DECITABINE injection, powder, lyophilized, for solution
Decitabine by
Drug Labeling and Warnings
Decitabine by is a Prescription medication manufactured, distributed, or labeled by Sandoz Inc, MSN LABORATORIES PRIVATE LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DECITABINE FOR INJECTION safely and effectively. See full prescribing information for DECITABINE FOR INJECTION.
DECITABINE for injection, for intravenous use
Initial U.S. Approval: 2006INDICATIONS AND USAGE
Decitabine for injection is a nucleoside metabolic inhibitor indicated for treatment of patients with myelodysplastic syndromes (MDS) including previously treated and untreated, de novo and secondary MDS of all French-American-British subtypes (refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia) and intermediate-1, intermediate-2, and high-risk International Prognostic Scoring System groups.(1)
DOSAGE AND ADMINISTRATION
- Three Day Regimen
Administer decitabine for injection at a dose of 15 mg/m2 by continuous intravenous infusion over 3 hours repeated every 8 hours for 3 days. Repeat cycle every 6 weeks. (2.1)
- Five Day Regimen
Administer decitabine for injection at a dose of 20 mg/m2 by continuous intravenous infusion over 1 hour repeated daily for 5 days. Repeat cycle every 4 weeks. (2.2)
DOSAGE FORMS AND STRENGTHS
For Injection: 50 mg of decitabine as a lyophilized powder in a single-dose vial for reconstitution. (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Neutropenia and thrombocytopenia: Perform complete blood counts and platelet counts. (5.1)
- Embryo-fetal Toxicity: Can cause fetal harm. Advise patients of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.2, 8.1, 8.3)
ADVERSE REACTIONS
Most common adverse reactions (>50%) are neutropenia, thrombocytopenia, anemia, and pyrexia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
Lactation: Advise not to breastfeed. (8.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Starting Dosage
2.2 Dose Modifications for Toxicity
2.3 Instructions for Intravenous Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
5.2 Embryo-fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis and Impairment of Fertility
14 CLINICAL STUDIES
14.1 Controlled Trial in Myelodysplastic Syndrome
14.2 Single-arm Studies in Myelodysplastic Syndrome
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
Decitabine for injection is indicated for treatment of adult patients with myelodysplastic syndromes (MDS) including previously treated and untreated, de novo and secondary MDS of all French-American-British subtypes (refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia) and intermediate-1, intermediate-2, and high- risk International Prognostic Scoring System groups.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Starting Dosage
Pre-Medications and Baseline Testing
Consider pre-medicating for nausea with antiemetics.
Conduct baseline laboratory testing: CBC with platelets, serum hepatic panel, and serum creatinine.
Decitabine for Injection Regimen Options
Three Day Regimen
Administer decitabine for injection at a dose of 15 mg/m2 by continuous intravenous infusion over 3 hours repeated every 8 hours for 3 days. Repeat cycles every 6 weeks upon hematologic recovery (ANC at least 1,000/μL and platelets at least 50,000/μL) for a minimum of 4 cycles. A complete or partial response may take longer than 4 cycles. Delay and reduce dose for hematologic toxicity (see Dose Modifications for Toxicity).
Five Day Regimen
Administer decitabine for injection at a dose of 20 mg/m2 by continuous intravenous infusion over 1 hour daily for 5 days. Delay and reduce dose for hematologic toxicity (see Dose Modifications for Toxicity). Repeat cycles every 4 weeks upon hematologic recovery (ANC at least 1,000/μL and platelets at least 50,000/μL) for a minimum of 4 cycles. A complete or partial response may take longer than 4 cycles.2.2 Dose Modifications for Toxicity
Hematologic Toxicity
If hematologic recovery from a previous decitabine for injection treatment cycle requires more than 6 weeks, delay the next cycle of decitabine for injection therapy and reduce decitabine for injection dose temporarily by following this algorithm:
Recovery requiring more than 6, but less than 8 weeks – delay decitabine for injection dosing for up to 2 weeks and reduce the dose temporarily to 11 mg/m2 every 8 hours (33 mg/m2/day, 99 mg/m2/cycle) upon restarting therapy.
Recovery requiring more than 8, but less than 10 weeks - Perform bone marrow aspirate to assess for disease progression. In the absence of progression, delay the decitabine for injection dose up to 2 more weeks and reduce the dose to 11 mg/m2 every 8 hours (33 mg/m2/day, 99 mg/m2/cycle) upon restarting therapy, then maintain or increase dose in subsequent cycles as clinically indicated.
Non-Hematologic Toxicity
Delay subsequent decitabine for injection treatment for any the following non-hematologic toxicities and do not restart until toxicities resolve:
Serum creatinine greater than or equal to 2 mg/dL
SGPT, total bilirubin greater than or equal to 2 times ULN
Active or uncontrolled infection2.3 Instructions for Intravenous Administration
Decitabine for injection is a cytotoxic drug and caution should be exercised when handling and preparing decitabine for injection1.
Aseptically reconstitute decitabine for injection with room temperature (20°C to 25°C) 10 mL of Sterile Water for Injection, USP. Upon reconstitution, the final concentration of the reconstituted decitabine for injection solution is 5 mg/mL. You must dilute the reconstituted solution with 0.9% Sodium Chloride Injection or 5% Dextrose Injection prior to administration. Temperature of the diluent (0.9% Sodium Chloride Injection or 5% Dextrose Injection) depends on time of administration after preparation.
