CALCITONIN SALMON spray, metered
Calcitonin Salmon by
Drug Labeling and Warnings
Calcitonin Salmon by is a Prescription medication manufactured, distributed, or labeled by Apotex Corp., Apotex Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CALCITONIN SALMON NASAL SPRAY safely and effectively. See full prescribing information for CALCITONIN SALMON NASAL SPRAY.
CALCITONIN SALMON nasal spray
Initial U.S. Approval: 1975INDICATIONS AND USAGE
Calcitonin Salmon (synthetic origin) Nasal Spray is a calcitonin, indicated for the treatment of postmenopausal osteoporosis in women greater than 5 years postmenopause when alternative treatments are not suitable. Fracture reduction efficacy has not been demonstrated (1.1)
Limitations of Use:
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Nasal Spray: 2200 USP Calcitonin Salmon Units per mL of calcitonin salmon in a 2 mL or 3.7 mL fill glass bottle with attached pump. Each actuation delivers 200 USP Calcitonin Salmon Units of calcitonin salmon (3)
CONTRAINDICATIONS
Hypersensitivity to calcitonin salmon or any of the excipients (4)
WARNINGS AND PRECAUTIONS
- Serious hypersensitivity reactions including anaphylactic shock have been reported. Consider skin testing prior to treatment in patients with suspected hypersensitivity to calcitonin salmon (5.1)
- Hypocalcemia has been reported. Ensure adequate intake of calcium and vitamin D (5.2)
- Nasal adverse reactions, including severe ulceration can occur. Periodic nasal examinations are recommended (5.3)
- Malignancy: A meta-analysis of 21 clinical trials suggests an increased risk of overall malignancies in calcitonin salmon-treated patients (5.4, 6.1)
- Circulating antibodies to calcitonin salmon may develop, and may cause loss of response to treatment (5.5)
ADVERSE REACTIONS
Most common adverse reactions (3% or greater) are rhinitis, epistaxis and other nasal symptoms, back pain, arthralgia, and headache (6)
To report SUSPECTED ADVERSE REACTIONS, contact Apotex Corp. at 1-800-706-5575 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.DRUG INTERACTIONS
- Concomitant use of calcitonin salmon and lithium may lead to a reduction in plasma lithium concentrations due to increased urinary clearance of lithium. The dose of lithium may require adjustment (7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2017
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Treatment of Postmenopausal Osteoporosis
1.2 Important Limitations of Use
2 DOSAGE AND ADMINISTRATION
2.1 Basic Dosing Information
2.2 Priming (Activation) of Pump
2.3 Recommendations for Calcium and Vitamin D Supplementation
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
5.2 Hypocalcemia
5.3 Nasal Adverse Reactions
5.4 Malignancy
5.5 Antibody Formation
5.6 Urine Sediment Abnormalities
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
6.3 Immunogenicity
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Treatment of Postmenopausal Osteoporosis
Calcitonin Salmon (synthetic origin) Nasal Spray is indicated for the treatment of postmenopausal osteoporosis in women greater than 5 years postmenopause. Fracture reduction efficacy has not been demonstrated. Calcitonin Salmon (synthetic origin) Nasal Spray should be reserved for patients for whom alternative treatments are not suitable (e.g., patients for whom other therapies are contraindicated or for patients who are intolerant or unwilling to use other therapies).
1.2 Important Limitations of Use
- Due to the possible association between malignancy and calcitonin salmon use, the need for continued therapy should be re-evaluated on a periodic basis [see Warnings and Precautions (5.4)].
- Calcitonin Salmon (synthetic origin) Nasal Spray has not been shown to increase spinal bone mineral density in early postmenopausal women.
-
2 DOSAGE AND ADMINISTRATION
2.1 Basic Dosing Information
The recommended dose of calcitonin salmon nasal spray is 1 spray (200 USP Calcitonin Salmon Units) per day administered intranasally, alternating nostrils daily.
2.2 Priming (Activation) of Pump
Unopened calcitonin salmon nasal spray should be stored in the refrigerator. Before using the first dose of calcitonin salmon nasal spray, the patient should wait until it has reached room temperature. To prime the pump before it is used for the first time, the bottle should be held upright and the two white side arms of the pump depressed toward the bottle, repeat until a full spray is released. The pump is primed once the first full spray is emitted. To administer, the nozzle should first be carefully placed into the nostril while the patient’s head is in the upright position, then the pump should be firmly depressed toward the bottle. The pump should not be primed before each daily dose.
-
3 DOSAGE FORMS AND STRENGTHS
Calcitonin Salmon (synthetic origin) Nasal Spray consists of one glass bottle with attached pump. The bottle contains 2 mL or 3.7 mL of calcitonin salmon clear solution at a concentration of 2200 USP Calcitonin Salmon Units per mL. A primed pump delivers 0.09 mL (200 USP Calcitonin Salmon Units) calcitonin salmon per actuation.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions have been reported in patients receiving calcitonin salmon nasal spray, e.g., bronchospasm, swelling of the tongue or throat, anaphylaxis and anaphylactic shock. Reports of serious hypersensitivity reactions with injectable calcitonin salmon have also been reported, including reports of death attributed to anaphylaxis. The usual provisions should be made for emergency treatment if such a reaction occurs. Hypersensitivity reactions should be differentiated from generalized flushing and hypotension [see Contraindications (4)].
