VAZALORE- aspirin capsule
VAZALORE by
Drug Labeling and Warnings
VAZALORE by is a Otc medication manufactured, distributed, or labeled by PLx Pharma Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Uses
-
Warnings
Warnings
Reye’s Syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy Alert: Aspirin may cause a severe allergic reaction which may include
n hives n facial wseelling n shock n astham (wheezing)
This product contains soy
Stomach bleeding warning: This product contains an NSAID, which may cause bleeding. The chance is higher if you:
- Are age 60 or older
- Have had stomach ulcers or bleeding problems
- Take a blood thinning (anticoagulant) or steroid drug
- Take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- Have 3 or more alcoholic drinks every day while using this product
- Take more or for a longer time than directed
- ACTIVE INGREDIENT
- ASK DOCTOR
- DO NOT USE
- ASK DOCTOR/PHARMACIST
-
Stop use and ask a doctor
Stop use and ask a doctor if
- An allergic reaction occurs. Seek medical help right away
- You experience amu of the following signs of stomach bleeding
- Feel faint - vomit blood - have bloody or black stool - have stomach pain that does not get better
- Pain gets worse or lasts more than 10 days
- Fever gets worse or lasts more than 3 days
- Ringing in the ears or loss of hearing occurs
- Redness and swelling is present in the painful area
- Any new symptoms occur. These could be signs of serious conditions
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- PURPOSE
- Questions or Comments
- Other Information
- INACTIVE INGREDIENT
- Directions
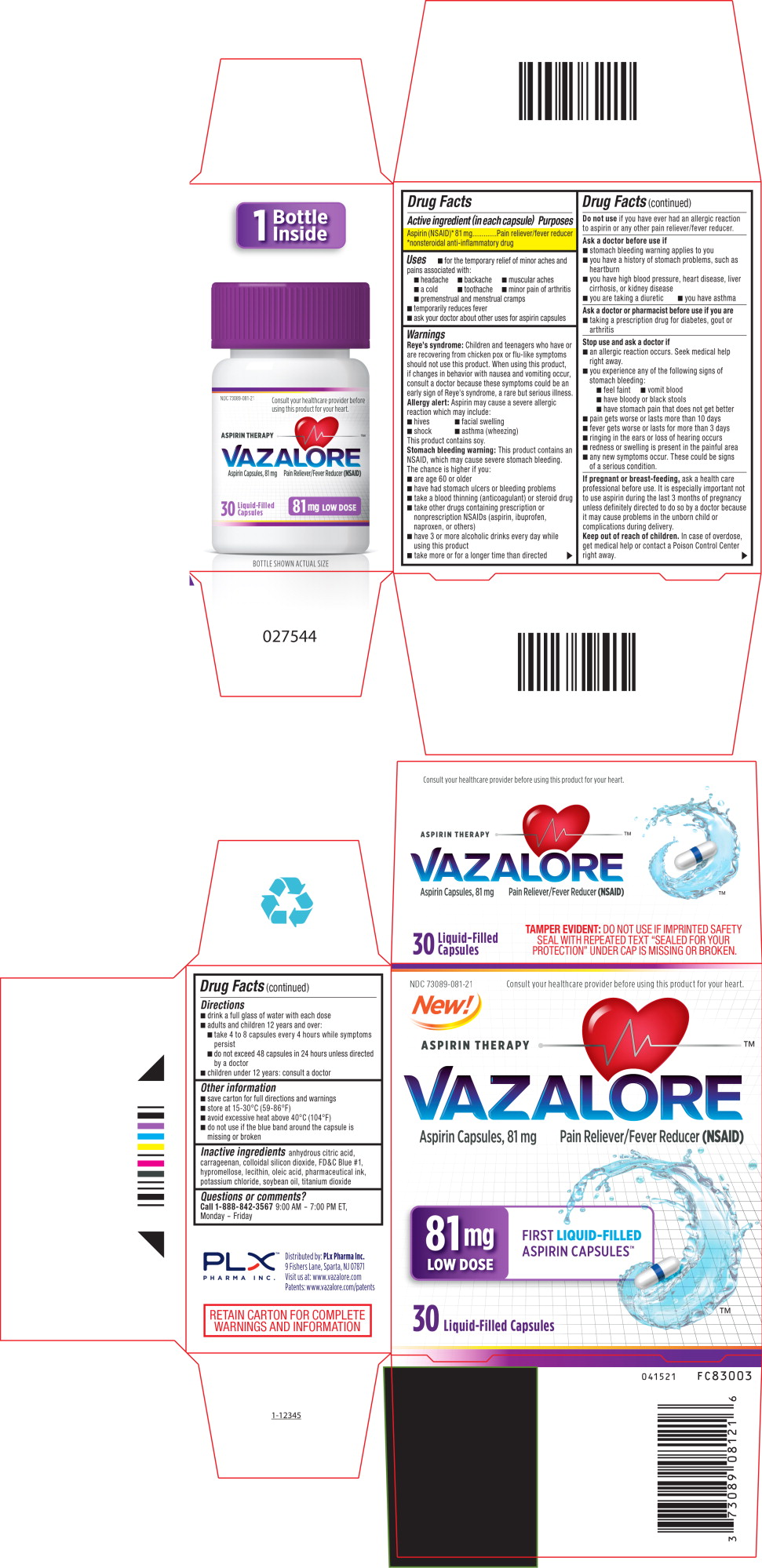
- 30 Count Blister Carton
- 30 Count Bottle Label
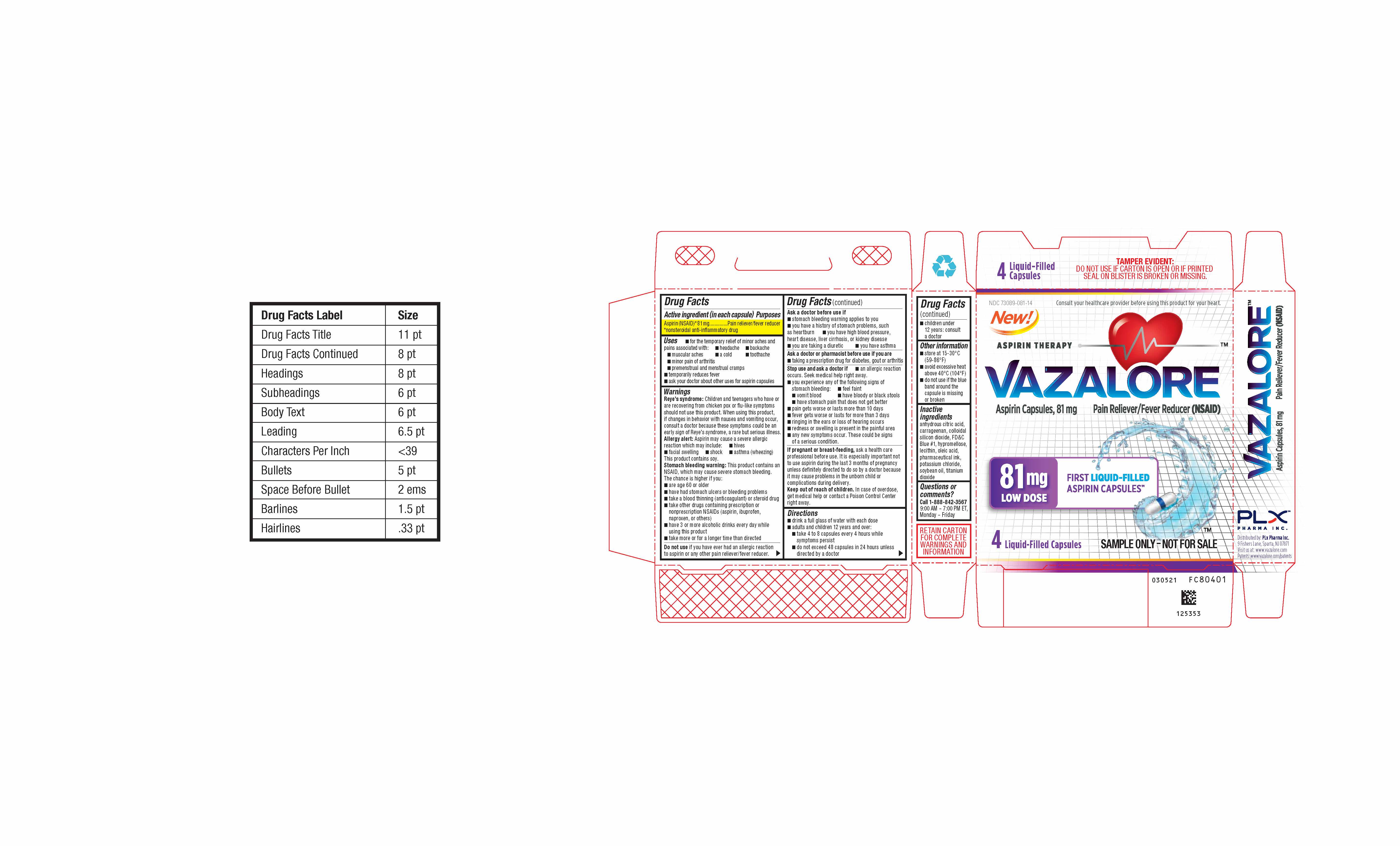