For Administration within 15 Minutes of Preparation
If decitabine for injection is intended to be administered within 15 minutes from the time of preparation, dilute the reconstituted solution with room temperature (20°C to 25°C) 0.9% Sodium Chloride Injection or 5% Dextrose Injection to a final concentration of 0.1 mg/mL to 1 mg/mL.
For Delayed Administration
If decitabine for injection is intended to be administered after 15 minutes of preparation, dilute the reconstituted solution with cold (2°C to 8°C) 0.9% Sodium Chloride Injection or 5% Dextrose Injection to a final concentration of 0.1 mg/mL to 1 mg/mL. Store at 2°C to 8°C for up to 4 hours. Diluted stored solution must be used within 4 hours from the time of preparation.
Use the diluted, refrigerated solution within 4 hours from the time of preparation or discard.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if there is evidence of particulate matter or discoloration.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Myelosuppression
Fatal and serious myelosuppression occurs in decitabine-treated patients. Myelosuppression (anemia, neutropenia, and thrombocytopenia) is the most frequent cause of decitabine dose reduction, delay, and discontinuation. Neutropenia of any grade occurred in 90% of decitabine-treated patients with grade 3 or 4 occurring in 87% of patients. Thrombocytopenia of any grade occurred in 89% of patients with grade 3 or 4 occurring in 85% of patients. Grade 3 or 4 febrile neutropenia occurred in 23% of patients. Anemia of any grade occurred in 82% of patients. Perform complete blood count with platelets at baseline, prior to each cycle, and as needed to monitor response and toxicity. Manage toxicity using dose-delay, dose-reduction, growth factors, and anti-infective therapies as needed [see Dosage and Administration (2.2)]. Myelosuppression and worsening neutropenia may occur more frequently in the first or second treatment cycles, and may not necessarily indicate progression of underlying MDS.
5.2 Embryo-fetal Toxicity
Decitabine can cause fetal harm when administered to a pregnant woman. Based on its mechanism of action, decitabine alters DNA synthesis and is expected to result in adverse reproductive effects [see Clinical Pharmacology (12.1)]. In preclinical studies in mice and rats, decitabine was teratogenic, fetotoxic, and embryotoxic. If this drug is used during pregnancy, or if a patient becomes pregnant while receiving this drug, the patient should be apprised of the potential hazard to the fetus. Advise women of childbearing potential to avoid becoming pregnant while taking decitabine and for 6 months following the last dose. Advise men with female partners of childbearing potential to avoid fathering a child while receiving treatment with decitabine, and for 3 months following the last dose, Counsel patients of childbearing potential to use effective contraception during this time [see Use in Specific Populations (8.1, 8.3)].
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of decitabine was studied in 3 single-arm studies (N = 66, N = 98, N = 99) and 1 controlled supportive care study (N = 83 decitabine, N = 81 supportive care). The data described below reflect exposure to decitabine in 83 patients in the MDS trial. In the trial, patients received 15 mg/m2 intravenously every 8 hours for 3 days every 6 weeks. The median number of decitabine cycles was 3 (range 0 to 9).
Most Commonly Occurring Adverse Reactions: neutropenia, thrombocytopenia, anemia, fatigue, pyrexia, nausea, cough, petechiae, constipation, diarrhea, and hyperglycemia.
Adverse Reactions Most Frequently (≥1%) Resulting in Clinical Intervention and or Dose Modification in the Phase 3 Trials in the decitabine Arm:
- Discontinuation: thrombocytopenia, neutropenia, pneumonia, Mycobacterium avium complex infection, cardio-respiratory arrest, increased blood bilirubin, intracranial hemorrhage, abnormal liver function tests.
- Dose Delayed: neutropenia, pulmonary edema, atrial fibrillation, central line infection, febrile neutropenia.
- Dose Reduced: neutropenia, thrombocytopenia, anemia, lethargy, edema, tachycardia, depression, pharyngitis.
Table 1 presents all adverse reactions occurring in at least 5% of patients in the decitabine group and at a rate greater than supportive care.