For patients with suspected hypersensitivity to calcitonin salmon, skin testing should be considered prior to treatment utilizing a dilute, sterile solution of a calcitonin salmon injectable product. Healthcare providers may wish to refer patients who require skin testing to an allergist. A detailed skin testing protocol is available from the Customer Service department of Apotex Corp. (1-800-706-5575).
5.2 Hypocalcemia
Hypocalcemia associated with tetany (i.e., muscle cramps, twitching) and seizure activity has been reported with calcitonin therapy. Hypocalcemia must be corrected before initiating therapy with calcitonin salmon nasal spray. Other disorders affecting mineral metabolism (such as vitamin D deficiency) should also be effectively treated. In patients with these conditions, serum calcium and symptoms of hypocalcemia should be monitored during therapy with calcitonin salmon nasal spray. Use of calcitonin salmon nasal spray is recommended in conjunction with an adequate intake of calcium and vitamin D [see Dosage and Administration (2.3)].
5.3 Nasal Adverse Reactions
Adverse reactions related to the nose including rhinitis and epistaxis have been reported. Development of mucosal alterations may occur. Therefore, periodic nasal examinations with visualization of the nasal mucosa, turbinates, septum and mucosal blood vessels are recommended prior to start of treatment with calcitonin salmon nasal spray, periodically during the course of therapy, and at any time nasal symptoms occur.
Calcitonin salmon nasal spray should be discontinued if severe ulceration of the nasal mucosa occurs, as indicated by ulcers greater than 1.5 mm in diameter or penetrating below the mucosa, or those associated with heavy bleeding. Although smaller ulcers often heal without withdrawal of calcitonin salmon nasal spray, medication should be discontinued temporarily until healing occurs [see Adverse Reactions (6.1)].
5.4 Malignancy
In a meta-analysis of 21 randomized, controlled clinical trials with calcitonin salmon (nasal spray or investigational oral formulations), the overall incidence of malignancies reported was higher among calcitonin salmon-treated patients (4.1%) compared with placebo-treated patients (2.9%). This suggests an increased risk of malignancies in calcitonin salmon-treated patients compared to placebo-treated patients. The benefits for the individual patient should be carefully considered against possible risks [see Adverse Reactions (6.1)].
5.5 Antibody Formation
Circulating antibodies to calcitonin salmon have been reported with calcitonin salmon nasal spray. The possibility of antibody formation should be considered in any patient with an initial response to calcitonin salmon nasal spray who later stops responding to treatment [see Adverse Reactions (6.3)].
5.6 Urine Sediment Abnormalities
Coarse granular casts and casts containing renal tubular epithelial cells were reported in young adult volunteers at bed rest who were given injectable calcitonin salmon to study the effect of immobilization on osteoporosis. There was no other evidence of renal abnormality and the urine sediment normalized after calcitonin salmon was stopped. Periodic examinations of urine sediment should be considered. Urine sediment abnormalities have not been reported in ambulatory volunteers treated with calcitonin salmon nasal spray.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- Hypersensitivity Reactions, including anaphylaxis [see Warnings and Precautions (5.1)]
- Hypocalcemia [see Warnings and Precautions (5.2)]
- Nasal Adverse Reactions [see Warnings and Precautions (5.3)]
- Malignancy [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of calcitonin salmon nasal spray in the treatment of postmenopausal osteoporosis was assessed in 5 randomized, double-blind, placebo controlled trials that enrolled postmenopausal women, aged 45 to 75 years. The duration of the trials ranged from 1 to 2 years. The incidence of adverse reactions reported in studies involving postmenopausal osteoporotic patients chronically exposed to calcitonin salmon nasal spray (N=341) and to placebo nasal spray (N=131), and reported in greater than 3% of calcitonin salmon nasal spray treated patients are presented in the following table. Other than flushing, nausea, possible allergic reactions, and possible local irritative effects in the respiratory tract, a relationship to calcitonin salmon nasal spray has not been established.
Table 1: Adverse Reactions Occurring in at Least 3% of Postmenopausal Patients Treated with Calcitonin Salmon Nasal Spray †Symptom of nose includes: nasal crusts, dryness, redness or erythema, nasal sores, irritation, itching, thick feeling, soreness, pallor, infection, stenosis, runny/blocked, small wound, bleeding wound, tenderness, uncomfortable feeling and sore across bridge of nose. Calcitonin Salmon
Nasal SprayPlacebo
Nasal SprayAdverse Reaction N=341
% of PatientsN=131
% of PatientsRhinitis 12 7 Symptom of Nose† 11 16 Back Pain 5 2 Arthralgia 4 5 Epistaxis 4 5 Headache 3 5 Nasal Adverse Reactions
In all postmenopausal patients treated with calcitonin salmon nasal spray, the most commonly reported nasal adverse reactions included rhinitis (12%), epistaxis (4%), and sinusitis (2%). Smoking did not have a contributory effect on the occurrence of nasal adverse reactions.Adverse reactions reported in 1% to 3% of patients treated with calcitonin salmon nasal spray include: influenza-like symptoms, erythematous rash, arthrosis, myalgia, sinusitis, upper respiratory tract infection, bronchospasm, abdominal pain, nausea, dizziness, paresthesia, abnormal lacrimation, conjunctivitis, lymphadenopathy, infection, and depression.
