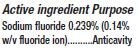dentalux charcoal by Lidl US LLC
dentalux charcoal by
Drug Labeling and Warnings
dentalux charcoal by is a Otc medication manufactured, distributed, or labeled by Lidl US LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DENTALUX CHARCOAL- sodium fluoride paste, dentifrice
Lidl US LLC
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Warnings
Keep out of reach of children under 6 years of age. If more than used for brushing is accidentally swallowed, get medical help or contact Poison Control Center right away.
tube:

box:

Directions
- adults and children 2 years of age and older: brush teeth thoroughly, preferably after each meal or at least twice a day, or as directed by a dentist or doctor.
- do not swallow
- to minimize swallowing use a pea-sized amount in children und 6
- teach children under 6 years of age good brushing and rinsing habits until capable of using without supervision
- children under 2 years of age: consult a dentist or doctor
tube:

box:

| DENTALUX CHARCOAL
sodium fluoride paste, dentifrice |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Lidl US LLC (079389709) |
Revised: 3/2023
Document Id: f81f5efd-c318-e0bf-e053-6394a90aa029
Set id: c5e7fdeb-4b83-b14a-e053-2a95a90ad333
Version: 4
Effective Time: 20230330
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.