Table 1 : Adverse Reactions Reported in ≥ 5% of Patients in the Decitabine Group and at a Rate Greater than Supportive Care in Phase 3 MDS Trial
Decitabine
N = 83 (%)
Supportive Care
N = 81 (%)
Blood and lymphatic system disorders
Neutropenia
75 (90)
58 (72)
Thrombocytopenia
74 (89)
64 (79)
Anemia NOS
68 (82)
60 (74)
Febrile neutropenia
24 (29)
5 (6)
Leukopenia NOS
23 (28)
11 (14)
Lymphadenopathy
10 (12)
6 (7)
Thrombocythemia
4 (5)
1 (1)
Cardiac disorders
Pulmonary edema NOS
5 (6)
0 (0)
Eye disorders
Vision blurred
5 (6)
0 (0)
Gastrointestinal disorders
Nausea
35 (42)
13 (16)
Constipation
29 (35)
11 (14)
Diarrhea NOS
28 (34)
13 (16)
Vomiting NOS
21 (25)
7 (9)
Abdominal pain NOS
12 (14)
5 (6)
Oral mucosal petechiae
11 (13)
4 (5)
Stomatitis
10 (12)
5 (6)
Dyspepsia
10 (12)
1 (1)
Ascites
8 (10)
2 (2)
Gingival bleeding
7 (8)
5 (6)
Hemorrhoids
7 (8)
3 (4)
Loose stools
6 (7)
3 (4)
Tongue ulceration
6 (7)
2 (2)
Dysphagia
5 (6)
2 (2)
Oral soft tissue disorder NOS
5 (6)
1 (1)
Lip ulceration
4 (5)
3 (4)
Abdominal distension
4 (5)
1 (1)
Abdominal pain upper
4 (5)
1 (1)
Gastro-esophageal reflux disease
4 (5)
0 (0)
Glossodynia
4 (5)
0 (0)
General disorders and administrative site disorders
Pyrexia
44 (53)
23 (28)
Edema peripheral
21 (25)
13 (16)
Rigors
18 (22)
14 (17)
Edema NOS
15 (18)
5 (6)
Pain NOS
11 (13)
5 (6)
Lethargy
10 (12)
3 (4)
Tenderness NOS
9 (11)
0 (0)
Fall
7 (8)
3 (4)
Chest discomfort
6 (7)
3 (4)
Intermittent pyrexia
5 (6)
3 (4)
Malaise
4 (5)
1 (1)
Crepitations NOS
4 (5)
1 (1)
Catheter site erythema
4 (5)
1 (1)
Catheter site pain
4 (5)
0 (0)
Injection site swelling
4 (5)
0 (0)
Hepatobiliary disorders
Hyperbilirubinemia
12 (14)
4 (5)
Infections and infestations
Pneumonia NOS
18 (22)
11 (14)
Cellulitis
10 (12)
6 (7)
Candidal infection NOS
8 (10)
1 (1)
Catheter related infection
7 (8)
0 (0)
Urinary tract infection NOS
6 (7)
1 (1)
Staphylococcal infection
6 (7)
0 (0)
Oral candidiasis
5 (6)
2 (2)
Sinusitis NOS
4 (5)
2 (2)
Bacteremia
4 (5)
0 (0)
Injury, poisoning and procedural complications
Transfusion reaction
6 (7)
3 (4)
Abrasion NOS
4 (5)
1 (1)
Investigations
Cardiac murmur NOS
13 (16)
9 (11)
Blood alkaline phosphatase NOS increased
9 (11)
7 (9)
Aspartate aminotransferase increased
8 (10)
7 (9)
Blood urea increased
8 (10)
1 (1)
Blood lactate dehydrogenase increased
7 (8)
5 (6)
Blood albumin decreased
6 (7)
0 (0)
Blood bicarbonate increased
5 (6)
1 (1)
Blood chloride decreased
5 (6)
1 (1)
Protein total decreased
4 (5)
3 (4)
Blood bicarbonate decreased
4 (5)
1 (1)
Blood bilirubin decreased
4 (5)
1 (1)
Metabolism and nutrition disorders
Hyperglycemia NOS
27 (33)
16 (20)
Hypoalbuminemia
20 (24)
14 (17)
Hypomagnesemia
20 (24)
6 (7)
Hypokalemia
18 (22)
10 (12)
Hyponatremia
16 (19)
13 (16)
Appetite decreased NOS
13 (16)
12 (15)
Anorexia
13 (16)
8 (10)
Hyperkalemia
11 (13)
3 (4)
Dehydration
5 (6)
4 (5)
Musculoskeletal and connective tissue disorders
Arthralgia
17 (20)
8 (10)
Pain in limb
16 (19)
8 (10)
Back pain
14 (17)
5 (6)
Chest wall pain
6 (7)
1 (1)
Musculoskeletal discomfort
5 (6)
0 (0)
Myalgia
4 (5)
1 (1)
Nervous system disorders
Headache
23 (28)
11 (14)
Dizziness
15 (18)
10 (12)
Hypoesthesia
9 (11)
1 (1)
Psychiatric disorders
Insomnia
23 (28)
11 (14)
Confusional state
10 (12)
3 (4)
Anxiety
9 (11)
8 (10)
Renal and urinary disorders
Dysuria
5 (6)
3 (4)
Urinary frequency
4 (5)
1 (1)
Respiratory, thoracic and Mediastinal disorders
Cough
33 (40)
25 (31)
Pharyngitis
13 (16)
6 (7)
Crackles lung
12 (14)
1 (1)
Breath sounds decreased
8 (10)
7 (9)
Hypoxia
8 (10)
4 (5)
Rales
7 (8)
2 (2)
Postnasal drip
4 (5)
2 (2)
Skin and subcutaneous tissue disorders
Ecchymosis
18 (22)
12 (15)
Rash NOS
16 (19)
7 (9)
Erythema
12 (14)
5 (6)
Skin lesion NOS
9 (11)
3 (4)
Pruritis
9 (11)
2 (2)
Alopecia
7 (8)
1 (1)
Urticaria NOS
5 (6)
1 (1)
Swelling face
5 (6)
0 (0)
Vascular disorders
Petechiae
32 (39)
13 (16)
Pallor
19 (23)
10 (12)
Hypotension NOS
5 (6)
4 (5)
Hematoma NOS
4 (5)
3 (4)
In a single-arm MDS study (N=99) decitabine was dosed at 20 mg/m2 intravenous, infused over one hour daily for 5 consecutive days of a 4-week cycle. Table 2 presents all adverse reactions occurring in at least 5% of patients.