Malignancy
A meta-analysis of 21 randomized, controlled clinical trials with calcitonin salmon (nasal spray or investigational oral formulations) was conducted to assess the risk of malignancies in calcitonin salmon-treated patients compared to placebo-treated patients. The trials in the meta-analysis ranged in duration from 6 months to 5 years and included a total of 10883 patients (6151 treated with calcitonin salmon and 4732 treated with placebo). The overall incidence of malignancies reported in these 21 trials was higher among calcitonin salmon-treated patients (254/6151 or 4.1%) compared with placebo-treated patients (137/4732 or 2.9%). Findings were similar when analyses were restricted to the 18 nasal spray only trials [calcitonin salmon 122/2712 (4.5%); placebo 30/1309 (2.3%)].The meta-analysis results suggest an increased risk of overall malignancies in calcitonin salmon-treated patients compared to placebo-treated patients when all 21 trials are included and when the analysis is restricted to the 18 nasal spray only trials (see Table 2). It is not possible to exclude an increased risk when calcitonin salmon is administered by the subcutaneous, intramuscular, or intravenous route because these routes of administration were not investigated in the meta-analysis. The increased malignancy risk seen with the meta-analysis was heavily influenced by a single large 5-year trial, which had an observed risk difference of 3.4% [95% CI (0.4%, 6.5%)]. Imbalances in risks were still observed when analyses excluded basal cell carcinoma (see Table 2); the data were not sufficient for further analyses by type of malignancy. A mechanism for these observations has not been identified. Although a definitive causal relationship between calcitonin salmon use and malignancies cannot be established from this meta-analysis, the benefits for the individual patient should be carefully evaluated against all possible risks [see Warnings and Precautions (5.4)].
Table 2: Risk Difference for Malignancies in Calcitonin Salmon-Treated Patients Compared with Placebo-Treated Patients 1 The overall adjusted risk difference is the difference between the percentage of patients who had any malignancy (or malignancy excluding basal cell carcinoma) in calcitonin salmon and placebo treatment groups, using the Mantel-Haenszel (MH) fixed-effect method. A risk difference of 0 is suggestive of no difference in malignancy risks between the treatment groups.
2 The corresponding 95% confidence interval for the overall adjusted risk difference also based on MH fixed-effect method.Patients Malignancies Risk Difference1 (%) 95% Confidence Interval2
(%)All (nasal spray + oral) All 1.0 (0.3, 1.6) All (nasal spray + oral) Excluding basal cell carcinoma 0.5 (-0.1, 1.2) All (nasal spray only) All 1.4 (0.3, 2.6) All (nasal spray only) Excluding basal cell carcinoma 0.8 (-0.2, 1.8) 6.2 Postmarketing Experience
Because postmarketing adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been reported during post-approval use of calcitonin salmon nasal spray.
Allergic / Hypersensitivity Reactions
Serious allergic reactions have been reported in patients receiving calcitonin salmon nasal spray, including anaphylaxis and anaphylactic shock.Hypocalcemia
Hypocalcemia with paresthesia has been reported.Body as a whole
facial or peripheral edemaCardiovascular
hypertension, vasodilatation, syncope, chest painNervous system
dizziness, seizure, visual or hearing impairment, tinnitusRespiratory/Special Senses
cough, bronchospasm, dyspnea, loss of taste/smellSkin
rash/dermatitis, pruritus, alopecia, increased sweatingGastrointestinal
diarrheaNervous system disorders
tremor6.3 Immunogenicity
Consistent with the potentially immunogenic properties of medicinal products containing peptides, administration of calcitonin salmon nasal spray may trigger the development of anti-calcitonin antibodies. In a two-year calcitonin salmon nasal spray clinical study that evaluated immunogenicity, a measurable antibody titer was found in 69% of patients treated with calcitonin salmon nasal spray and 3% of placebo-treated patients. Antibody formation may be associated with a loss of response to treatment [see Warnings and Precautions (5.5)].
The incidence of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of a positive antibody test result may be influenced by several factors, including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of antibodies to calcitonin salmon nasal spray with the incidence of antibodies to other calcitonin-containing products may be misleading.
- Hypersensitivity Reactions, including anaphylaxis [see Warnings and Precautions (5.1)]
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Calcitonin salmon nasal spray is not indicated for use in females of reproductive potential. There are no data with the use of calcitonin salmon nasal spray in pregnant women. In an animal reproduction study, subcutaneous administration of calcitonin-salmon to pregnant rabbits during organogenesis at 418 times the recommended parenteral human dose caused a decrease in fetal birth weights. No adverse developmental outcome was observed in the rat with subcutaneous administration of calcitonin-salmon at 9 times the recommended human parenteral dose based on body surface area (see Data).