- 4 Count Blister Card
- 4 Count Blister Carton
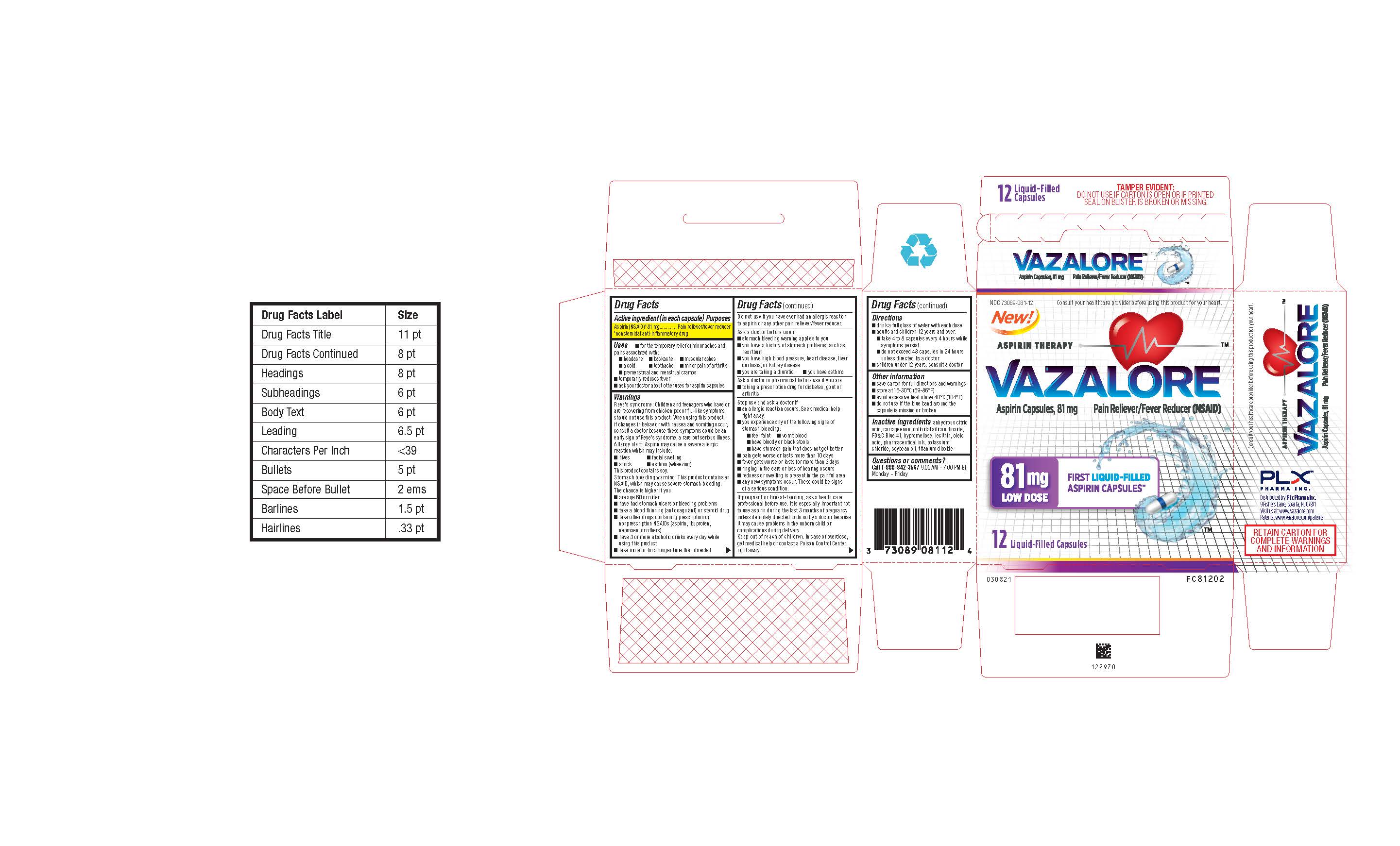
- 12 Count Blister Carton
-
INGREDIENTS AND APPEARANCE
VAZALORE
aspirin capsuleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 73089-081 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 81 mg in 81 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) CARRAGEENAN (UNII: 5C69YCD2YJ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) OLEIC ACID (UNII: 2UMI9U37CP) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SOYBEAN OIL (UNII: 241ATL177A) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (White with Blue Band and A81 Imprint) Score no score Shape CAPSULE (White Capsule with Blue Band and A81 Imprint) Size 14mm Flavor Imprint Code A81 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 73089-081-21 1 in 1 CARTON 08/05/2021 1 81 mg