Table 2 : Adverse Reactions Reported in ≥ 5% of Patients in a Single-arm Study*
Decitabine
N = 99 (%)
Blood and lymphatic system disorders
Anemia
31 (31%)
Febrile neutropenia
20 (20%)
Leukopenia
6 (6% )
Neutropenia
38 (38% )
Pancytopenia
5 (5% )
Thrombocythemia
5 (5% )
Thrombocytopenia
27 (27%)
Cardiac disorders
Cardiac failure congestive
5 (5% )
Tachycardia
8 (8% )
Ear and labyrinth disorders
Ear pain
6 (6% )
Gastrointestinal disorders
Abdominal pain
14 (14%)
Abdominal pain upper
6 (6% )
Constipation
30 (30%)
Diarrhea
28 (28%)
Dyspepsia
10 (10%)
Dysphagia
5 (5% )
Gastro-esophageal reflux disease
5 (5% )
Nausea
40 (40%)
Oral pain
5 (5% )
Stomatitis
11 (11%)
Toothache
6 (6% )
Vomiting
16 (16%)
General disorders and administration site conditions
Asthenia
15 (15%)
Chest pain
6 (6% )
Chills
16 (16%)
Fatigue
46 (46%)
Mucosal inflammation
9 (9% )
Edema
5 (5% )
Edema peripheral
27 (27%)
Pain
5 (5% )
Pyrexia
36 (36%)
Infections and infestations
Cellulitis
9 (9% )
Oral candidiasis
6 (6% )
Pneumonia
20 (20%)
Sinusitis
6 (6% )
Staphylococcal bacteremia
8 (8% )
Tooth abscess
5 (5% )
Upper respiratory tract infection
10 (10%)
Urinary tract infection
7 (7% )
Injury, poisoning and procedural complications
Contusion
9 (9% )
Investigations
Blood bilirubin increased
6 (6% )
Breath sounds abnormal
5 (5% )
Weight decreased
9 (9% )
Metabolism and nutrition disorders
Anorexia
23 (23%)
Decreased appetite
8 (8% )
Dehydration
8 (8% )
Hyperglycemia
6 (6% )
Hypokalemia
12 (12%)
Hypomagnesemia
5 (5% )
Musculoskeletal and connective tissue disorders
Arthralgia
17 (17%)
Back pain
18 (18%)
Bone pain
6 (6% )
Muscle spasms
7 (7% )
Muscular weakness
5 (5% )
Musculoskeletal pain
5 (5% )
Myalgia
9 (9% )
Pain in extremity
18 (18%)
Nervous system disorders
Dizziness
21 (21%)
Headache
23 (23%)
Psychiatric disorders
Anxiety
9 (9% )
Confusional state
8 (8% )
Depression
9 (9% )
Insomnia
14 (14%)
Respiratory, thoracic and mediastinal disorders
Cough
27 (27%)
Dyspnea
29 (29%)
Epistaxis
13 (13%)
Pharyngolaryngeal pain
8 (8% )
Pleural effusion
5 (5% )
Sinus congestion
5 (5% )
Skin and subcutaneous tissue disorders
Dry skin
8 (8% )
Ecchymosis
9 (9% )
Erythema
5 (5% )
Night sweats
5 (5% )
Petechiae
12 (12%)
Pruritus
9 (9% )
Rash
11 (11%)
Skin lesion
5 (5% )
Vascular disorders
Hypertension
6 (6% )
Hypotension
11 (11%)
* In this single arm study, investigators reported adverse events based on clinical signs and symptoms rather than predefined laboratory abnormalities. Thus, not all laboratory abnormalities were recorded as adverse events.
No overall difference in safety was detected between patients greater than 65 years of age and younger patients in these myelodysplasia trials. No significant gender differences in safety or efficacy were detected. Patients with renal or hepatic dysfunction were not studied. Insufficient numbers of non-white patients were available to draw conclusions in these clinical trials.
Serious adverse reactions that occurred in patients receiving decitabine regardless of causality, not previously reported in Tables 1 and 2 include:- Blood and Lymphatic System Disorders: myelosuppression, splenomegaly.
- Cardiac Disorders: myocardial infarction, cardio-respiratory arrest, cardiomyopathy, atrial fibrillation, supraventricular tachycardia.
- Gastrointestinal Disorders: gingival pain, upper gastrointestinal hemorrhage.
- General Disorders and Administrative Site Conditions: chest pain, catheter site hemorrhage.
- Hepatobiliary Disorders: cholecystitis.
- Infections and Infestations: fungal infection, sepsis, bronchopulmonary aspergillosis, peridiverticular abscess, respiratory tract infection, pseudomonal lung infection, Mycobacterium avium complex infection.
- Injury, Poisoning and Procedural Complications: post procedural pain, post procedural hemorrhage.
- Nervous System Disorders: intracranial hemorrhage.
- Psychiatric Disorders: mental status changes.