Data
Animal Data
Calcitonin salmon has been shown to cause a decrease in fetal birth weights in rabbits when given by subcutaneous injection in doses 4 to 18 times the parenteral dose (of 54 International Units/m2) and 70 to 278 times the intranasal dose recommended for human use based on body surface area.
No embryo/fetal toxicities related to calcitonin salmon nasal spray were reported from maternal subcutaneous daily doses in rats up to 80 International Units/kg/day from gestation day 6 to 15.
8.2 Lactation
Risk Summary
Calcitonin salmon nasal spray is not indicated for use in females of reproductive potential. There is no information on the presence of calcitonin-salmon in human milk, the effects on the breastfed child, or the effects on milk production. Calcitonin has been shown to inhibit lactation in rats.
8.5 Geriatric Use
In a multi-centered, double-blind, randomized clinical study of calcitonin salmon nasal spray, 279 patients were less than 65 years old, while 467 patients were 65 to 74 years old and 196 patients were 75 years old and older. Compared to subjects less than 65 years old, the incidence of nasal adverse reactions (rhinitis, irritation, erythema, and excoriation) was higher in patients over the age of 65, particularly among those over the age of 75. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
-
10 OVERDOSAGE
The pharmacologic actions of calcitonin salmon nasal spray suggest that hypocalcemic tetany could occur in overdose. Therefore, provisions for parenteral administration of calcium should be available for the treatment of overdose.
Single doses of calcitonin salmon nasal spray up to 1600 International Units, doses up to 800 International Units per day for 3 days and chronic administration of doses up to 600 International Units per day have been studied without serious adverse effects.
-
11 DESCRIPTION
Calcitonin is a polypeptide hormone secreted by the parafollicular cells of the thyroid gland in mammals and by the ultimobranchial gland of birds and fish.
Calcitonin Salmon (synthetic origin) Nasal Spray is a synthetic polypeptide of 32 amino acids in the same linear sequence that is found in calcitonin of salmon origin. This is shown by the following graphic formula:

It is provided in a 2 mL or 3.7 mL fill glass bottle as a solution for nasal administration. This is sufficient medication for at least 14 doses (2 mL fill) or 30 doses (3.7 mL fill).
Active Ingredient: calcitonin salmon 2200 USP Calcitonin Salmon Units per mL (corresponding to 200 USP Calcitonin Salmon Units per 0.09 mL actuation).
Inactive Ingredients: benzalkonium chloride, hydrochloric acid (added as necessary to adjust pH), purified water and sodium chloride.
The activity of calcitonin salmon nasal spray is stated in International Units based on bioassay in comparison with the International Reference Preparation of calcitonin salmon for Bioassay, distributed by the National Institute of Biological Standards and Control, Holly Hill, London.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Calcitonin salmon is a calcitonin receptor agonist. Calcitonin salmon acts primarily on bone, but direct renal effects and actions on the gastrointestinal tract are also recognized. Calcitonin salmon appears to have actions essentially identical to calcitonins of mammalian origin, but its potency per mg is greater and it has a longer duration of action.
The actions of calcitonin on bone and its role in normal human bone physiology are still not completely elucidated, although calcitonin receptors have been discovered in osteoclasts and osteoblasts.
12.2 Pharmacodynamics
The information below, describing the clinical pharmacology of calcitonin, has been derived from studies with injectable calcitonin salmon. The mean bioavailability of calcitonin salmon nasal spray is approximately 3% of the injectable calcitonin salmon in healthy subjects and, therefore, the conclusions concerning the clinical pharmacology of this preparation may be different.
Bone
Single injections of calcitonin salmon caused a marked transient inhibition of the ongoing bone resorptive process. With prolonged use, there is a persistent, smaller decrease in the rate of bone resorption. Histologically, this is associated with a decreased number of osteoclasts and an apparent decrease in their resorptive activity.In healthy adults, who have a relatively low rate of bone resorption, the administration of exogenous calcitonin salmon results in decreases in serum calcium within the limits of the normal range. In healthy children and in patients whose bone resorption is more rapid, decreases in serum calcium are more pronounced in response to calcitonin salmon.
