANY. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
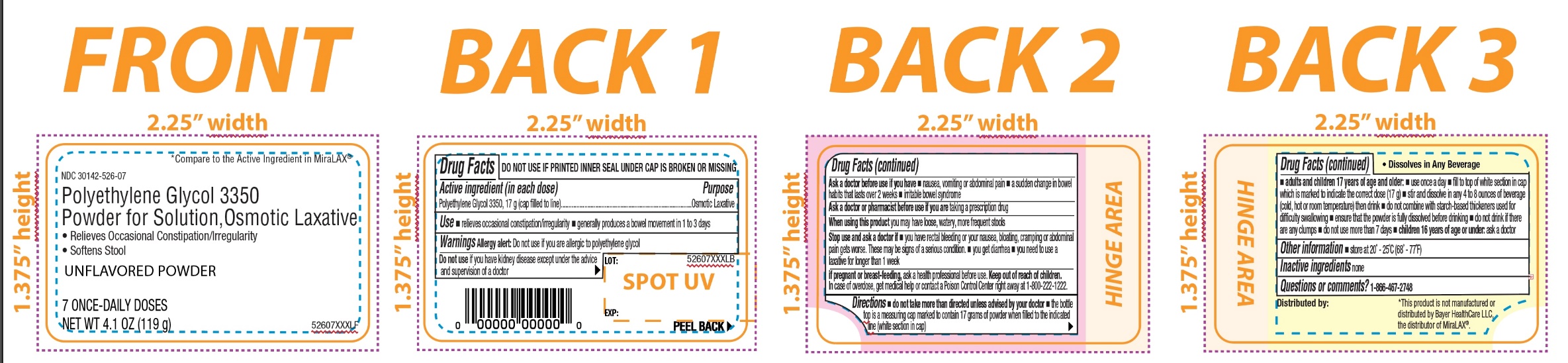
- Active ingredient (in each dose)
- Purpose
- Use
-
Warnings
Ask a doctor before use if you have
- nausea, vomiting or abdominal pain
- a sudden change in bowel habits that lasts over 2 weeks
- irritable bowel syndrome
-
Directions
- do not take more than directed unless advised by your doctor
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line (white section in cap)
-
adults and children 17 years of age and older:
- use once a day
- fill to top of white section in cap which is marked to indicate the correct dose (17 g)
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- do not combine with starch-based thickeners used for difficulty swallowing
- ensure that the powder is fully dissolved before drinking
- do not drink if there are any clumps
- do not use no more than 7 days
- children 16 years of age or under: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
*Compare to the Active Ingredient in MiraLAX®
NDC: 30142-526-30
Polyethylene Glycol 3350,
Powder for Solution,
Osmotic Laxative
- Relieves Occasional Constipation/Irregularity
- Softens Stool
UNFLAVORED POWDER
30 ONCE-DAILY DOSESNET WT 17.9 OZ (510g)
- Dissolves in ANY Beverage
*This product is not manufactured or distributed by Bayer HealthCare LLC, the distributer of MiraLAX®
-
Principal Display Panel
*Compare to the Active Ingredient in MiraLAX®
NDC: 30142-526-07
Polyethylene Glycol 3350,
Powder for Solution,
Osmotic Laxative
- Relieves Occasional Constipation/Irregularity
- Softens Stool
UNFLAVORED POWDER
7 ONCE-DAILY DOSESNET WT 4.1 OZ (119g)
-
INGREDIENTS AND APPEARANCE
KROGER
polyethylene glycol 3350 powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 30142-526 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 17 g in 17 g Product Characteristics Color white (Colorless upon dissolution) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 30142-526-30 510 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/03/2022 2 NDC: 30142-526-07 119 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 10/03/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202071 10/03/2022 Labeler - KROGER COMPANY (006999528)
Trademark Results [Kroger]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 KROGER 97704202 not registered Live/Pending |
The Kroger Co. of Michigan 2022-12-05 |
 KROGER 85489540 4164728 Live/Registered |
The Kroger Co. of Michigan 2011-12-07 |
 KROGER 78777677 3250834 Live/Registered |
The Kroger Co. of Michigan 2005-12-20 |
 KROGER 78483412 3027881 Live/Registered |
The Kroger Co. of Michigan 2004-09-14 |
 KROGER 76190440 2665432 Live/Registered |
Kroger Co. of Michigan, The 2001-01-08 |
 KROGER 76189921 2623620 Live/Registered |
Kroger Co. of Michigan, The 2001-01-08 |
 KROGER 76189920 2673940 Live/Registered |
Kroger Co. of Michigan, The 2001-01-08 |
 KROGER 76189917 2673939 Live/Registered |
Kroger Co. of Michigan, The 2001-01-08 |
 KROGER 76182163 2634257 Live/Registered |
Kroger Co. of Michigan, The 2000-12-18 |
 KROGER 76182125 2645252 Live/Registered |
Kroger Co. of Michigan, The 2000-12-18 |
 KROGER 75332788 2205297 Dead/Cancelled |
Kroger Co. of Michigan, The 1997-07-30 |
 KROGER 72218878 0813182 Live/Registered |
KROGER CO., THE 1965-05-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.