- Renal and Urinary Disorders: renal failure, urethral hemorrhage.
- Respiratory, Thoracic and Mediastinal Disorders: hemoptysis, lung infiltration, pulmonary embolism, respiratory arrest, pulmonary mass.
- Allergic Reaction: Hypersensitivity (anaphylactic reaction) to decitabine has been reported in a Phase 2 trial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of decitabine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cases of Sweet's Syndrome (acute febrile neutrophilic dermatosis) have been reported.
-
7 DRUG INTERACTIONS
Drug interaction studies with decitabine have not been conducted. In vitro studies in human liver microsomes suggest that decitabine is unlikely to inhibit or induce cytochrome P450 enzymes. In vitro metabolism studies have suggested that decitabine is not a substrate for human liver cytochrome P450 enzymes. As plasma protein binding of decitabine is negligible (less than 1%), interactions due to displacement of more highly protein bound drugs from plasma proteins are not expected.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on findings from human data, animal studies, and the mechanism of action, decitabine can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1)].
Limited published data on decitabine use throughout the first trimester during pregnancy describe adverse developmental outcomes including major birth defects (structural abnormalities). In animal reproduction studies, administration of decitabine to pregnant mice and rats during organogenesis caused adverse developmental outcomes and was teratogenic, fetotoxic, and embryotoxic starting at doses approximately 7% of the recommended human dose on a mg/m2 basis (see Data). Advise pregnant women of the potential risk to a fetus. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk of major birth defects and miscarriage in the U.S. general population is 2 to 4% and 15 to 20% of clinically recognized pregnancies, respectively.
Data
Human Data
A single published case report of decitabine pregnancy exposure in a 39-year old woman with a hematologic malignancy described multiple structural abnormalities after 6 cycles of therapy in the 18th week of gestation. These abnormalities included holoprosencephaly, absence of nasal bone, mid-facial deformity, cleft lip and palate, polydactyly and rocker-bottom feet. The pregnancy was terminated.
Animal Data
In utero exposure to decitabine causes temporal related defects in the rat and/or mouse, which include growth suppression, exencephaly, defective skull bones, rib/sternabrae defects, phocomelia, digit defects, micrognathia, gastroschisis, micromelia. Decitabine inhibits proliferation and increases apoptosis of neural progenitor cells of the fetal CNS and induces palatal clefting in the developing murine fetus. Studies in mice have also shown that decitabine administration during osteoblastogenesis (day 10 of gestation) induces bone loss in offspring.
In mice exposed to single IP (intraperitoneal) injections (0, 0.9 and 3.0 mg/m2, approximately 2% and 7% of the recommended daily clinical dose, respectively) over gestation days 8, 9, 10 or 11, no maternal toxicity was observed but reduced fetal survival was observed after treatment at 3 mg/m2 and decreased fetal weight was observed at both dose levels. The 3 mg/m2 dose elicited characteristic fetal defects for each treatment day, including supernumerary ribs (both dose levels), fused vertebrae and ribs, cleft palate, vertebral defects, hindlimb defects and digital defects of fore- and hindlimbs.
In rats given a single IP injection of 2.4, 3.6 or 6 mg/m2 (approximately 5, 8, or 13% the daily recommended clinical dose, respectively) on gestation days 9-12, no maternal toxicity was observed. No live fetuses were seen at any dose when decitabine was injected on gestation day 9. A significant decrease in fetal survival and reduced fetal weight at doses greater than 3.6 mg/m2 was seen when decitabine was given on gestation day 10. Increased incidences of vertebral and rib anomalies were seen at all dose levels, and induction of exophthalmia, exencephaly, and cleft palate were observed at 6.0 mg/m2. Increased incidence of foredigit defects was seen in fetuses at doses greater than 3.6 mg/m2. Reduced size and ossification of long bones of the forelimb and hindlimb were noted at 6.0 mg/m2.
The effect of decitabine on postnatal development and reproductive capacity was evaluated in mice administered a single 3 mg/m2 IP injection (approximately 7% the recommended daily clinical dose) on day 10 of gestation. Body weights of males and females exposed in utero to decitabine were significantly reduced relative to controls at all postnatal time points. No consistent effect on fertility was seen when female mice exposed in utero were mated to untreated males. Untreated females mated to males exposed in utero showed decreased fertility at 3 and 5 months of age (36% and 0% pregnancy rate, respectively). Follow up studies indicated that treatment of pregnant mice with decitabine on gestation day 10 was associated with a reduced pregnancy rate resulting from effects on sperm production in the F1-generation.
8.2 Lactation
Risk Summary
There are no data on the presence of decitabine or its metabolites in human milk, the effects on the breastfed child, or the effects on milk production. Because many drugs are excreted in human milk, and because of the potential for serious adverse reactions from decitabine in a nursing child, advise lactating women to avoid breastfeeding during treatment with decitabine and for at least 1 week after the last dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Conduct pregnancy testing of females of reproductive potential prior to initiating decitabine.
Contraception
Females Advise females of reproductive potential to avoid pregnancy and use effective contraception while receiving decitabine and for 6 months following the last dose. Males Advise males with female partners of reproductive potential to use effective contraception while receiving treatment with decitabine and for 3 months following the last dose [see Nonclinical Toxicology (13.1)].