Kidney
Studies with injectable calcitonin salmon show increases in the excretion of filtered phosphate, calcium, and sodium by decreasing their tubular reabsorption. Comparable studies have not been conducted with calcitonin salmon nasal spray.Gastrointestinal Tract
Some evidence from studies with injectable preparations suggests that calcitonin salmon may have effects on the gastrointestinal tract. Short-term administration of injectable calcitonin salmon results in marked transient decreases in the volume and acidity of gastric juice and in the volume and the trypsin and amylase content of pancreatic juice. Whether these effects continue to be elicited after each injection of calcitonin salmon during chronic therapy has not been investigated. These studies have not been conducted with calcitonin salmon nasal spray.Calcium Homeostasis
In two clinical studies designed to evaluate the pharmacodynamic response to calcitonin salmon nasal spray, administration of calcitonin salmon 100 to 1600 International Units to healthy volunteers resulted in rapid and sustained decreases within the normal range for both total serum calcium and serum ionized calcium. Single doses of calcitonin salmon greater than 400 International Units did not produce any further biological response to the drug.12.3 Pharmacokinetics
The bioavailability of calcitonin salmon nasal spray relative to intramuscular administration in healthy volunteers is between 3% and 5%. Calcitonin salmon nasal spray is absorbed rapidly by the nasal mucosa with a mean Tmax of about 13 minutes. The terminal half-life of calcitonin salmon has been calculated to be around 18 minutes and no evidence of accumulation was observed with multiple dosing.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
The incidence of pituitary adenomas was increased in rats after one and two years of subcutaneous exposure to synthetic calcitonin salmon. The significance of this finding to humans is unknown because pituitary adenomas are very common in rats as they age, the pituitary adenomas did not transform into metastatic tumors, there were no other clear treatment-related neoplasms, and synthetic calcitonin salmon related neoplasms were not observed in mice after two years of dosing.Rat findings
The only clear neoplastic finding in rats dosed subcutaneously with synthetic calcitonin salmon was an increase in the incidence of pituitary adenomas in male Fisher 344 rats and female Sprague Dawley rats after one year of dosing and male Sprague Dawley rats dosed for one and two years. In female Sprague Dawley rats, the incidence of pituitary adenomas after two years was high in all treatment groups (between 80% and 92% including the control groups) such that a treatment-related effect could not be distinguished from natural background incidence. The lowest dose in male Sprague Dawley rats that developed an increased incidence of pituitary adenomas after two years of dosing (1.7 International Units/kg/day) is approximately 2 times the maximum recommended intranasal dose in humans (200 International Units/day) based on body surface area conversion between rats and humans and a 20-fold conversion factor to account for decreased clinical exposure via the intranasal route. The findings suggest that calcitonin salmon reduced the latency period for development of non-functioning pituitary adenomas.Mouse findings
No carcinogenicity potential was evident in male or female mice dosed subcutaneously for two years with synthetic calcitonin salmon at doses up to 800 International Units/kg/day. The 800 International Units/kg/day dose is approximately 390 times the maximum recommended intranasal dose in humans (200 International Units) based on scaling for body surface area and a 20-fold conversion factor to account for low clinical exposure via the intranasal route.Mutagenesis
Synthetic calcitonin salmon tested negative for mutagenicity using Salmonella typhimurium (5 strains) and Escherichia coli (2 strains), with and without rat liver metabolic activation, and was not clastogenic in a chromosome aberration test in Chinese Hamster V79 cells. There was no evidence that calcitonin salmon was clastogenic in the in vivo mouse micronucleus test.Fertility
Effects of calcitonin salmon on fertility have not been assessed in animals. -
14 CLINICAL STUDIES
Two randomized, placebo-controlled, two-year trials were conducted in 266 postmenopausal women who were greater than 5 years postmenopause with spinal, forearm or femoral bone mineral density (BMD) at least one standard deviation below the normal value for healthy premenopausal women (T-score < -1). In both studies, a total of 144 patients received calcitonin salmon nasal spray 200 International Units or placebo daily. The intent-to-treat population comprised 139 patients who had at least one follow-up BMD measurement. In study 1, patients also received 500 mg daily calcium supplements, while in study 2, patients received no calcium supplementation. The primary endpoint for both studies was percent change in lumbar spine BMD at 2 years. Calcitonin salmon nasal spray increased lumbar vertebral BMD relative to placebo in women with low bone mass who were greater than 5 years post menopause (see Table 3 below).
Table 3: Calcitonin Salmon Nasal Spray: Lumbar Spine Bone Mineral Density In Women Greater Than 5 years Postmenopause With Low Bone Mass ITT: Intent To Treat
IU: International Units
NS: nasal spray
†p-values by parametric testing (2-tailed 2-sample t-test)Lumbar Spine Bone Mineral Density,
Mean Change From Baseline (in %) at Month 24Study 1
(with calcium supplement)
n (ITT) = 100Study 2
(no calcium supplement)
n (ITT) = 39Calcitonin Salmon 200 IU NS daily +1.56 +1.02 Placebo +0.20 -1.85 Treatment Difference +1.36 +2.87 p-value† < 0.05 < 0.005 No effects of calcitonin salmon nasal spray on cortical bone of the forearm or hip were demonstrated.
In clinical studies of postmenopausal osteoporosis, bone biopsy and radial bone mass assessments at baseline and after 26 months of daily injectable calcitonin salmon indicate that calcitonin therapy results in the formation of normal bone.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Calcitonin Salmon (synthetic origin) Nasal Spray is available as a metered dose clear solution in a 2 mL or 3.7 mL fill clear glass bottle that contains 2,200 USP Calcitonin Salmon Units calcitonin salmon per mL. A pump is attached to the bottle. After priming, the pump will deliver 200 USP Calcitonin Salmon Units per activation (0.09 mL per spray) (2 mL fill: NDC: 60505-0823-0; 3.7 mL fill: NDC: 60505-0823-6).Storage and Handling
Store unopened bottle in refrigerator between 2°C to 8°C (36°F to 46°F). Protect from freezing.Store bottle in use at room temperature between 20°C to 25°C (68°F to 77°F) in an upright position, for up to 30 days (2 mL fill) or 35 days (3.7 mL fill). Each bottle contains at least 14 doses (2 mL fill) or 30 doses (3.7 mL fill).
