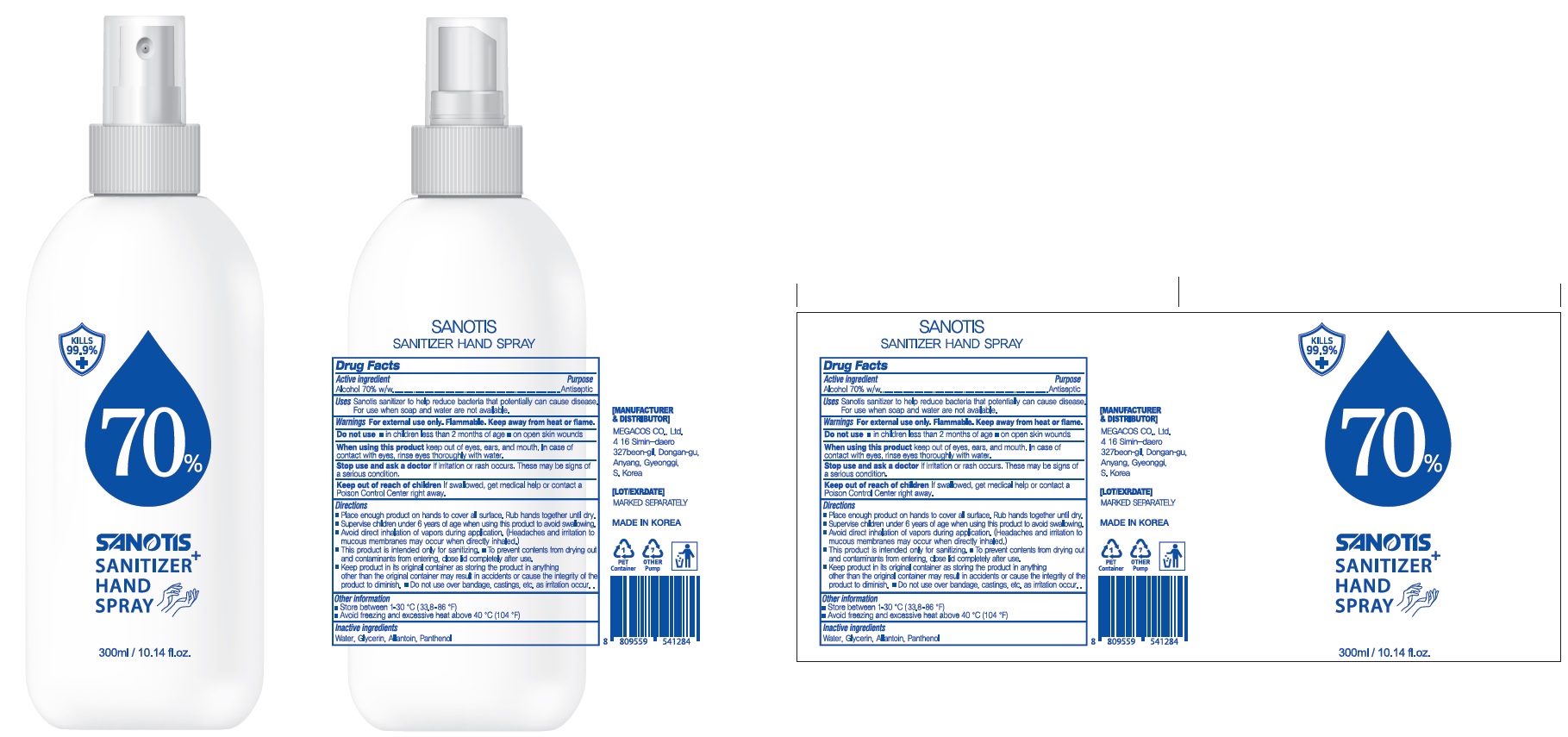
SANOTIS SANITIZER HAND by MEGACOS MANUFACTURING Co., Ltd.
SANOTIS SANITIZER HAND by
Drug Labeling and Warnings
SANOTIS SANITIZER HAND by is a Otc medication manufactured, distributed, or labeled by MEGACOS MANUFACTURING Co., Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SANOTIS SANITIZER HAND- alcohol spray
MEGACOS MANUFACTURING Co., Ltd.
----------
WARNINGS
For external use only. Flammable. Keep away from heat or flame
--------------------------------------------------------------------------------------------------------
Do not use
in children less than 2 months of age
on open skin wounds
--------------------------------------------------------------------------------------------------------
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
----------------------------------------------------------------------- ---------------------------------
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
KEEP OUT OF REACH OF CHILDREN
If swallowed, get medical help or contact a Poison Control Center right away.
Uses
Sanotis sanitizer to help reduce bacteria that potentially can cause disease.
For use when soap and water are not available.
Directions
Place enough product on hands to cover all surface. Rub hands together until dry.
Supervise children under 6 years of age when using this product to avoid swallowing.
Avoid direct inhalation of vapors during application. (Headaches and irritation to mucous membranes may occur when directly inhaled.)
This product is intended only for sanitizing.
To prevent contents from drying out and contaminants from entering, close lid completely after use.
Keep product in its original container as storing the product in anything other than the original container may result in accidents or cause the integrity of the product to diminish.
Do not use over bandage, castings, etc. as irritation occur.
| SANOTIS SANITIZER HAND
alcohol spray |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - MEGACOS MANUFACTURING Co., Ltd. (694745986) |
| Registrant - MEGACOS MANUFACTURING Co., Ltd. (694745986) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| MEGACOS MANUFACTURING Co., Ltd. | 694745986 | manufacture(78063-040) | |