Infertility
Based on findings of decitabine in animals, male fertility may be compromised by treatment with decitabine [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of decitabine in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of patients exposed to decitabine in the controlled clinical trial, 61 of 83 patients were age 65 and over, while 21 of 83 patients were age 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
- 10 OVERDOSAGE
-
11 DESCRIPTION
Decitabine for injection contains decitabine (5-aza-2’-deoxycitidine), an analogue of the natural nucleoside 2’-deoxycytidine. Decitabine is a fine, white to almost white powder with the molecular formula of C8H12N4O4 and a molecular weight of 228.21. Its chemical name is 4-amino-1-(2-deoxy-β-D-erythro-pentofuranosyl)-1,3,5-triazin-2(1H)-one and it has the following structural formula:
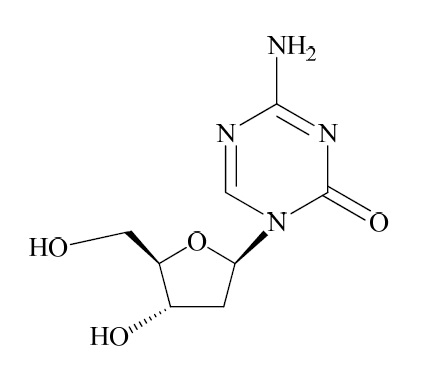
Decitabine is soluble in dimethyl sulfoxide; sparingly soluble in water; slightly soluble in ethanol and water (50:50), in methanol and water (50:50), and in methanol.
Decitabine for injection is a sterile white to almost white lyophilized cake or powder supplied in a clear colorless glass vial. Each 20 mL, single dose, glass vial contains 50 mg decitabine, 68 mg monobasic potassium phosphate (potassium dihydrogen phosphate) and 11.6 mg sodium hydroxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Decitabine is believed to exert its antineoplastic effects after phosphorylation and direct incorporation into DNA and inhibition of DNA methyltransferase, causing hypomethylation of DNA and cellular differentiation or apoptosis. Decitabine inhibits DNA methylation in vitro, which is achieved at concentrations that do not cause major suppression of DNA synthesis. Decitabine-induced hypomethylation in neoplastic cells may restore normal function to genes that are critical for the control of cellular differentiation and proliferation. In rapidly dividing cells, the cytotoxicity of decitabine may also be attributed to the formation of covalent adducts between DNA methyltransferase and decitabine incorporated into DNA. Non-proliferating cells are relatively insensitive to decitabine.
12.2 Pharmacodynamics
Decitabine has been shown to induce hypomethylation both in vitro and in vivo. However, there have been no studies of decitabine-induced hypomethylation and pharmacokinetic parameters.
12.3 Pharmacokinetics
Pharmacokinetic parameters were evaluated in patients. Eleven patients received 20 mg/m2 infused over 1 hour intravenously (treatment Option 2). Fourteen patients received 15 mg/m2 infused over 3 hours (treatment Option 1). PK parameters are shown in Table 3. Plasma concentration-time profiles after discontinuation of infusion showed a biexponential decline. The CL of decitabine was higher following treatment Option 2. Upon repeat doses, there was no systemic accumulation of decitabine or any changes in PK parameters. Population PK analysis (N=35) showed that the cumulative AUC per cycle for treatment Option 2 was 2.3-fold lower than the cumulative AUC per cycle following treatment Option 1.
Table 3 : Mean (CV% or 95% CI) Pharmacokinetic Parameters of Decitabine
Dose
Cmax
(ng/mL)AUC0-∞
(ng·h/mL)
T1/2
(h)CL
(L/h/m2)
AUCCumulative*
(ng·h/mL)
15 mg/m2 3-hr infusion every 8 hours for 3 days (Option 1)†
73.8
(66)163
(62)
0.62
(49)
125
(53)1332
(1010-1730)
20 mg/m2 1-hr infusion daily for 5 days (Option 2)‡
147
(49)
115
(43)0.54
(43)
210
(47)570
(470-700)
*N=35 Cumulative AUC per cycle,†N=14, ‡N=11
The exact route of elimination and metabolic fate of decitabine is not known in humans. One of the pathways of elimination of decitabine appears to be deamination by cytidine deaminase found principally in the liver but also in granulocytes, intestinal epithelium and whole blood.
Specific Populations
Patients with Renal Impairment
There are no data on the use of decitabine in patients with renal impairment; therefore, if decitabine is administered to these patients, monitor them frequently for excessive toxicity.
Patients with Hepatic Impairment
There are no data on the use of decitabine in patients with hepatic impairment; therefore, if decitabine is administered to these patients, monitor them frequently for excessive toxicity.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis and Impairment of Fertility
Carcinogenicity studies with decitabine have not been conducted.
The mutagenic potential of decitabine was tested in several in vitro and in vivo systems. Decitabine increased mutation frequency in L5178Y mouse lymphoma cells, and mutations were produced in an Escherichia coli lac-I transgene in colonic DNA of decitabine-treated mice. Decitabine caused chromosomal rearrangements in larvae of fruit flies.