Discard bottle after 14 doses (2 mL fill) or 30 doses (3.7 mL fill).
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Patient Information and Instructions for Use).
- Instruct patients on pump assembly, priming of the pump, and nasal introduction of Calcitonin Salmon (synthetic origin) Nasal Spray. Although instructions for patients are supplied with the individual bottle, procedures for use should be demonstrated to each patient [see Dosage and Administration (2.2)]. Patients should notify their healthcare provider if they develop significant nasal irritation [see Warnings and Precautions (5.3)].
- Inform patients of the potential increase in risk of malignancy [see Warnings and Precautions (5.4)].
- Advise patients to maintain an adequate calcium (at least 1000 mg elemental calcium per day) and vitamin D (at least 400 International Units per day) intake [see Dosage and Administration (2.3)].
- Instruct patients to seek emergency medical help or go to the nearest hospital emergency room right away if they develop any signs or symptoms of a serious allergic reaction.
- Advise patients how to correctly store unopened and opened product [see How Supplied/Storage and Handling (16)]. Advise patients that the bottle should be discarded after 14 doses (2 mL fill) or 30 doses (3.7 mL fill), because after 14 or 30 doses, each spray may not deliver the correct amount of medication even if the bottle is not completely empty.
APOTEX INC.
CALCITONIN SALMON (SYNTHETIC ORIGIN) NASAL SPRAYManufactured by Manufactured for Apotex Inc. Apotex Corp. Toronto, Ontario Weston, FL Canada M9L 1T9 33326 Revised: November 2017
-
INSTRUCTIONS FOR USE
Calcitonin Salmon (synthetic origin)
(kal" si toe' nin)
Nasal Spray
For Nasal Use Only.
Important information about your Calcitonin Salmon (synthetic origin) Nasal Spray:
- A single spray of Calcitonin Salmon (synthetic origin) Nasal Spray contains 1 daily dose of medicine.
- Each Calcitonin Salmon (synthetic origin) Nasal Spray bottle contains the right amount of medicine. The bottle may not be completely filled to the top. This is normal.
- This package contains 1 bottle of Calcitonin Salmon (synthetic origin) Nasal Spray with attached pump.
- Store unopened bottles of Calcitonin Salmon (synthetic origin) Nasal Spray in the refrigerator between 2°C to 8°C (36°F to 46°F). Do not freeze.
- After you open your bottle of Calcitonin Salmon (synthetic origin) Nasal Spray, store it at room temperature between 20°C to 25°C (68°F to 77°F) in an upright position. Do not shake the bottle.
Preparing your Calcitonin Salmon (synthetic origin) Nasal Spray: Step 1. Remove the bottle from your refrigerator and let it reach room temperature. Check the medicine in the bottle to make sure it is clear and colorless without particles. Step 2. Remove the clear plastic dust cap and the blue safety clip from the nasal spray pump. See Figure 1. The safety clip prevents the accidental discharge of the spray in your pocket or purse. 
Step 3. Check to see if your Calcitonin Salmon (synthetic origin) Nasal Spray has been primed. - To make sure you get the right dose of Calcitonin Salmon (synthetic origin) Nasal Spray medicine you must prime each new bottle and pump before you use it for the first time.
- Check to see if the bottle and pump has already been primed by pressing on the pump 1 time. See Figure 2.
- If you see a full spray from the pump, the bottle and pump has already been primed for you.
- If you do not see a full spray, you must prime the bottle and pump.
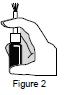
Step 4. Priming your Calcitonin Salmon (synthetic origin) Nasal Spray: - Hold the bottle upright with your pointer finger and middle finger on the 2 side arms of the pump, and your thumb on the bottom of the bottle. Firmly press down on the arms of the pump and press down again if needed until you see a full spray of medicine. See Figure 2.
- Now your Calcitonin Salmon (synthetic origin) Nasal Spray is ready for you to use.
- Do not prime the pump before you use it each day because this will waste your medicine.
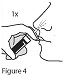
Giving your Calcitonin Salmon (synthetic origin) Nasal Spray dose: Step 5. Insert the nasal spray pump in 1 side of your nose. See Figure 3. - Keep your head upright. Carefully tilt the bottle and place the nose spray pump into 1 side of your nose.

Step 6. Firmly press down on the nose spray pump to release the medicine. See Figure 4. - Give 1 spray of Calcitonin Salmon (synthetic origin) Nasal Spray, 1 time daily, in 1 side of your nose (nostril). Spray your medicine in a different side of your nose each day.
- You do not need to breathe or inhale while you are giving your dose.
- You may not feel the spray inside your nose.
- Some of the medicine may drip out of your nose. This is normal, and you are still getting all of the medicine you need.
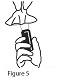
Cleaning your Calcitonin Salmon (synthetic origin) Nasal Spray pump: Step 7. Wipe the nose spray pump with a clean, damp cloth 1 to 2 times a week. See Figure 5. - Dry the nose spray pump with a clean cloth.