in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC: 73089-081-12 1 in 1 CARTON 08/05/2021 2 81 mg in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 73089-081-14 81 mg in 1 CARTON; Type 0: Not a Combination Product 08/05/2021 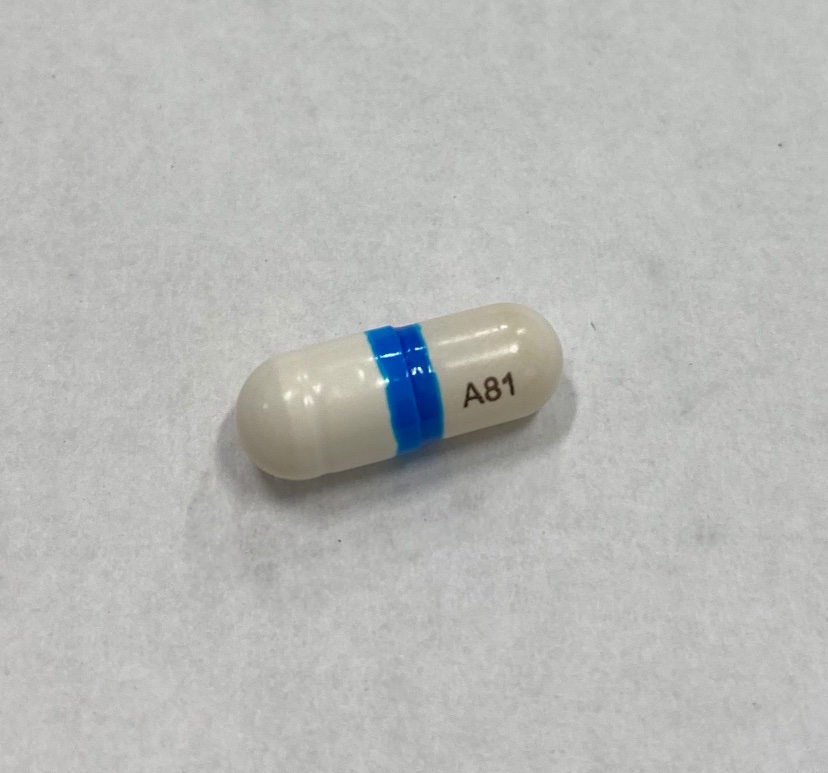
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA203697 08/05/2021 Labeler - PLx Pharma Inc (079325568) Registrant - PLx Pharma Inc (079325568)
Trademark Results [VAZALORE]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 VAZALORE 90157665 not registered Live/Pending |
PLX PHARMA INC. 2020-09-03 |
 VAZALORE 88756309 not registered Live/Pending |
PLx Pharma Inc. 2020-01-13 |
 VAZALORE 88677340 not registered Live/Pending |
PLx Pharma, Inc. 2019-11-01 |
 VAZALORE 87679090 not registered Live/Pending |
PLX PHARMA INC. 2017-11-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.