In male mice given IP injections of 0.15, 0.3 or 0.45 mg/m2 decitabine (approximately 0.3% to 1% the recommended clinical dose) 3 times a week for 7 weeks, decitabine did not affect survival, body weight gain or hematological measures (hemoglobin and WBC counts). Testes weights were reduced, abnormal histology was observed and significant decreases in sperm number were found at doses ≥ 0.3 mg/m2.
In females mated to males dosed with ≥ 0.3 mg/m2 decitabine, pregnancy rate was reduced and preimplantation loss was significantly increased.
-
14 CLINICAL STUDIES
14.1 Controlled Trial in Myelodysplastic Syndrome
A randomized open-label, multicenter, controlled trial evaluated 170 adult patients with myelodysplastic syndromes (MDS) meeting French-American-British (FAB) classification criteria and International Prognostic Scoring System (IPSS) High-Risk, Intermediate-2 and Intermediate-1 prognostic scores. Eighty-nine patients were randomized to decitabine therapy plus supportive care (only 83 received decitabine), and 81 to Supportive Care (SC) alone. Patients with Acute Myeloid Leukemia (AML) were not intended to be included. Of the 170 patients included in the study, independent review (adjudicated diagnosis) found that 12 patients (9 in the decitabine arm and 3 in the SC arm) had the diagnosis of AML at baseline. Baseline demographics and other patient characteristics in the Intent-to-Treat (ITT) population were similar between the 2 groups, as shown in Table 4.
Table 4 : Baseline Demographics and Other Patient Characteristics (ITT)
Demographics and Other Patient Characteristic
Decitabine
N = 89
Supportive Care
N = 81
Age (years)
Mean (±SD)
69±10
67±10
Median (IQR)
70 (65-76)
70 (62-74)
(Range: min-max)
(31-85)
(30-82)
Gender n (%)
Male
59 (66)
57 (70)
Female
30 (34)
24 (30)
Race n (%)
White
83 (93)
76 (94)
Black
4 (4)
2 (2)
Other
2 (2)
3 (4)
Weeks Since MDS Diagnosis
Mean (±SD)
86±131
77±119
Median (IQR)
29 (10-87)
35 (7-98)
(Range: min-max)
(2-667)
(2-865)
Previous MDS Therapy n (%)
Yes
27 (30)
19 (23)
No
62 (70)
62 (77)
RBC Transfusion Status n (%)
Independent
23 (26)
27 (33)
Dependent
66 (74)
54 (67)
Platelet Transfusion Status n (%)
Independent
69 (78)
62 (77)
Dependent
20 (22)
19 (23)
IPSS Classification n (%)
Intermediate-1
28 (31)
24 (30)
Intermediate-2
38 (43)
36 (44)
High Risk
23 (26)
21 (26)
FAB Classification n (%)
RA
12 (13)
12 (15)
RARS
7 (8)
4 (5)
RAEB
47 (53)
43 (53)
RAEB-t
17 (19)
14 (17)
CMML
6 (7)
8 (10)
Patients randomized to the decitabine arm received decitabine intravenously infused at a dose of 15 mg/m2 over a 3-hour period, every 8 hours, for 3 consecutive days. This cycle was repeated every 6 weeks, depending on the patient’s clinical response and toxicity. Supportive care consisted of blood and blood product transfusions, prophylactic antibiotics, and hematopoietic growth factors. The study endpoints were overall response rate (complete response + partial response) and time to AML or death. Responses were classified using the MDS International Working Group (IWG) criteria; patients were required to be RBC and platelet transfusion independent during the time of response. Response criteria are given in Table 5.
Table 5 :Response Criteria for Phase 3 MDS Trial*
Complete Response (CR)
≥ 8 weeks
Bone Marrow
On repeat aspirates:
- <5% myeloblasts
- No dysplastic changes
Peripheral Blood
In all samples during response:
- Hgb >11 g/dL (no transfusions or erythropoietin
- ANC ≥1500/μL (no growth factor)
- Platelets ≥100,000/μL (no thrombopoietic agent)
- No blasts and no dysplasia
Partial Response (PR)
≥ 8 weeks
Bone Marrow
On repeat aspirates:
- ≥ 50% decrease in blasts over pretreatment values
- Improvement to a less advanced MDS FAB classification
Peripheral Blood
Same as for CR
*Cheson BD, Bennett JM, et al. Report of an International Working Group to Standardize Response Criteria for MDS. Blood. 2000; 96:3671-3674.
The overall response rate (CR+PR) in the ITT population was 17% in decitabine-treated patients and 0% in the SC group (p<0.001). (See Table 6.)
The overall response rate was 21% (12/56) in decitabine-treated patients considered evaluable for response (i.e., those patients with pathologically confirmed MDS at baseline who received at least 2 cycles of treatment). The median duration of response (range) for patients who responded to decitabine was 288 days (116-388) and median time to response (range) was 93 days (55-272). All but one of the decitabine-treated patients who responded did so by the fourth cycle. Benefit was seen in an additional 13% of decitabine -treated patients who had hematologic improvement, defined as a response less than PR lasting at least 8 weeks, compared to 7% of SC patients. Decitabine treatment did not significantly delay the median time to AML or death versus supportive care.