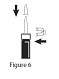
Storing your Calcitonin Salmon (synthetic origin) Nasal Spray: Step 8. Replace the blue safety clip, then replace the protective cap on the nasal spray unit. See Figure 6. - Be careful not to push down on the pump while you are putting the cap back on it.
- Do not refrigerate Calcitonin Salmon (synthetic origin) Nasal Spray between doses.
- Store Calcitonin Salmon (synthetic origin) Nasal Spray upright.
- Do not shake the bottle.
When should I throw away Calcitonin Salmon (synthetic origin) Nasal Spray?
- Unopened, refrigerated bottles can be used until the expiration date stamped on the bottle and box.
- Throw away Calcitonin Salmon (synthetic origin) Nasal Spray after you use 14 doses (2 mL fill) or 30 doses (3.7 mL fill) (sprays).
- Throw away Calcitonin Salmon (synthetic origin) Nasal Spray bottles left at room temperature (opened or unopened) for more than 30 days (2 mL fill) or 35 days (3.7 mL fill).
For more information on Calcitonin Salmon (synthetic origin) Nasal Spray, call Apotex Corp. at 1-800-706-5575.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
APOTEX INC.
CALCITONIN SALMON (SYNTHETIC ORIGIN) NASAL SPRAYManufactured by Manufactured for Apotex Inc. Apotex Corp. Toronto, Ontario Weston, FL Canada M9L 1T9 33326 Revised: November 2017
-
PATIENT INFORMATION
Calcitonin Salmon (synthetic origin)
(kal" si toe' nin)
Nasal Spray
Read this Patient Information before you start using Calcitonin Salmon (synthetic origin) Nasal Spray and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What is Calcitonin Salmon (synthetic origin) Nasal Spray?
Calcitonin Salmon (synthetic origin) Nasal Spray is a prescription medicine used to treat osteoporosis in women more than 5 years after menopause. Calcitonin Salmon (synthetic origin) Nasal Spray should be used for women who cannot use other treatments or who choose not to use other treatments for osteoporosis.
It is not known if Calcitonin Salmon (synthetic origin) Nasal Spray lowers the chance of having bone fractures.
Calcitonin Salmon (synthetic origin) Nasal Spray has not been shown to be effective in women less than 5 years after menopause.
It is not known if Calcitonin Salmon (synthetic origin) Nasal Spray is safe and effective in children under 18 years of age.
Who should not use Calcitonin Salmon (synthetic origin) Nasal Spray?
Do not use Calcitonin Salmon (synthetic origin) Nasal Spray if you:
- are allergic to calcitonin salmon or any of the ingredients in Calcitonin Salmon (synthetic origin) Nasal Spray. See the end of this leaflet for a complete list of ingredients in Calcitonin Salmon (synthetic origin) Nasal Spray.
What should I tell my healthcare provider before using Calcitonin Salmon (synthetic origin) Nasal Spray?
Before you use Calcitonin Salmon (synthetic origin) Nasal Spray, tell your healthcare provider if you:
- have any other medical conditions
- have low calcium levels in your blood
- are pregnant or plan to become pregnant. It is not known if Calcitonin Salmon (synthetic origin) Nasal Spray can harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if Calcitonin Salmon passes into your breast milk. You and your healthcare provider should decide if you will use Calcitonin Salmon (synthetic origin) Nasal Spray or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Especially tell your healthcare provider if you take:
- lithium. Your healthcare provider may need to change your dose of lithium while you use Calcitonin Salmon (synthetic origin) Nasal Spray.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use Calcitonin Salmon (synthetic origin) Nasal Spray?
- For detailed instructions, see the Instructions for Use at the end of this Patient Information leaflet.
- Use Calcitonin Salmon (synthetic origin) Nasal Spray exactly as your healthcare provider tells you to use it.
- Do not use Calcitonin Salmon (synthetic origin) Nasal Spray until your healthcare provider shows you and you understand how to use it correctly.
- Use 1 spray of Calcitonin Salmon (synthetic origin) Nasal Spray, 1 time each day, in 1 nostril (inside your nose).
- Start with 1 spray in your left nostril on your first day, followed by 1 spray in your right nostril on the second day.
- Continue to switch nostrils for your dose each day.
- Your healthcare provider should check your nose before you start using Calcitonin Salmon (synthetic origin) Nasal Spray and often while you are using it.
- Tell your healthcare provider if you start to have discomfort (irritation) in your nose that bothers you while you use Calcitonin Salmon (synthetic origin) Nasal Spray.
- Your health care provider should prescribe calcium and vitamin D to help prevent low calcium levels in your blood while you use Calcitonin Salmon (synthetic origin) Nasal Spray.
- Take your calcium and vitamin D as your healthcare provider tells you to.
- There are 14 doses (2 mL fill) or 30 doses (3.7 mL fill) (sprays) of Calcitonin Salmon (synthetic origin) Nasal Spray in each bottle. After either 14 doses (2 mL fill) or 30 doses (3.7 mL fill), each spray may not give you the right amount of medicine, even if the bottle is not completely empty. Keep track of the number of doses of medicine used from your bottle.
- If you use too much Calcitonin Salmon (synthetic origin) Nasal Spray, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of Calcitonin Salmon (synthetic origin) Nasal Spray?
Calcitonin Salmon (synthetic origin) Nasal Spray may cause serious side effects, including:
-
allergic reactions
Some people have had an allergic reaction when using calcitonin salmon nasal spray. Some reactions may be serious and can be life threatening. Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of these symptoms of an allergic reaction.- trouble breathing
- swelling of your face, throat or tongue
- fast heartbeat
- chest pain
- feel dizzy or faint
-
low calcium levels in your blood (hypocalcemia)
Calcitonin Salmon (synthetic origin) Nasal Spray may lower the calcium levels in your blood. If you have low blood calcium before you start using Calcitonin Salmon (synthetic origin) Nasal Spray, it may get worse during treatment. Your low blood calcium must be treated before you use Calcitonin Salmon (synthetic origin) Nasal Spray. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your healthcare provider right away if you have any of these symptoms of low blood calcium:- numbness or tingling in your fingers, toes, or around your mouth
- do blood tests while you use Calcitonin Salmon (synthetic origin) Nasal Spray
- prescribe calcium and vitamin D to help prevent low calcium levels in your blood while you use Calcitonin Salmon (synthetic origin) Nasal Spray.
-
nose irritation
Irritation of your nose can happen while you are using Calcitonin Salmon (synthetic origin) Nasal Spray, especially if you are over 65 years of age. Call your healthcare provider right way if you have any of these symptoms of nose irritation:- crusting
- dryness
- redness or swelling
- nose sores (ulcers)
- nose bleeds
-
risk of cancer
People who use calcitonin salmon, the medicine in Calcitonin Salmon (synthetic origin) Nasal Spray, may have an increased risk of cancer. -
increase of certain cells (sediment) in your urine
Your healthcare provider should test your urine often while you are using Calcitonin Salmon (synthetic origin) Nasal Spray.
The most common side effects of Calcitonin Salmon (synthetic origin) Nasal Spray include:
- back pain
- muscle aches
- headache
- runny nose
These are not all the possible side effects of Calcitonin Salmon (synthetic origin) Nasal Spray. For more information, ask your healthcare provider or pharmacist.
Tell your healthcare provider right away if you have any side effect that bothers you or does not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store Calcitonin Salmon (synthetic origin) Nasal Spray?
- Store open bottles of Calcitonin Salmon (synthetic origin) Nasal Spray at room temperature between 20°C to 25°C (68°F to 77°F) for 30 days (2 mL fill) or 35 days (3.7 mL fill).
- Store unopened bottles of Calcitonin Salmon (synthetic origin) Nasal Spray in the refrigerator between 2°C to 8°C (36°F to 46°F). Do not freeze.
- Store Calcitonin Salmon (synthetic origin) Nasal Spray bottles in an upright position.
- Safely throw away Calcitonin Salmon (synthetic origin) Nasal Spray in the trash after you have used 14 doses (2 mL fill) or 30 doses (3.7 mL fill) (sprays).
Keep Calcitonin Salmon (synthetic origin) Nasal Spray and all other medicines out of the reach of children.
General information about the safe and effective use of Calcitonin Salmon (synthetic origin) Nasal Spray.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Calcitonin Salmon (synthetic origin) Nasal Spray for a condition for which it was not prescribed. Do not give Calcitonin Salmon (synthetic origin) Nasal Spray to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information summarizes the most important information about Calcitonin Salmon (synthetic origin) Nasal Spray. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Calcitonin Salmon (synthetic origin) Nasal Spray that is written for health professionals.
For more information, call 1-800-706-5575.
What are the ingredients in Calcitonin Salmon (synthetic origin) Nasal Spray?
Active Ingredients: calcitonin salmon
Inactive Ingredients: benzalkonium chloride, hydrochloric acid, purified water and sodium chloride.
-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - PACKAGE CARTON LABEL
APOTEX CORP. NDC: 60505-0823-6
Calcitonin Salmon (synthetic origin) Nasal Spray
(Calcitonin Salmon Nasal Solution, USP)
Rx
30 Doses per bottle
2200 USP Calcitonin Salmon Units/mL (200 USP Calcitonin Salmon Units/spray)
FOR INTRANASAL USE ONLY
REFRIGERATE UNTIL OPENED

-
INGREDIENTS AND APPEARANCE
CALCITONIN SALMON
calcitonin salmon spray, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60505-0823 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCITONIN SALMON (UNII: 7SFC6U2VI5) (CALCITONIN SALMON - UNII:7SFC6U2VI5) CALCITONIN SALMON 200 [iU] Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60505-0823-0 1 in 1 CARTON 12/09/2008 1 14 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 2 NDC: 60505-0823-6 1 in 1 CARTON 12/09/2008 2 30 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076396 12/09/2008 Labeler - Apotex Corp. (845263701) Registrant - Apotex Inc. (209429182) Establishment Name Address ID/FEI Business Operations Richmond Hill 255092496 analysis(60505-0823) , manufacture(60505-0823)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.