Table 6 : Analysis of Response (ITT)
Parameter
Decitabine
N=89
Supportive Care
N=81
Overall Response Rate (CR+PR)*
15 (17%)†
0 (0%)
Complete Response (CR)
8 (9%)
0 (0%)
Partial Response (PR)
7 (8%)
0 (0%)
Duration of Response
Median time to (CR+PR) response - Days (range)
93 (55-272)
NA
Median Duration of (CR+PR) response - Days (range)
288 (116-388)
NA
*In the statistical analysis plan, a p-value of ≤ 0.024 was required to achieve statistical significance.
† p-value <0.001 from two-sided Fisher's Exact Test comparing Decitabine vs. Supportive Care.
All patients with a CR or PR were RBC and platelet transfusion independent in the absence of growth factors.
Responses occurred in patients with an adjudicated baseline diagnosis of AML.14.2 Single-arm Studies in Myelodysplastic Syndrome
Three open-label, single-arm, multicenter studies were conducted to evaluate the safety and efficacy of decitabine in MDS patients with any of the FAB subtypes. In one study conducted in North America, 99 patients with IPSS Intermediate-1, Intermediate-2, or high-risk prognostic scores received decitabine by intravenous infusion at a dose of 20 mg/m2 IV over 1-hour daily, on days 1 to 5 of week 1 every 4 weeks (1 cycle). The results were consistent with the results of the controlled trial and summarized in Table 8.
Table 7 : Baseline Demographics and Other Patient Characteristics (ITT)
Demographic or Other Patient Characteristic
Decitabine
N = 99
Age (years)
Mean (±SD)
71±9
Median (Range: min-max)
72 (34-87)
Gender n (%)
Male
71 (72)
Female
28 (28)
Race n (%)
White
86 (87)
Black
6 (6)
Asian
4 (4)
Other
3 (3)
Days From MDS Diagnosis to First Dose
Mean (±SD)
444±626
Median (Range: min-max)
154 (7-3079)
Previous MDS Therapy n (%)
Yes
27 (27)
No
72 (73)
RBC Transfusion Status n (%)
Independent
33 (33)
Dependent
66 (67)
Platelet Transfusion Status n (%)
Independent
84 (85)
Dependent
15 (15)
IPSS Classification n (%)
Low Risk
1 (1)
Intermediate–1
52 (53)
Intermediate–2
23 (23)
High Risk
23 (23)
FAB Classification n (%)
RA
20 (20)
RARS
17 (17)
RAEB
45 (45)
RAEB-t
6 (6)
CMML
11 (11)
Table 8 : Analysis of Response (ITT)*
Parameter
Decitabine
N=99
Overall Response Rate (CR+PR)
Complete Response (CR)
Partial Response (PR)
16 (16%)
15 (15%)
1 (1%)
Duration of Response
Median time to (CR+PR) response - Days (range)
Median Duration of (CR+PR) response - Days (range)
162 (50-267)
443 (72-722†)
†indicates censored observation
* Cheson BD, Bennett JM, et al. Report of an International Working Group to Standardize Response Criteria for MDS. Blood. 2000; 96:3671-3674.
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Decitabine for injection 50 mg is white to almost white lyophilized cake or powder in 20 mL clear glass vial.
NDC: 0781-3296-80, 50 mg single-dose vial individually packaged in a carton.
Storage
Store vials at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. -
17 PATIENT COUNSELING INFORMATION
Advise women of childbearing potential to avoid pregnancy and to use effective contraception while receiving decitabine and for 6 months after last dose [ see Warnings and Precautions (5.2)].
Advise men not to father a child while receiving treatment with decitabine, and for 3 months after the last dose. Advise men with female partners of childbearing potential to use effective contraception [see Warnings and Precautions (5.2) and Nonclinical Toxicology (13.1)].
Advise patients to avoid breastfeeding while receiving decitabine and for at 1 week after the last dose [see Use in Specific Populations (8.2)]. Advise patients of the risk of myelosuppression and to report any symptoms of infection, anemia, or bleeding to their healthcare provider as soon as possible. Advise patients for the need for laboratory monitoring [see Warnings and Precautions (5.1)].
Manufactured by
MSN Laboratories Private Limited
Telangana 509228, India
for Sandoz Inc.
Princeton, NJ 08540 -
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Principal Display Panel
NDC: 0781-3296-80
Decitabine for Injection
50 mg per vial
Rx Only
For INTRAVENOUS
INFUSION ONLY
WARNING: Cytotoxic Agent
Single-Dose Sterile Vial
Discard Unused Portion
SANDOZ

-
INGREDIENTS AND APPEARANCE
DECITABINE
decitabine injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0781-3296 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DECITABINE (UNII: 776B62CQ27) (DECITABINE - UNII:776B62CQ27) DECITABINE 50 mg in 10 mL Inactive Ingredients Ingredient Name Strength POTASSIUM PHOSPHATE, MONOBASIC (UNII: 4J9FJ0HL51) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0781-3296-80 1 in 1 CARTON 03/01/2020 1 10 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA212265 03/01/2020 Labeler - Sandoz Inc (005387188) Registrant - MSN LABORATORIES PRIVATE LIMITED (650786952) Establishment Name Address ID/FEI Business Operations MSN LABORATORIES PRIVATE LIMITED 650786952 ANALYSIS(0781-3296) , MANUFACTURE(0781-3296)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.